Материал: 2019 ESC acute pulmonaryembolism
ESC Guidelines |
31 |
|
|
©ESC 2019 |
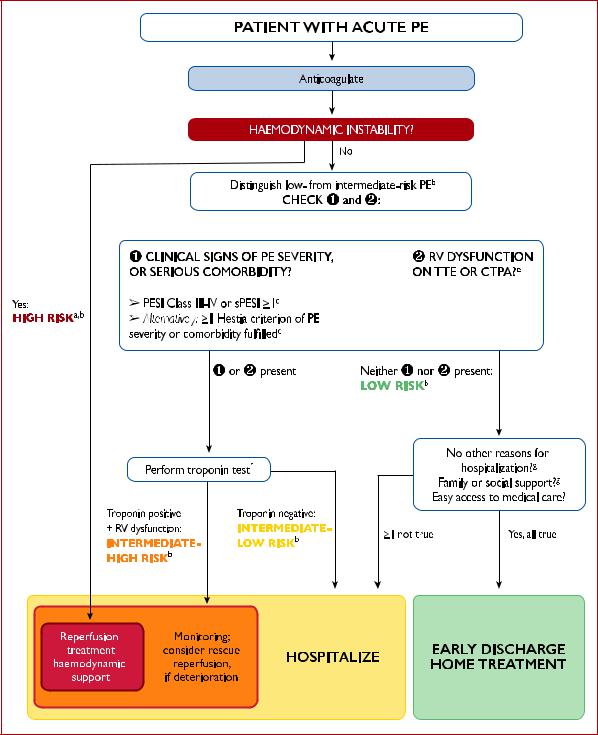
Figure 6 Central Illustration. Risk-adjusted management strategy for acute pulmonary embolism.
CTPA = computed tomography pulmonary angiography/angiogram; PE = pulmonary embolism; PESI = Pulmonary Embolism Severity Index; RV = right ventricular; sPESI = simplified Pulmonary Embolism Severity Index; TTE = transthoracic echocardiogram.
aSee also emergency management algorithm shown in the online Supplementary Data. bRefer to Table 8 for definition of high, intermediate-high-, intermediate-low-, and low-risk PE. cCancer, heart failure and chronic lung disease are included in the PESI and sPESI (Table 7). dSee Supplementary Data Table 12 for the Hestia criteria.
ePrognostically relevant imaging (TTE or CTPA) findings in patients with acute PE, are graphically presented in Figure 3. fA cardiac troponin test may already have been performed during initial diagnostic work-up.
gIncluded in the Hestia criteria.
2019 September 13 on guest by 1093/eurheartj/ehz405/5556136.abstract/doi/10-article-com/eurheartj/advance.oup.https://academic from Downloaded

32 |
ESC Guidelines |
slightly different criteria or combinations thereof were used to ensure the above three requirements.
The Hestia exclusion criteria (Supplementary Data Table 12) represent a checklist of clinical parameters or questions that can be obtained/answered at the bedside. They integrate aspects of PE severity, comorbidity, and the feasibility of home treatment. If the answer to one or more of the questions is ‘yes’, then the patient cannot be discharged early. In a single-arm management trial that used these criteria to select candidates for home treatment, the 3 month rate of recurrent VTE was 2.0% (0.8 4.3%) in patients with acute PE who were discharged within 24 h.317 In a subsequent non-inferiority trial that randomized 550 patients to direct discharge based on the Hestia criteria alone vs. additional NT-proBNP testing and discharge if levels were <500 pg/mL, the primary outcome (30 day PEor bleeding-related mortality, cardiopulmonary resuscitation, or admission to an intensive care unit) was very low in both arms. The results suggest no incremental value of natriuretic-peptide testing in patients who are eligible for home treatment based on the Hestia criteria, although the study was not powered to exclude this possibility.318
The PESI and its simplified form, the sPESI (Table 7), also integrate clinical parameters of PE severity and comorbidity to permit assessment of overall 30 day mortality. Compared with the Hestia criteria, the PESI is more standardized, but it contains a less-comprehensive list of aggravating conditions; moreover, the sPESI excludes all patients with cancer from the low-risk category (compare Table 7 with Supplementary Data Table 12). The PESI was not primarily developed as a tool to select candidates for home treatment, but it has been used—in combination with additional feasibility criteria—in a trial of 344 patients randomized to inpatient vs. outpatient treatment of PE.178 One (0.6%) patient in each treatment group died within 90 days.178
In patients who were included in prospective cohort studies and treated at home, with or without a short hospitalization period, the 3 month rates of thromboembolic recurrence, major bleeding, and death were 1.75, 1.43, and 2.83%, respectively.327
In summary, the currently available evidence indicates that both the Hestia rule and the PESI or sPESI appear capable of reliably identifying patients who are (i) at low PE-related risk, and (ii) free of serious comorbidity. Consequently, either may be used for clinical triage according to local experience and preference. If a PESIor sPESIbased approach is chosen, it must be combined with assessment of the feasibility of early discharge and home treatment; this assessment is already integrated into the Hestia criteria.
A more difficult decision related to immediate or early discharge is whether the exclusion of intermediate-risk PE on clinical grounds alone is adequate, or whether the assessment of RV dysfunction or myocardial injury (see section 5) by an imaging test or a laboratory biomarker is necessary to provide maximal safety for the patient in this ‘vulnerable’ early period. A systematic review and meta-analysis of cohort studies suggested that the prognostic sensitivity is increased further when clinical criteria (e.g. PESI or sPESI) are combined with imaging findings, or laboratory biomarker levels.234 A multicentre prospective management trial tested this hypothesis, investigating the efficacy and safety of early discharge, and ambulatory rivaroxaban treatment, in patients selected by clinical criteria and an absence of RV dysfunction.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
Overall, 20% of the screened unselected patients with PE were included. At the predefined interim analysis of 525 patients (50% of the planned population), the 3 month rate of symptomatic or fatal recurrent VTE was 0.6% (one-sided upper 99.6% CI 2.1%), permitting the early rejection of the null hypothesis and termination of the trial. Major bleeding occurred in six (1.2%) of the patients in the safety population. There were no PE-related deaths.319 In view of the existing evidence—and taking into consideration (i) the catastrophic scenario of early death if a patient with acute PE is falsely judged to be at low risk on clinical grounds alone and discharged ‘too early’ (as described in a prematurely terminated trial328), and (ii) the ease and minimal additional effort of assessing RV size and function at presentation by echocardiography, or on the CTPA performed to diagnose the PE event itself329 (section 5)—it is wise to exclude RV dysfunction and right heart thrombi if immediate or early (within the first 24 48 h) discharge of the patient is planned.
8 Chronic treatment and prevention of recurrence
The aim of anticoagulation after acute PE is to complete the treatment of the acute episode and prevent recurrence of VTE over the long-term. Current drugs and regimens for the initial phase, and the first months of anticoagulant treatment, are described insection 6.
Most of the randomized studies focusing on long-term anticoagulation for VTE have included patients with DVT, with or without PE; only two randomized studies have specifically focused on patients with PE.330,331 The incidence of recurrent VTE does not appear to depend on the clinical manifestation of the first event (i.e. it is similar after PE and after proximal DVT). However, in patients who have had a PE, VTE more frequently recurs as PE, while in patients who have had a DVT, it tends to recur more frequently as DVT.332 As a consequence, the case fatality rate of recurrent VTE in patients who have previously had a PE is twice as high as that of VTE recurrence after DVT.333,334
Landmark clinical trials have evaluated various durations of anticoagulant treatment with VKAs for VTE.330,331,335 337 The findings of these studies permit the following conclusions. First, all patients with PE should receive >3 months of anticoagulant treatment. Second, after withdrawal of anticoagulant treatment, the risk of recurrence is expected to be similar if anticoagulants are stopped after 3 6 months compared with longer treatment periods (e.g. 12 24 months). Third, extended oral anticoagulant treatment reduces the risk for recurrent VTE by <90%, but this benefit is partially offset by the risk of bleeding.
Oral anticoagulants are highly effective in preventing recurrent VTE during treatment, but they do not eliminate the risk of subsequent recurrence after the discontinuation of treatment.330,331 Based on this fact on the one hand, and considering the bleeding risk of anticoagulation treatment on the other, the clinically important question is how to best select candidates for extended or indefinite anticoagulation. Involvement of the patient in the decision-making process is crucial to optimize and maintain treatment adherence.
2019 September 13 on guest by 1093/eurheartj/ehz405/5556136.abstract/doi/10-article-com/eurheartj/advance.oup.https://academic from Downloaded
ESC Guidelines |
33 |
|
|
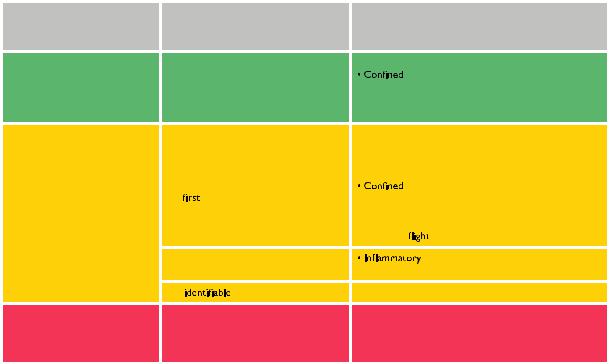
Table 11 Categorization of risk factors for venous thromboembolism based on the risk of recurrence over the longterm
Estimated risk for long-term |
Risk factor category |
Examplesb |
|
|
recurrencea |
for index PEb |
|
|
|
|
Major transient or reversible factors |
• Surgery with general anaesthesia for >30 min |
|
|
|
to bed in hospital (only “bathroom |
|
||
|
associated with >10-fold increased risk |
|
||
Low (<3% per year) |
privileges”) for ≥3 days due to an acute illness, or acute |
|
||
for the index VTE event (compared to |
|
|||
|
exacerbation of a chronic illness |
|
||
|
patients without the risk factor) |
|
||
|
• Trauma with fractures |
|
||
|
|
|
|
|
|
|
|
• Minor surgery (general anaesthesia for <30 min) |
|
|
|
|
• Admission to hospital for <3 days with an acute illness |
|
|
Transient or reversible factors |
• Oestrogen therapy/contraception |
|
|
|
• Pregnancy or puerperium |
|
||
|
associated with ≤10-fold increased risk |
to bed out of hospital for ≥3 days with |
|
|
|
for |
(index) VTE |
an acute illness |
|
Intermediate (3–8% per year) |
|
|
• Leg injury (without fracture) associated with reduced |
|
|
|
|
mobility for ≥3 days |
|
|
|
|
• Long-haul |
|
|
Non-malignant persistent risk factors |
bowel disease |
|
|
|
• Active autoimmune disease |
|
||
|
|
|
|
|
|
No |
risk factor |
|
|
High (>8% per year) |
|
|
• Active cancer |
2019 |
|
|
• One or more previous episodes of VTE in the absence |
||
|
|
|
|
|
|
|
|
of a major transient or reversible factor |
©ESC |
|
|
|
• Antiphospholipid antibody syndrome |
|
|
|
|
|
|
PE = pulmonary embolism; VTE = venous thromboembolism.
aIf anticoagulation is discontinued after the first 3 months (based on data from Baglin et al.340 and Iorio et al.341).
bThe categorization of risk factors for the index VTE event is in line with that proposed by the International Society on Thrombosis and Haemostasis.338 The present Guidelines avoid terms such as ‘provoked’, ‘unprovoked’, or ‘idiopathic’ VTE.
8.1 Assessment of venous thromboembolism recurrence risk
The risk for recurrent VTE after discontinuation of treatment is related to the features of the index PE (or, in the broader sense, VTE) event. A study, which followed patients after a first episode of acute PE, found that the recurrence rate after discontinuation of treatment was 2.5% per year after PE associated with transient risk factors, compared with 4.5% per year after PE occurring in the absence of known cancer, known thrombophilia, or any transient risk factor.331 Similar observations were made in other prospective studies in patients with DVT.337 Advancing the concept further, randomized anticoagulation trials over the past 15 years, which have focused on secondary VTE prevention, have classified patients into distinct groups based on their risk of VTE recurrence after discontinuation of anticoagulant treatment. In general, these groups are: (i) patients in whom a strong (major) transient or reversible risk factor, most commonly major surgery or trauma, can be identified as being responsible for the acute (index) episode; (ii) patients in whom the index episode might be partly explained by the presence of a weak (minor) transient or reversible risk factor, or if a non-malignant risk factor for thrombosis persists; (iii) patients in whom the index episode occurred in the
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
absence of any identifiable risk factor (the present Guidelines avoid terms such as ‘unprovoked’ or ‘idiopathic’ VTE); (iv) patients with one or more previous episodes of VTE, and those with a major persistent pro-thrombotic condition such as antiphospholipid antibody syndrome; and (v) patients with active cancer.338
Table 11 shows examples of transient/reversible and persistent risk factors for VTE, classified by the risk of long-term recurrence. As active cancer is a major risk factor for recurrence of VTE, but also for bleeding while on anticoagulant treatment,339 section 8.4 is specifically dedicated to the management of PE in patients with cancer.
Overall, assessment of the VTE recurrence risk after acute PE, in the absence of a major transient or reversible risk factor, is a complex issue. Beyond the examples listed in Table 11, patients who are carriers of some forms of hereditary thrombophilia, notably those with confirmed deficiency of antithrombin, protein C, or protein S, and patients with homozygous factor V Leiden or homozygous prothrombin G20210A mutation, are often candidates for indefinite anticoagulant treatment after a first episode of PE occurring in the absence of a major reversible risk factor. In view of these possible implications, testing for thrombophilia (including antiphospholipid
2019 September 13 on guest by 1093/eurheartj/ehz405/5556136.abstract/doi/10-article-com/eurheartj/advance.oup.https://academic from Downloaded

34 |
ESC Guidelines |
antibodies and lupus anticoagulant)342 may be considered in patients in whom VTE occurs at a young age (e.g. aged <50 years) and in the absence of an otherwise identifiable risk factor, especially when this occurs against the background of a strong family history of VTE. In such cases, testing may help to tailor the regimen and dose of the anticoagulant agent over the long-term. On the other hand, no evidence of a clinical benefit of extended anticoagulant treatment is currently available for carriers of heterozygous factor V Leiden or prothrombin 20210A mutation.
A number of risk prediction models have been developed for the assessment of the risk of recurrence in an individual patient (Supplementary Data Table 13).343,344 The clinical value and, in particular, the possible therapeutic implications of these models in the NOAC era are unclear.
8.2 Anticoagulant-related bleeding risk
Incidence estimates from cohort studies conducted more than 15 years ago reported an 3% annual incidence of major bleeding in patients treated with VKAs.345 Meta-analyses of phase III studies focusing on the first 3 - 12 months of anticoagulant treatment showed an 40% reduction in the risk for major bleeding with NOACs compared with VKAs.346 The risk of major bleeding is higher in the first month of anticoagulant treatment, and then declines and remains stable over time. Based on currently available evidence, risk factors include: (i) advanced age (particularly >75 years); (ii) previous bleeding (if not associated with a reversible or treatable cause) or anaemia; (iii) active cancer; (iv) previous stroke, either haemorrhagic or ischaemic; (v) chronic renal or hepatic disease; (vi) concomitant antiplatelet therapy or non-steroidal anti-inflammatory drugs (to be avoided, if possible); (vii) other serious acute or chronic illness; and (viii) poor anticoagulation control.
Existing bleeding risk scores and their current validation status are reviewed in Supplementary DataTable 14. The patient’s bleeding risk should be assessed, either by implicit judgement after evaluating individual risk factors or by the use of a bleeding risk score, at the time of initiation of anticoagulant treatment. It should be reassessed periodically (e.g. once a year in patients at low risk, and every 3 or 6 months in patients at high risk for bleeding). Bleeding risk assessment should be used to identify and treat modifiable bleeding risk factors, and it may influence decision-making on the duration and regimen/dose of anticoagulant treatment after acute PE.
8.3 Regimens and treatment durations with non-vitamin K antagonist oral anticoagulants, and with other non-vitamin K antagonist antithrombotic drugs
All patients with PE should be treated with anticoagulants for >3
months.347 Beyond this period, the balance between the risk of VTE recurrence and that of bleeding, which has been used to select candidates for extended anticoagulation after a first VTE event in the VKA era, is currently being revisited based on the lower bleeding rates with NOACs. However, despite the improved safety of these drugs compared with VKAs, treatment
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
with NOACs is not without risk. Phase III clinical trials on the extended treatment of VTE have shown that the rate of major bleeding may be 1%, and that of clinically relevant non-major (CRNM) bleeding as high as 6%. Bleeding rates may be higher in everyday clinical practice.
The NOAC trials that focused on extended VTE treatment are summarized in Supplementary Data Table 15. In all studies, patients with PE made up approximately one-third of the entire study population, while the remaining two-thirds were patients with proximal DVT but no clinically overt PE. Patients needed to have completed the initial and long-term anticoagulation phase to be included in the extended studies.
Dabigatran was compared with warfarin or placebo in two different studies (Supplementary Data Table 15).350 In these studies, dabigatran was non-inferior to warfarin for the prevention of confirmed recurrent symptomatic VTE or VTE-related death, and more effective than placebo for the prevention of symptomatic recurrent VTE or unexplained death.350 The rate of major bleeding was 0.9% with dabigatran compared to 1.8% with warfarin (HR 0.52, 95% CI 0.27 1.02).350
Rivaroxaban was compared with placebo or aspirin in two different studies in patients who had completed 6 12 months of anticoagulation treatment for a first VTE event (Supplementary Data Table 15). Treatment with rivaroxaban [20 mg once a day (o.d.)] reduced recurrent VTE by 80%, with a 6.0% incidence of major or CRNM bleeding as compared to 1.2% with placebo.351 Rivaroxaban given at a dose of 20 or 10 mg o.d. was compared with aspirin (100 mg o.d.) in 3365 patients.352 Both doses of rivaroxaban reduced symptomatic recurrent fatal or non-fatal VTE by 70% in comparison with aspirin. No significant differences in the rates of major or
CRNM bleeding were shown between either dose of rivaroxaban and aspirin.352
Patients with VTE were randomized to receive two different doses of apixaban [2.5 or 5 mg twice a day (bis in die: b.i.d.)] or placebo after
6 12 |
months of initial anticoagulation (Supplementary Data |
|
353 |
||
Table 15). |
Both doses of apixaban reduced the incidence of symp- |
|
tomatic recurrent VTE or death from any cause compared with placebo, with no safety concerns.353
Patients at high bleeding risk—based on the investigator’s judgement, the patient’s medical history, and the results of laboratory examinations—were excluded from the extension studies mentioned above; this was also the case for studies on extended anticoagulation with VKAs.330,331 This fact should be taken into account during triage of a patient for extended anticoagulation with one of the above regimens.
In a randomized, open-label study in high-risk patients with antiphospholipid syndrome (testing triple positive for lupus anticoagulant, anticardiolipin, and anti-b2-glycoprotein I), rivaroxaban was associated with an increased rate of thromboembolic and major bleeding events compared with warfarin (HR for the composite primary outcome 6.7; 95% CI 1.5 30.5).354 At present, NOACs are not an alternative to VKAs for patients with antiphospholipid syndrome.
In two trials with a total of 1224 patients, extended therapy with aspirin (after termination of standard oral anticoagulation) was
2019 September 13 on guest by 1093/eurheartj/ehz405/5556136.abstract/doi/10-article-com/eurheartj/advance.oup.https://academic from Downloaded

ESC Guidelines |
35 |
associated with a 30 35% reduction in the risk of recurrence compared with placebo (Supplementary Data Table 15).355,356 However, more recently, another trial demonstrated the superiority of anticoagulation with rivaroxaban, either 20 or 10 mg o.d., over aspirin for secondary prophylaxis of VTE recurrence.352
A randomized, placebo controlled study evaluated sulodexide (2250 lipasemic unit capsules b.i.d.) for the prevention of recurrence
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
in 615 patients with a first VTE event without an identifiable risk factor, who had completed 3 12 months of oral anticoagulant treatment (Supplementary Data Table 15).357 Sulodexide reduced the risk of recurrence by 50% with no apparent increase in bleeding events. However, only 8% of patients in this study had PE as the index VTE event.357
8.4 Recommendations for the regimen and duration of anticoagulation after pulmonary embolism in patients without cancer
|
Recommendations |
Classa |
Levelb |
|
|
|
|
|
|
|
|
|
Therapeutic anticoagulation for > 3 months is recommended for all patients with PE.347 |
I |
A |
|
|
|
Patients in whom discontinuation of anticoagulation after 3 months is recommended |
|
|
|
|
|
For patients with first PE/VTE secondary to a major transient/reversible risk factor, discontinuation of therapeutic oral |
I |
B |
|
|
|
anticoagulation is recommended after 3 months.331,340,341 |
|
|
||
|
|
|
|
|
|
|
Patients in whom extension of anticoagulation beyond 3 months is recommended |
|
|
|
|
|
Oral anticoagulant treatment of indefinite duration is recommended for patients presenting with recurrent VTE (that is, |
I |
B |
|
|
|
with at least one previous episode of PE or DVT) not related to a major transient or reversible risk factor.358 |
|
|
||
|
|
|
|
|
|
|
Oral anticoagulant treatment with a VKA for an indefinite period is recommended for patients with antiphospholipid anti- |
I |
B |
|
|
|
body syndrome.359 |
|
|
||
|
|
|
|
|
|
|
Patients in whom extension of anticoagulation beyond 3 months should be consideredc,d |
|
|
|
|
|
Extended oral anticoagulation of indefinite duration should be considered for patients with a first episode of PE and no |
IIa |
A |
|
|
|
identifiable risk factor.330,331,347,351 353 |
|
|
||
|
|
|
|
|
|
|
Extended oral anticoagulation of indefinite duration should be considered for patients with a first episode of PE associated |
IIa |
C |
|
|
|
with a persistent risk factor other than antiphospholipid antibody syndrome.330,352,353 |
|
|
||
|
|
|
|
|
|
|
Extended oral anticoagulation of indefinite duration should be considered for patients with a first episode of PE associated |
IIa |
C |
|
|
|
with a minor transient or reversible risk factor.330,331,352 |
|
|
||
|
|
|
|
|
|
|
NOAC dose in extended anticoagulatione |
|
|
|
|
|
If extended oral anticoagulation is decided after PE in a patient without cancer, a reduced dose of the NOACs apixaban |
IIa |
A |
|
|
|
(2.5 mg b.i.d.) or rivaroxaban (10 mg o.d.) should be considered after 6 months of therapeutic anticoagulation.352,353 |
|
|
||
|
|
|
|
|
|
|
Extended treatment with alternative antithrombotic agents |
|
|
|
|
|
In patients who refuse to take or are unable to tolerate any form of oral anticoagulants, aspirin or sulodexide may be con- |
IIb |
B |
|
|
|
sidered for extended VTE prophylaxis.355 357 |
|
|
||
|
|
|
|
|
|
|
Follow-up of the patient under anticoagulation |
|
|
|
|
|
In patients who receive extended anticoagulation, it is recommended that their drug tolerance and adherence, hepatic and |
I |
C |
|
|
|
renalf function, and bleeding risk be reassessed at regular intervals.259 |
|
|
||
|
|
|
|
|
b.i.d. = bis in die (twice a day); DVT = deep vein thrombosis; NOAC(s) = non-vitamin K antagonist oral anticoagulant(s); o.d. = omni die (once a day); PE = pulmonary embolism; VKA = vitamin K antagonist; VTE = venous thromboembolism.
aClass of recommendation. bLevel of evidence.
cThe patient’s bleeding risk should be assessed (see Supplementary Data Table 14 for prediction models) to identify and treat modifiable bleeding risk factors, and it may influence decision-making on the duration and regimen/dose of anticoagulant treatment.
dRefer to Supplementary Data Table 9 for therapeutic decisions in specific clinical situations.
eIf dabigatran or edoxaban is chosen for extended anticoagulation after PE, the dose should remain unchanged, as reduced-dose regimens were not investigated in dedicated extension trials.313,350
fEspecially for patients receiving NOACs.
2019 September 13 on guest by 1093/eurheartj/ehz405/5556136.abstract/doi/10-article-com/eurheartj/advance.oup.https://academic from Downloaded