Материал: 2019 ESC acute pulmonaryembolism
36 |
ESC Guidelines |
|
|
8.5 Management of pulmonary embolism in patients with cancer
Five RCTs compared LMWH vs. conventional VTE treatment (heparin followed by VKA) in the treatment of VTE in cancer-associated thrombosis.360 364 In 2003, a significant reduction in VTE recurrence was reported with LMWH compared with conventional (VKA) treatment without an increase in bleeding complications.362 In a more recent trial, long-term administration of tinzaparin failed to achieve a statistically significant reduction in overall VTE recurrence over conventional treatment (HR 0.65, 95% CI 0.41 1.03); however, the overall rate of recurrent VTE in the control arm was lower than that previously observed, probably as a result of the recruitment of patients with a lower cancer-specific thrombotic risk.360 Overall, LMWHs were found to decrease the risk of recurrent VTE by 40% with a risk of major bleeding complications similar to that of VKAs.365 Accordingly, LMWHs have become the standard of care. However, these agents are associated with a relevant cost and burden for patients. In addition, the absolute rate of recurrent VTE while on LMWH remains high (7 9%) compared with that observed in noncancer patients with VTE on conventional treatment (1.5 3%).365
NOACs could make the treatment of VTE easier and more convenient in patients with cancer, due to their oral administration in fixed-dose regimens and their lower cost compared with LMWH. However, only 3 9% of patients included in phase III studies with NOACs for the treatment of VTE had concomitant cancer.260,261,312,314,351 A randomized, open-label trial compared edoxaban with LMWH in the secondary prevention of VTE in 1050 patients with cancer-associated thrombosis (mostly symptomatic or asymptomatic PE).366 Edoxaban (60 mg o.d., reduced to 30 mg in subjects with moderate renal impairment, low body weight, or concomitant need for strong inhibitors of glycoprotein-P) was started after 5 days of LMWH and treatment was given for >6 months. Edoxaban was non-inferior to dalteparin in the prevention of VTE recurrence or major bleeding over 12 months after randomization (HR 0.97, 95% CI 0.70 1.36). Major bleeding occurred in 6.9% of the patients in the edoxaban arm and 4.0% in the dalteparin arm (difference in risk 2.9 percentage points, 95% CI 0.1 5.6). This difference appears to have been mainly accounted for by the high rate of bleeding in patients with gastrointestinal cancer allocated to the edoxaban group.366 Similar results were reported by a randomized, open-label pilot trial comparing rivaroxaban with dalteparin in 406 patients with VTE and cancer, 58% of whom had metastases.367 A significant decrease in the risk of recurrent VTE was observed with rivaroxaban (HR 0.43, 95% CI 0.19 0.99). The 6 month cumulative rate of major bleeding, which was mostly gastrointestinal, was 6% (95% CI 3 11%) for rivaroxaban and 4% (95% CI 2 8%) for dalteparin (HR 1.83, 95% CI 0.68 4.96). Corresponding rates of CRNM bleeds were 13%
(95% CI 9 19%) and 4% (95% CI 2 9%), respectively (HR 3.76, 95% CI 1.63 8.69).367
Based on the currently available evidence, as described above, patients with acute PE and cancer, particularly those with gastrointestinal cancer, should be encouraged to continue LMWH for > 3 6 months. This also applies to patients in whom oral treatment is unfeasible due to problems of intake or absorption, and to those with
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
severe renal impairment. In all other cases, especially in patients with an anticipated low risk of bleeding and without gastrointestinal tumours, the choice between LMWH and edoxaban or rivaroxaban is left to the discretion of the physician, and the patient’s preference.
Owing to the high risk for recurrence, patients with cancer should receive indefinite anticoagulation after a first episode of VTE. Although existing evidence is limited, it is conceivable that once cancer is cured the risk for recurrence decreases and anticoagulation can be stopped. However, the definition of cured cancer is not always clear. The risk of recurrence of PE in cancer was assessed in a cohort study of 543 patients and was validated in an independent set of 819 patients.368 The proposed score to predict the risk of recurrence included breast cancer (minus 1 point), Tumour Node Metastasis stage I or II (minus 1 point), and female sex, lung cancer, and previous VTE (plus 1 point each). Patients with a score <0 were at low risk (<4.5%) and those with a score >1 were at high (>19%) risk of VTE
recurrence over the first 6 months.368
After the first 3 6 months, extended anticoagulation may consist of continuation of LMWH or transition to an oral anticoagulant. Two cohort studies have assessed the safety of extended treatment with LMWH (<12 months) in cancer-associated thrombosis.369,370 In both studies, the incidence of bleeding complications was higher in the first months and then reached a plateau that remained unchanged after the sixth month. In the absence of conclusive evidence, the decision to continue with LMWH or to change to VKA or a NOAC should be made on a case-by-case basis after consideration of the success of anticancer therapy, the estimated risk of recurrence of VTE, the bleeding risk, and the preference of the patient. Periodic reassessment of the risk-to-benefit ratio of continued anticoagulant treatment is mandatory.
As mentioned in section 5, venous filters are principally indicated when anticoagulation is impossible due to active haemorrhage or an excessive bleeding risk. However, the risk of VTE recurrence in the absence of anticoagulation is particularly high in patients with cancer, and the insertion of a filter should not delay the initiation of anticoagulation as soon as it is safe to do so. There is no evidence to support the use of venous filters as an adjunct to anticoagulation treatment in patients with cancer.
A number of studies have reported that a proportion of patients presenting with PE in the absence of identifiable risk factors develop cancer within the first year after diagnosis.371 Consequently, the optimal strategy to achieve early diagnosis of these occult cancers was investigated. Two large randomized trials failed to show that comprehensive CT of the abdomen or 18F deoxy-fluoro-glucose positron emission tomography were able to detect more cancers than limited screening in patients with an unprovoked VTE.372,373 Therefore, based on current evidence, the search for occult cancer after an episode of VTE may be restricted to careful history taking, physical examination, basic laboratory tests, and a chest X-ray (if no CTPA was performed to diagnose PE).372,374,375
In patients with cancer, incidental PE should be managed in the same manner as symptomatic PE, whether it involves segmental or more proximal branches, multiple subsegmental vessels, or a single subsegmental vessel in association with detectable DVT.376,377
2019 September 13 on guest by 1093/eurheartj/ehz405/5556136.abstract/doi/10-article-com/eurheartj/advance.oup.https://academic from Downloaded

ESC Guidelines |
37 |
|
|
8.6 Recommendations for the regimen and the duration of anticoagulation after pulmonary embolism in patients with active cancer
|
Recommendations |
Classa |
Levelb |
|
|
|
|
|
|
|
|
|
For patients with PE and cancer, weight-adjusted subcutaneous LMWH should be considered for the first 6 months over |
IIa |
A |
|
|
|
VKAs.360 363 |
|
|
||
|
|
|
|
|
|
|
Edoxaban should be considered as an alternative to weight-adjusted subcutaneous LMWH in patients without gastrointes- |
IIa |
B |
|
|
|
tinal cancer.366 |
|
|
||
|
|
|
|
|
|
|
Rivaroxaban should be considered as an alternative to weight-adjusted subcutaneous LMWH in patients without gastroin- |
IIa |
C |
|
|
|
testinal cancer.367 |
|
|
||
|
|
|
|
|
|
|
For patients with PE and cancer, extended anticoagulation (beyond the first 6 months)c should be considered for an indef- |
IIa |
B |
|
|
|
inite period or until the cancer is cured.378 |
|
|
||
|
|
|
|
|
|
|
In patients with cancer, management of incidental PE in the same manner as symptomatic PE should be considered, if it |
|
|
|
|
|
involves segmental or more proximal branches, multiple subsegmental vessels, or a single subsegmental vessel in associa- |
IIa |
B |
|
|
|
tion with proven DVT.376,377 |
|
|
|
|
|
DVT = deep vein thrombosis; LMWH = low-molecular weight heparin; PE = pulmonary embolism; VKAs = vitamin K antagonists. |
|
|
|
|
|
aClass of recommendation. |
|
|
|
|
|
bLevel of evidence. |
|
|
|
|
|
cRefer to Supplementary Data Table 9 for further guidance on therapeutic decisions after the first 6 months. |
|
|
|
|
9 Pulmonary embolism and pregnancy
9.1 Epidemiology and risk factors for pulmonary embolism in pregnancy
Acute PE remains one of the leading causes of maternal death in high-income countries.379,380 For example, in the UK and Ireland, thrombosis and thromboembolism were the most common causes of direct maternal death (death resulting from the pregnancy rather than pre-existing conditions) in the triennium 2013 15, resulting in 1.13 mortalities per 100 000 maternities (https://www.npeu.ox.ac.uk/mbrrace-uk). VTE risk is higher in pregnant women compared with non-pregnant women of similar age; it increases during pregnancy and reaches a peak during the post-partum period.381 The baseline pregnancy-related risk increases further in the presence of additional VTE risk factors, including in vitro fertilization: in a cross-sectional study derived from a Swedish registry, the HR for VTE following in vitro fertilization was 1.77 (95% CI 1.41 2.23) overall and 4.22 (95% CI 2.46 7.20) during the first trimester.382 Other important and common risk factors include prior VTE, obesity, medical comorbidities, stillbirth, pre-eclampsia, post-partum haemorrhage, and caesarean section; documented risk assessment is therefore essential.383
The recommendations provided in these Guidelines are in line with those included in the 2018 ESC Guidelines on the management of cardiovascular diseases during pregnancy.384
9.2 Diagnosis of pulmonary embolism in pregnancy
9.2.1 Clinical prediction rules and D-dimers
Diagnosis of PE during pregnancy can be challenging as symptoms frequently overlap with those of normal pregnancy. The overall
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
prevalence of confirmed PE is low among women investigated for the disease, between 2 and 7%.385 388 D-dimer levels continuously increase during pregnancy, and levels are above the threshold for VTE ‘rule-out’ in almost one-quarter of pregnant women in the third trimester.390 The results of a multinational prospective management study of 441 pregnant women presenting to emergency departments with clinically suspected PE suggest that a diagnostic strategy based on the assessment of clinical probability, D-dimer measurement, CUS, and CTPA may safely exclude PE in pregnancy.388 In that study, PE exclusion on the basis of a negative D-dimer result (without imaging) was possible in 11.7% of the 392 women with a non-high pre-test probability (Geneva) score, a rate that was reduced to 4.2% in the third trimester.388 A further prospective management study evaluated a combination of a pregnancy-adapted YEARS algorithm with D-dimer levels in 498 women with suspected PE during pregnancy. PE was ruled out without CTPA in women deemed to be at low PE risk according to the combination of the algorithm and D-dimer results. At 3 months, only one woman with PE excluded on the basis of the algorithm developed a popliteal DVT (0.21%, 95% CI 0.04 1.2) and no women developed PE.391
9.2.2 Imaging tests
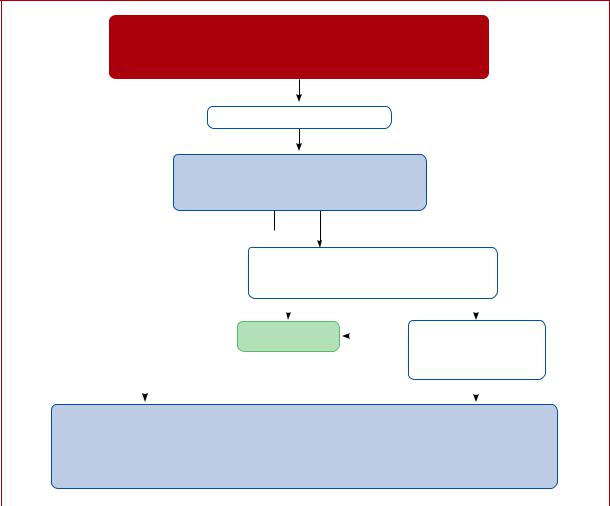
A proposed algorithm for the investigation of suspected PE in women who are pregnant, or <6 weeks post-partum, is shown in Figure 7. Both maternal and foetal radiation exposure are low using modern imaging techniques (Table 12).385,392 398 For V/Q scans and CTPA, foetal radiation doses are well below the
threshold associated with foetal radiation complications (which is 50 100 mSv).399,400 In the past, CTPA has been reported to cause high radiation exposure to the breast; however, CT technology has evolved, and several techniques can now reduce radiation exposure without compromising image quality. These
2019 September 13 on guest by 1093/eurheartj/ehz405/5556136.abstract/doi/10-article-com/eurheartj/advance.oup.https://academic from Downloaded
38 |
ESC Guidelines |
|
|
SUSPECTED PE DURING PREGNANCY
High pretest probability, or intermediate/low probability and positive D-dimer result
Anticoagulate with LMWH
•Chest X-raya
•Compression proximal duplex ultrasound, if symptoms or signs suggestive of DVTb
Proximal DVT not present
|
|
|
|
|
|
|
|
|
|
SPECIFIC INVESTIGATION FOR PE |
|||||||
Proximal |
• If chest X-ray normal => CTPA or perfusion lung scan |
|||||||
• If chest X-ray abnormala => CTPAc |
|
|
|
|||||
DVT present |
|
|
|
|||||
|
|
|
Negative |
|
Indeterminate or positive |
|||
|
PE ruled out |
Negative |
Review by radiologist or |
|||||
|
|
|
||||||
|
|
|
nuclear physician |
|||||
|
|
|
|
|
|
|||
|
|
|
|
|
|
experienced in diagnosis |
||
|
|
|
|
|
|
of PE in pregnancy |
||
|
|
|
|
|
|
|
Positive |
|
|
|
|
|
|
|
|
||
• Continue with LMWH at therapeutic dosed |
|
|
|
|||||
• Assess PE severity and the risk of early deathe |
|
|
|
|||||
• Refer to multidisciplinary team with experience of PE management in pregnancy |
||||||||
• Provide plan to guide management of pregnancy, labour and delivery, postnatal and future care |
||||||||
|
|
|
|
|
|
|
|
©ESC2019 |
Figure 7 Diagnostic workup and management of suspected pulmonary embolism during pregnancy, and up to 6 weeks post-partum.
CTPA = computed tomography pulmonary angiography; CUS = compression ultrasonography; DVT = deep vein thrombosis; LMWH = low-molecular- weight heparin; PE = pulmonary embolism.
aIf chest X-ray abnormal, consider also alternative cause of chest symptoms.
bDVT in pelvic veins may not be ruled out by CUS. If the entire leg is swollen, or there is buttock pain or other symptoms suggestive of pelvic thrombosis, consider magnetic resonance venography to rule out DVT.
cCTPA technique must ensure very low foetal radiation exposure (see Table 12).
dPerform full blood count (to measure haemoglobin and platelet count) and calculate creatinine clearance before administration. Assess bleeding risk and ensure absence of contra-indications.
eSee Table 8.
include reducing the anatomical coverage of the scan,393 reducing the kilovoltage, using iterative reconstructive techniques, and reducing the contrast-monitoring component of the CTPA.392,393 Modern CTPA imaging techniques may therefore expose the maternal breast to median doses as low as 3 4 mGy (Table 12).392 The effect on maternal cancer risk with modern CTPA techniques is negligible (lifetime cancer risk is reportedly increased by a factor of 1.0003 1.0007); avoiding CTPA on the grounds of maternal cancer risk is therefore not justified.394
A normal perfusion scan and a negative CTPA appear equally
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
safe for ruling out PE in pregnancy, as suggested by retrospective series.385,386,402 404 Inconclusive results can be a problem (4 33% of investigations),385,386,405 especially late in pregnancy.405 A recent survey of 24 sites in the UK, representing a population of 15.5 million, revealed a similar rate of inadequate or indeterminate CTPA and scintigraphy scans, suggesting that the initial choice of imaging is best determined by local expertise and resources.406
V/Q SPECT is associated with low foetal and maternal radiation exposure, and has promise in PE diagnosis in pregnancy.407 However,
2019 September 13 on guest by 1093/eurheartj/ehz405/5556136.abstract/doi/10-article-com/eurheartj/advance.oup.https://academic from Downloaded

ESC Guidelines |
39 |
Table 12 Estimated amounts of radiation absorbed in
procedures used to diagnose pulmonary embolism (based on various references385,392–398)
Test |
Estimated |
Estimated maternal |
|
foetal radiation |
radiation exposure |
|
exposure |
to breast tissue |
|
(mGy)a |
(mGy)a |
Chest X-ray |
<0.01 |
<0.1 |
Perfusion lung scan with |
|
|
technetium-99m- |
|
|
labelled albumin |
|
|
Low dose: 40 MBq |
0.02 0.20 |
0.16 0.5 |
High dose: 200 MBq |
0.20 0.60 |
1.2 |
Ventilation lung scan |
0.10 0.30 |
<0.01 |
CTPA |
0.05 0.5 |
3 10 |
CTPA = computed tomography pulmonary angiography; mGy = milligray; MBq = megabecquerel; PE = pulmonary embolism.
aIn this section, absorbed radiation dose is expressed in mGy to reflect the radiation exposure to single organs, or the foetus, as a result of various diagnostic techniques. Compare with Table 6, in which effective radiation dose is expressed in millisieverts to reflect the effective doses of all organs that have been exposed.
further evaluation of this technique is required before its widespread incorporation into diagnostic algorithms. For MRA, the long-term effects of gadolinium contrast on the foetus are not known. In nonpregnant patients, technically inadequate images are frequently obtained and the rate of inconclusive scan results is high.140 Therefore, use of this technique for diagnosing or ruling out PE during pregnancy cannot be recommended at present. Conventional pulmonary angiography involves significantly higher radiation exposure of the foetus (2.2 3.7 mSv) and should be avoided during pregnancy.400
Overdiagnosis of PE is a potential pitfall that can have significant, lifelong implications for a pregnant woman, including the risk of bleeding at the time of delivery, the withholding of oestrogen contraception, and the requirement for thromboprophylaxis during future pregnancies. Consequently, avoiding PE overdiagnosis in pregnancy is as important as not missing a PE diagnosis.
9.3 Treatment of pulmonary embolism in pregnancy
LMWH is the treatment of choice for PE during pregnancy.384 In contrast to VKAs and NOACs, LMWH does not cross the placenta, and consequently does not confer a risk of foetal haemorrhage or teratogenicity. Moreover, while UFH is also safe in pregnancy, LMWH has more predictable pharmacokinetics and a more favourable risk profile.408 411 Although no RCT has evaluated the optimal dose of LMWH for the treatment of PE during pregnancy, currently published data favour similar dosing to non-pregnant patients, either with o.d. or b.i.d. regimens based on early pregnancy weight.408,410 For the majority of patients receiving LMWH treatment for PE during pregnancy,412,413 it remains uncertain whether using serial measurements of plasma anti-activated coagulation factor X activity to guide dosing may be of clinical benefit. It is important to bear in mind that: (i) LMWH has a predictable pharmacokinetic profile, (ii) data on optimal
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
anti-activated coagulation factor levels are lacking, and (iii) the assay itself has limitations.414 In addition, there are no solid data on the clinical benefit vs. harm of frequent, weight-based dose adjustments of LMWH during pregnancy. Thus, anti-activated coagulation factor X monitoring may be reserved for specific high-risk circumstances such as recurrent VTE, renal impairment, and extremes of body weight.
The use of UFH has been associated with heparin-induced thrombocytopenia and bone loss. It remains uncertain whether, and to what extent, the risk of bone loss is increased with LMWH use. In a recent observational cohort study, in which bone mineral density was measured by dual-energy X-ray absorptiometry 4 7 years after the last delivery in 152 women (92 of whom received prolonged LMWH during pregnancy), lumbar spine bone mineral density was similar in LWMH-treated women and controls following adjustment for potential confounders. No osteoporosis or osteoporotic fractures were reported.415
Fondaparinux may be considered if there is an allergy or adverse response to LMWH, although solid data are lacking and minor transplacental passage has been demonstrated.416 VKAs cross the placenta and are associated with a well-defined embryopathy during the first trimester. Administration of VKAs in the third trimester can result in foetal and neonatal haemorrhage, as well as placental abruption. Warfarin may be associated with central nervous system anomalies in the foetus throughout pregnancy. NOACs are contraindicated in pregnant patients.417
The management of labour and delivery requires particular attention. In women receiving therapeutic LMWH, strong consideration should be given to planned delivery in collaboration with the multidisciplinary team to avoid the risk of spontaneous labour while fully anticoagulated. The incidence of spinal haematoma after regional anaesthesia is unknown in pregnant women under anticoagulation treatment. If regional analgesia is considered for a woman receiving therapeutic LMWH, >24 h should have elapsed since the last LMWH dose before insertion of a spinal or epidural needle (assuming normal renal function and including risk assessment at extremes of body weight).
In high-risk situations, for example in patients with recent PE, it is recommended that LMWH be converted to UFH >36 h prior to delivery. The UFH infusion should be stopped 4 - 6 h prior to anticipated delivery and the activated partial thromboplastin time should be normal (i.e. not prolonged) prior to regional anaesthesia.418
Data are limited on the optimal timing of post-partum reinitiation of LMWH.419,420 Timing will depend upon the mode of delivery and an assessment of the thrombotic vs. bleeding risk by a multidisciplinary team. LMWH should not be given for >4 h after removal of the epidural catheter; the decision on timing and dose should consider whether the epidural insertion was traumatic, and take into account the risk profile of the woman. For example, an interim dose of a prophylactic LMWH dose may be considered post-operatively (after caesarean section), once at least 4 h have elapsed since epidural catheter removal, and allowing for an interval of >8 12 h between the prophylactic and the next therapeutic dose. Close collaboration between the obstetrician, the anaesthesiologist, and the attending physician is recommended.
Anticoagulant treatment should be administered for >6 weeks after delivery and with a minimum overall treatment duration of 3 months. LMWH and warfarin can be given to breastfeeding mothers; the use of NOACs is not recommended.417
2019 September 13 on guest by 1093/eurheartj/ehz405/5556136.abstract/doi/10-article-com/eurheartj/advance.oup.https://academic from Downloaded

40 |
ESC Guidelines |
|
|
High-risk, life-threatening PE during pregnancy is a rare, but potentially devastating, event. A recent systematic review included 127 cases of severe PE during pregnancy (and until 6 weeks post-partum) treated with thrombolysis, thrombectomy, and/or ECMO.421 Both highand intermediate-risk PE cases were included, and 23% of women experienced cardiac arrest. Reported survival rates were 94 and 86% following thrombolysis and surgical thrombectomy, respectively; however, these favourable rates may reflect reporting bias. Following thrombolysis, major bleeding occurred in 18 and 58% of cases during pregnancy and in the post-partum period, respectively.421 Finally, foetal deaths occurred in 12 and 20% of the cases following thrombolysis and thrombectomy, respectively.421 Thrombolytic treatment should not be used peri-partum, except in the setting of life-threatening PE. Typically, UFH is used in the acute treatment of high-risk PE.
Although the indications for vena cava filters are similar to those for non-pregnant patients (discussed in section 6), there is limited experience with their use in pregnancy and the risk associated with the procedure may be increased.
Suggestions for the anticoagulation management of PE in specific clinical situations (also) related to pregnancy, for which conclusive evidence is lacking, are presented in Supplementary Data Table 9.
9.3.1 Role of a multidisciplinary pregnancy heart team
A team of multidisciplinary colleagues should collaborate in the planning of ante-, peri-, and post-partum care pathways for women with cardiovascular diseases, including PE. As many members as possible of this team should have expertise in the management of PE during pregnancy and the post-partum period. Jointly agreed, written care pathways should be available (if timelines permit) for effective communication (an example is shown in Figure 7).
9.4 Amniotic fluid embolism
Amniotic fluid embolism (AFE) is a rare condition that occurs during pregnancy or shortly after delivery. It remains one of the leading causes of direct maternal death (i.e. death resulting from the pregnancy rather than from pre-existing conditions) in high-income countries.422 Diagnosis of AFE is challenging, being primarily a clinical diagnosis of exclusion. Awareness of AFE, prompt diagnosis, and aggressive life support are of critical importance. AFE is characterized by unexplained sudden cardiovascular or respiratory deterioration, often accompanied by disseminated intravascular coagulation,422 and occurring during pregnancy or after delivery.423,424 The reported incidence is approximately 2 7 per 100 000 maternities, with a mortality rate of 0.5 6 deaths per 100 000 deliveries.422,425,426 Reported case fatality rates vary, reflecting the challenges in making the diagnosis and the rarity of AFE. In a retrospective Californian study including more than 3.5 million deliveries, a case fatality rate of 13% was reported, as in other US and Canadian studies.425 Similarly, a case fatality rate of 19% was reported in a recent prospective UK population-based study with validated case criteria.422 Recent literature have suggested that risk factors for AFE may include pre-existing cardiac, cerebrovascular, and renal disorders, placenta previa, polyhydramnios, stillbirth, chorioamnionitis, hypertensive disorders, instrumental delivery, and caesarean section.422,425 Management of AFE is supportive, and
.
.
.
.
.
.
.
.
.
.
.
.
9.5 Recommendations for pulmonary embolism in pregnancy
|
Recommendations |
Classa |
Levelb |
|
|
|
|
|
|
|
Diagnosis |
|
|
|
|
Formal diagnostic assessment with validated |
|
|
|
|
methods is recommended if PE is suspected dur- |
I |
B |
|
|
ing pregnancy or in the post-partum period.388,391 |
|
|
|
|
D-dimer measurement and clinical prediction |
|
|
|
|
rules should be considered to rule out PE during |
IIa |
B |
|
|
pregnancy or the post-partum period.388,391 |
|
|
|
|
In a pregnant patient with suspected PE (par- |
|
|
|
|
ticularly if she has symptoms of DVT), venous |
IIa |
B |
|
|
CUS should be considered to avoid unneces- |
|
||
|
|
|
|
|
|
sary irradiation.388 |
|
|
|
|
Perfusion scintigraphy or CTPA (with a low-radi- |
|
|
|
|
ation dose protocol) should be considered to |
|
|
|
|
rule out suspected PE in pregnant women; CTPA |
IIa |
C |
|
|
should be considered as the first-line option if |
|
|
|
|
the chest X-ray is abnormal.385,386 |
|
|
|
|
Treatment |
|
|
|
|
A therapeutic, fixed dose of LMWH based on |
|
|
|
|
early pregnancy body weight is the recom- |
|
|
|
|
mended therapy for PE in the majority of preg- |
I |
B |
|
|
nant women without haemodynamic |
|
|
|
|
instability.408,410 |
|
|
|
|
Thrombolysis or surgical embolectomy should |
|
|
|
|
be considered for pregnant women with high- |
IIa |
C |
|
|
risk PE.421 |
|
|
|
|
Insertion of a spinal or epidural needle is not rec- |
|
|
|
|
ommended, unless >24 h have passed since the |
III |
C |
|
|
last therapeutic dose of LMWH. |
|
|
|
|
Administration of LMWH is not recom- |
|
|
|
|
mended within 4 h of removal of an epidural |
III |
C |
|
|
catheter. |
|
|
|
|
NOACs are not recommended during preg- |
III |
C |
|
|
nancy or lactation. |
|
||
|
|
|
|
|
|
Amniotic fluid embolism |
|
|
|
|
Amniotic fluid embolism should be considered |
|
|
|
|
in a pregnant or post-partum woman with |
|
|
|
|
otherwise unexplained cardiac arrest, sus- |
|
|
|
|
tained hypotension, or respiratory deteriora- |
IIa |
C |
|
|
tion, especially if accompanied by |
|
|
|
|
disseminated intravascular |
|
|
|
|
coagulation.422,425,426 |
|
|
|
CTPA = computed tomography pulmonary angiography; CUS = compression ultrasonography; DVT = deep vein thrombosis; LMWH = low-molecular weight heparin; NOACs = non-vitamin K antagonist oral anticoagulants; PE = pulmonary embolism. aClass of recommendation.
bLevel of evidence.
based on high-quality emergency care following the recognition and diagnosis of the condition, with prompt treatment of bleeding and coagulopathy.423 Awareness of AFE should be integral to the education of involved physicians and to emergency algorithms.
2019 September 13 on guest by 1093/eurheartj/ehz405/5556136.abstract/doi/10-article-com/eurheartj/advance.oup.https://academic from Downloaded