Материал: 2019 ESC acute pulmonaryembolism

26 |
ESC Guidelines |
was no difference between the two types of reperfusion treatment regarding 30 day mortality (15 and 13%, respectively), but thrombolysis was associated with a higher risk of stroke and reintervention at 30 days. No difference was found in terms of 5 year actuarial survival, but thrombolytic therapy was associated with a higher rate of recurrent PE requiring readmission compared with surgery (7.9 vs. 2.8%). However, the two treatments were not randomly allocated in this observational retrospective study, and the patients referred for surgery may have been selected. An analysis of the Society of Thoracic Surgery Database with multicentre data collection, including 214 patients submitted for surgical embolectomy for high- (n = 38) or intermediate-risk (n = 176) PE, revealed an in-hospital mortality rate of 12%, with the worst outcome (32%) in the group experiencing pre-operative cardiac arrest.299
Recent experience appears to support combining ECMO with surgical embolectomy, particularly in patients with high-risk PE with or without the need for cardiopulmonary resuscitation. Among patients who presented with intermediate-risk PE (n = 28), high-risk PE without cardiac arrest (n = 18), and PE with cardiac arrest (n = 9), the in-hospital and 1 year survival rates were 93 and 91%, respectively.300
6.4 Multidisciplinary pulmonary embolism teams
The concept of multidisciplinary rapid-response teams for the management of ‘severe’ (high-risk and selected cases of intermediaterisk) PE emerged in the USA, with increasing acceptance by the medical community and implementation in hospitals in Europe and worldwide. Set-up of PE response teams (PERTs) is encouraged, as they address the needs of modern systems-based healthcare.301 A PERT brings together a team of specialists from different disciplines including, for example, cardiology, pulmonology, haematology, vascular medicine, anaesthesiology/intensive care, cardiothoracic surgery, and (interventional) radiology. The team convenes in real time (face-to- face or via web conference) to enhance clinical decision-making. This allows the formulation of a treatment plan and facilitates its immediate implementation.301 The exact composition and operating mode of a PERT are not fixed, depending on the resources and expertise available in each hospital for the management of acute PE.
6.5 Vena cava filters
The aim of vena cava interruption is to mechanically prevent venous clots from reaching the pulmonary circulation. Most devices in current use are inserted percutaneously and can be retrieved after several weeks or months, or left in place over the long-term, if needed. Potential indications include VTE and absolute contraindication to anticoagulant treatment, recurrent PE despite adequate anticoagulation, and primary prophylaxis in patients with a high risk of VTE. Other potential indications for filter placement, including freefloating thrombi, have not been confirmed in patients without contraindications to therapeutic anticoagulation.
Only two phase III randomized trials have compared anticoagulation with or without vena cava interruption in patients with proximal DVT, with or without associated PE.302 304 In the Prevention of Recurrent Pulmonary Embolism by Vena Cava Interruption (PREPIC) study, insertion of a permanent vena cava
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
filter was associated with a significant reduction in the risk of recurrent PE and a significant increase in the risk of DVT, without a significant difference in the risk of recurrent VTE or death.303,304 The PREPIC-2 trial randomized 399 patients with PE and venous thrombosis to receive anticoagulant treatment, with or without a
retrievable vena cava filter. In this study, the rate of recurrent VTE was low in both groups and did not differ between groups.302 A systematic review and meta-analysis of published reports on the efficacy and safety of vena cava filters included 11 studies, with a total of 2055 patients who received a filter vs. 2149 controls.305 Vena cava filter placement was associated with a 50% decrease in the incidence of PE and an 70% increase in the risk of DVT over time. Neither all-cause mortality nor PE-related mortality differed between patients with or without filter placement.
The broad indication for placement of a venous filter in patients with recent (<1 month) proximal DVT and an absolute contraindication to anticoagulant treatment is based mainly on the perceived high risk of recurrent PE in this setting, and the lack of other treatment options.
Complications associated with vena cava filters are common and can be serious. A systematic literature review revealed penetration of the venous wall in 1699 (19%) of 9002 procedures; of these cases, 19% showed adjacent organ involvement and >8% were sympto-
matic.306 Lethal complications were rare (only two cases), but 5% of the patients required major interventions such as surgical removal of the filter, endovascular stent placement or embolization, endovascu-
6.6 Recommendations for acute-phase treatment of high-risk pulmonary embolisma
|
Recommendations |
Classb |
Levelc |
|
|
|
|
|
|
|
It is recommended that anticoagulation with |
|
|
|
|
UFH, including a weight-adjusted bolus injec- |
I |
C |
|
|
tion, be initiated without delay in patients with |
|
||
|
|
|
|
|
|
high-risk PE. |
|
|
|
|
Systemic thrombolytic therapy is recom- |
I |
B |
|
|
mended for high-risk PE. 282 |
|
||
|
|
|
|
|
|
Surgical pulmonary embolectomy is recom- |
|
|
|
|
mended for patients with high-risk PE, in whom |
I |
C |
|
|
thrombolysis is contraindicated or has failed.d 281 |
|
|
|
|
Percutaneous catheter-directed treatment |
|
|
|
|
should be considered for patients with high- |
IIa |
C |
|
|
risk PE, in whom thrombolysis is contraindi- |
|
||
|
|
|
|
|
|
cated or has failed.d |
|
|
|
|
Norepinephrine and/or dobutamine should be |
IIa |
C |
|
|
considered in patients with high-risk PE. |
|
||
|
|
|
|
|
|
ECMO may be considered, in combination with |
|
|
|
|
surgical embolectomy or catheter-directed treat- |
IIb |
C |
|
|
ment, in patients with PE and refractory circula- |
|
||
|
|
|
|
|
|
tory collapse or cardiac arrest.d 252 |
|
|
|
ECMO = extracorporeal membrane oxygenation; PE = pulmonary embolism; UFH = unfractionated heparin.
aSee Table 4 for definition of high-risk PE. After haemodynamic stabilization of the patient, continue with anticoagulation treatment as in intermediateor low-risk PE (section 6.7).
bClass of recommendation. cLevel of evidence.
dIf appropriate expertise and resources are available on-site.
2019 September 13 on guest by 1093/eurheartj/ehz405/5556136.abstract/doi/10-article-com/eurheartj/advance.oup.https://academic from Downloaded
ESC Guidelines |
27 |
|
|
6.7 Recommendations for acute-phase treatment of intermediateor low-risk pulmonary embolism
|
Recommendations |
Classa |
Levelb |
|
|
|
|
|
|
|
Initiation of anticoagulation |
|
|
|
|
Initiation of anticoagulation is recommended |
|
|
|
|
without delay in patients with high or inter- |
I |
C |
|
|
mediate clinical probability of PE,c while diag- |
|||
|
|
|
|
|
|
nostic workup is in progress. |
|
|
|
|
If anticoagulation is initiated parenterally, |
|
|
|
|
LMWH or fondaparinux is recommended |
I |
A |
|
|
(over UFH) for most patients.262,309 311 |
|
|
|
|
When oral anticoagulation is started in a |
|
|
|
|
patient with PE who is eligible for a NOAC |
|
|
|
|
(apixaban, dabigatran, edoxaban, or rivaroxa- |
I |
A |
|
|
ban), a NOAC is recommended in preference |
|
|
|
|
to a VKA.260,261,312 314 |
|
|
|
|
When patients are treated with a VKA, over- |
|
|
|
|
lapping with parenteral anticoagulation is rec- |
I |
A |
|
|
ommended until an INR of 2.5 (range |
|||
|
|
|
|
|
|
2.0 3.0) is reached.315,316 |
|
|
|
|
NOACs are not recommended in patients with |
|
|
|
|
severe renal impairment,d during pregnancy and |
III |
C |
|
|
lactation, and in patients with antiphospholipid |
|||
|
|
|
|
|
|
antibody syndrome.260,261,312 314 |
|
|
|
|
Reperfusion treatment |
|
|
|
|
Rescue thrombolytic therapy is recommended |
|
|
|
|
for patients with haemodynamic deterioration |
I |
B |
|
|
on anticoagulation treatment.282 |
|
|
|
|
As an alternative to rescue thrombolytic ther- |
|
|
|
|
apy, surgical embolectomye or percutaneous |
|
|
|
|
catheter-directed treatmente should be con- |
IIa |
C |
|
|
sidered for patients with haemodynamic dete- |
|
|
|
|
rioration on anticoagulation treatment. |
|
|
|
|
Routine use of primary systemic thrombolysis |
|
|
|
|
is not recommended in patients with inter- |
III |
B |
|
|
mediateor low-risk PE.c,f 179 |
|
|
|
CrCl = creatinine clearance; INR = international normalized ratio; LMWH = low-molecular weight heparin; NOAC(s) = non-vitamin K antagonist oral anticoagulant(s); PE = pulmonary embolism; UFH = unfractionated heparin; VKA = vitamin K antagonist.
aClass of recommendation. bLevel of evidence.
cSee Table 8 for definition of the PE severity and PE-related risk.
dDabigatran is not recommended in patients with CrCl <30 mL/min. Edoxaban should be given at a dose of 30 mg once daily in patients with CrCl of 15 - 50 mL/ min and is not recommended in patients with CrCl <15 mL/min. Rivaroxaban and apixaban are to be used with caution in patients with CrCl 15 - 29 mL/min, and their use is not recommended in patients with CrCl <15 mL/min.
eIf appropriate expertise and resources are available on-site.
fThe risk-to-benefit ratios of surgical embolectomy or catheter-directed procedures have not yet been established in intermediateor low-risk PE.
6.8 Recommendations for multidisciplinary pulmonary embolism teams
Recommendation |
Classa |
Levelb |
|
|
|
|
|
Set-up of a multidisciplinary team and a programme for the management of highand (in
selected cases) intermediate-risk PE should be IIa C considered, depending on the resources and
expertise available in each hospital.
PE = pulmonary embolism. aClass of recommendation. bLevel of evidence.
6.9 Recommendations for inferior vena cava filters
Recommendations |
Classa Levelb |
IVC filters should be considered in patients |
|
|
|
with acute PE and absolute contraindications |
IIa |
C |
|
to anticoagulation. |
|
|
|
IVC filters should be considered in cases of PE |
|
|
|
recurrence despite therapeutic |
IIa |
C |
|
anticoagulation. |
|
|
|
Routine use of IVC filters is not |
III |
A |
|
recommended.302 304 |
|||
|
|
||
IVC = inferior vena cava; PE = pulmonary embolism. |
|
|
|
aClass of recommendation. |
|
|
|
bLevel of evidence. |
|
|
6.10 Recommendations for early discharge and home treatment
Recommendation |
Classa |
Levelb |
|
|
|
|
|
Carefully selected patients with low-risk PE should be considered for early discharge and
continuation of treatment at home, if proper IIa A outpatient care and anticoagulant treatment
can be provided.c 178,206,317 319
PE = pulmonary embolism. aClass of recommendation. bLevel of evidence.
cSee section 7 and Figure 6 for further guidance on defining low-risk PE and deci- sion-making.
2019 September 13 on guest by 1093/eurheartj/ehz405/5556136.abstract/doi/10-article-com/eurheartj/advance.oup.https://academic from Downloaded
28 |
ESC Guidelines |
lar retrieval of the permanent filter, or percutaneous nephrostomy or ureteral stent placement.306 Further reported complications
include filter fracture and/or embolization, and DVT occasionally extending up to the vena cava.303,307,308
7 Integrated risk-adapted diagnosis and management
7.1 Diagnostic strategies
Various combinations of clinical assessments, plasma D-dimer measurements, and imaging tests have been proposed and validated for PE
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
diagnosis. These strategies have been tested in patients presenting with suspected PE in the emergency department or during their hospital stay,101,164,171,320 and more recently in the primary care setting.111 Withholding of anticoagulation without adherence to evidence-based diagnostic strategies was associated with a significant increase in the number of VTE episodes and sudden cardiac death at 3 month follow-up.12 The most straightforward diagnostic algorithms for suspected PE—with and without haemodynamic instability—are presented in Figures 4 and 5, respectively. However, it is recognized that the diagnostic approach for suspected PE may vary, depending on the availability of, and expertise in, specific tests in various hospitals and clinical settings.
Suspected PE in a patient with haemodynamic instabilitya
Bedside TTEb
RV dysfunction?c
|
|
No |
|
|
|
|
|
|
Yes |
|
|
|
|
|
|
|
|
|
|
|||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
CTPA immediately available |
|
|
|
|
|
|
|
|
|||||||
|
|
|
|
|
|
|
|
|
and |
|
feasible? |
|
|
|
|
|
|
|
|
|||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Nod |
|
|
|
|
Yes |
|
|
|
||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
CTPA |
||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Positive |
|
|
|
|
|
Negative |
|
||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Search for other causes of |
|
|
|
|
Treatment of |
|
|
|
|
|
Search for other causes of |
||||||||||||
|
shock or instability |
|
|
|
|
high-risk PEa |
|
|
|
|
|
|
|
2019©ESC |
||||||||||
|
|
|
|
|
|
|
|
shock or instability |
||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||
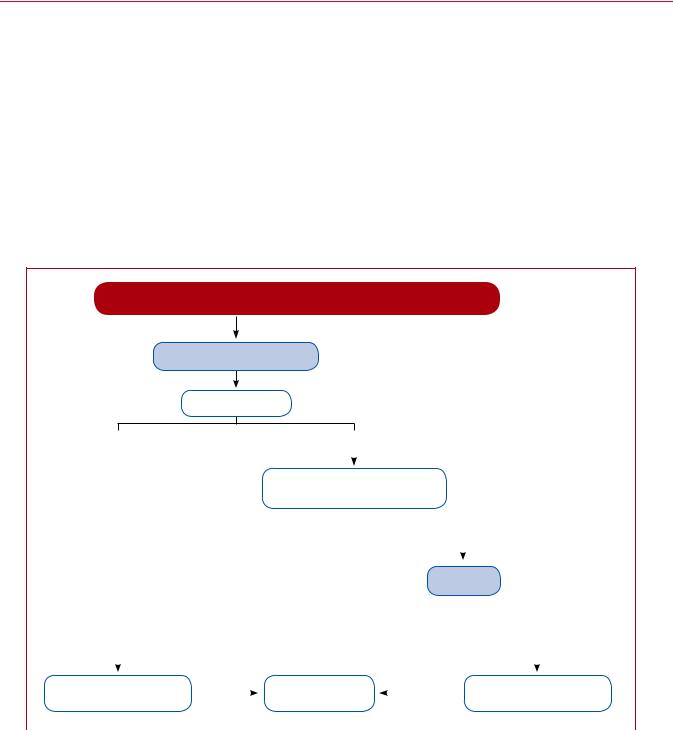
Figure 4 Diagnostic algorithm for patients with suspected high-risk pulmonary embolism presenting with haemodynamic instability. |
||||||||||||||||||||||||
CTPA = computed tomography pulmonary |
angiography; CUS = compression ultrasonography; |
DVT = deep vein thrombosis; LV = left ventricle; |
||||||||||||||||||||||
PE = pulmonary embolism; RV = right ventricle; TOE = transoesophageal echocardiography; TTE = transthoracic echocardiogram. aSee Table 4 for definition of haemodynamic instability and high-risk PE.
bAncillary bedside imaging tests may include TOE, which may detect emboli in the pulmonary artery and its main branches; and bilateral venous CUS, which may confirm DVT and thus VTE.
cIn the emergency situation of suspected high-risk PE, this refers mainly to a RV/LV diameter ratio >1.0; the echocardiographic findings of RV dysfunction, and the corresponding cut-off levels, are graphically presented in Figure 3, and their prognostic value summarized in Supplementary Data Table 3. dIncludes the cases in which the patient’s condition is so critical that it only allows bedside diagnostic tests. In such cases, echocardiographic findings of RV dysfunction confirm high-risk PE and emergency reperfusion therapy is recommended
2019 September 13 on guest by 1093/eurheartj/ehz405/5556136.abstract/doi/10-article-com/eurheartj/advance.oup.https://academic from Downloaded
ESC Guidelines |
29 |
|
|
|
Suspected PE in a patient without haemodynamic instabilitya |
|
|
|
||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Assess clinical probability of PE |
|
|
|
|||
|
|
|
|
Clinical judgement or prediction ruleb |
|
|
|
|||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Low or intermediate clinical probability, |
High clinical probability |
|
|
|
|||||
|
|
|
or PE unlikely |
or PE likely |
|
|
|
|||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
D-dimer test |
|
|
|
|
|
||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Negative |
Positive |
|
|
|
|
|
||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
CTPA |
CTPA |
|
|
|
|||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
No PE |
|
PE confirmedd |
No PE |
PE confirmedd |
|
||||
|
|
|
|
|
|
|
|
|
|
|
No treatmentc |
|
Treatmentc |
No treatmentc |
Treatmentc |
|
|||||
|
|
|
|
|
|
or investigate |
|
|
2019 |
|
|
|
|
|
|
|
furthere |
|
|
||
|
|
|
|
|
|
|
|
|
|
©ESC |
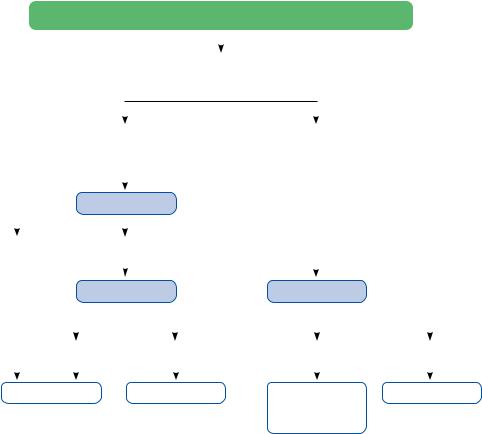
Figure 5 Diagnostic algorithm for patients with suspected pulmonary embolism without haemodynamic instability. CTPA = computed tomography pulmonary angiography/angiogram; PE = pulmonary embolism.
aThe proposed diagnostic strategy for pregnant women with suspected acute PE is discussed in section 9.
bTwo alternative classification schemes may be used for clinical probability assessment, i.e. a three-level scheme (clinical probability defined as low, intermediate, or high) or a two-level scheme (PE unlikely or PE likely). When using a moderately sensitive assay, D-dimer measurement should be restricted to patients with low clinical probability or a PE-unlikely classification, while highly sensitive assays may also be used in patients with intermediate clinical probability of PE due to a higher sensitivity and negative predictive value. Note that plasma D-dimer measurement is of limited use in suspected PE occurring in hospitalized patients.
cTreatment refers to anticoagulation treatment for PE.
dCTPA is considered diagnostic of PE if it shows PE at the segmental or more proximal level.
eIn case of a negative CTPA in patients with high clinical probability, investigation by further imaging tests may be considered before withholding PE-specific treatment.
The diagnostic strategy for suspected acute PE in pregnancy is discussed in section 9.
7.1.1 Suspected pulmonary embolism with haemodynamic instability
The proposed strategy is shown in Figure 4. The clinical probability is usually high and the differential diagnosis includes cardiac tamponade, acute coronary syndrome, aortic dissection, acute valvular dysfunction, and hypovolaemia. The most useful initial test in this situation is bedside TTE, which will yield evidence of acute RV dysfunction if acute PE is the cause of the patient’s haemodynamic decompensation. In a highly unstable patient, echocardiographic evidence of RV dysfunction is sufficient to prompt immediate reperfusion without further testing. This decision may be strengthened by
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
the (rare) visualization of right heart thrombi.155,157,321,322 Ancillary bedside imaging tests include TOE, which may allow direct visualization of thrombi in the pulmonary artery and its main branches, especially in patients with RV dysfunction. TOE should be cautiously performed in hypoxaemic patients. Moreover, bedside CUS can detect proximal DVT. As soon as the patient is stabilized using supportive treatment, final confirmation of the diagnosis by CT angiography should be sought.
For unstable patients admitted directly to the catheterization laboratory with suspected acute coronary syndrome, pulmonary angiography may be considered as a diagnostic procedure after the acute coronary syndrome has been excluded, provided that PE is a probable diagnostic alternative and particularly if percutaneous catheterdirected treatment is a therapeutic option.
2019 September 13 on guest by 1093/eurheartj/ehz405/5556136.abstract/doi/10-article-com/eurheartj/advance.oup.https://academic from Downloaded

30 |
ESC Guidelines |
7.1.2 Suspected pulmonary embolism without haemodynamic instability
7.1.2.1Strategy based on computed tomographic pulmonary angiography
The proposed strategy based on CTPA is shown in Figure 5. In patients admitted to the emergency department, measurement of plasma D- dimer is the logical first step following the assessment of clinical probability and allows PE to be ruled out in 30% of outpatients. D-dimer should not be measured in patients with a high clinical probability of PE, owing to a low negative predictive value in this population.323 It is also less useful in hospitalized patients because the number that needs to be tested to obtain a clinically relevant negative result is high.
In most centres, multidetector CTPA is the second-line test in patients with an elevated D-dimer level and the first-line test in patients with a high clinical probability of PE. CTPA is considered to be diagnostic of PE when it shows a clot at least at the segmental level
of the pulmonary arterial tree. False-negative results of CTPA have been reported in patients with a high clinical probability of PE;115
however, such discrepancies are infrequent and the 3 month thromboembolic risk was low in these patients.171 Accordingly, both the necessity of performing further tests and the nature of these tests remain controversial in these clinical situations.
7.1.2.2Strategy based on ventilation/perfusion scintigraphy
In hospitals in which V/Q scintigraphy is readily available, it is a valid option for patients with an elevated D-dimer and a contraindication to CTPA. Also, V/Q scintigraphy may be preferred over CTPA to avoid unnecessary radiation, particularly in younger patients and in female patients in whom thoracic CT might raise the lifetime risk of breast cancer.324 V/Q lung scintigraphy is diagnostic (with either normalor high-probability findings) in 30 50% of emergency ward patients with suspected PE.75,122,134,325 The proportion of diagnostic V/Q scans is higher in patients with a normal chest X-ray, and this might support the use of a V/Q scan as a first-line imaging test for PE in younger patients, depending on local availability.326 The number of patients with inconclusive findings may further be reduced by taking into account clinical probability. Thus, patients with a non-diagnostic lung scan and low clinical probability of PE have a low prevalence of confirmed PE,124,325 and the negative predictive value of this combination is further increased by the absence of a DVT on lower-limb CUS. If a highprobability lung scan is obtained from a patient with low clinical probability of PE, confirmation by other tests should be considered.
7.2 Treatment strategies
7.2.1 Emergency treatment of high-risk pulmonary embolism
The algorithm for a risk-adjusted therapeutic approach to acute PE is shown in Figure 6; an emergency management algorithm specifically for patients with suspected acute high-risk PE is proposed in Supplementary Data Figure 1. Primary reperfusion treatment, in most cases systemic thrombolysis, is the treatment of choice for patients with high-risk PE. Surgical pulmonary embolectomy or percutaneous catheter-directed treatment are alternative reperfusion options in patients with contraindications to thrombolysis, if expertise with either of these methods and the appropriate resources are available on-site.
Following reperfusion treatment and haemodynamic stabilization, patients recovering from high-risk PE can be switched from
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
parenteral to oral anticoagulation. As patients belonging to this risk category were excluded from the phase III NOAC trials, the optimal time point for this transition has not been determined by existing evidence but should instead be based on clinical judgement. The specifications concerning the higher initial dose of apixaban or rivaroxaban (for 1 and 3 weeks after PE diagnosis, respectively), or the minimum overall period (5 days) of heparin anticoagulation before switching to dabigatran or edoxaban, must be followed (see Supplementary Data Table 8 for tested and approved regimens).
7.2.2Treatment of intermediate-risk pulmonary embolism
For most cases of acute PE without haemodynamic compromise, parenteral or oral anticoagulation (without reperfusion techniques) is adequate treatment. As shown in Figure 6, normotensive patients with at least one indicator of elevated PE-related risk, or with aggravating conditions or comorbidity, should be hospitalized. In this group, patients with signs of RV dysfunction on echocardiography or CTPA (graphically presented in Figure 3), accompanied by a positive troponin test, should be monitored over the first hours or days due
to the risk of early haemodynamic decompensation and circulatory collapse.179 Routine primary reperfusion treatment, notably full-dose
systemic thrombolysis, is not recommended, as the risk of potentially
life-threatening bleeding complications appears too high for the expected benefits from this treatment.179 Rescue thrombolytic ther-
apy or, alternatively, surgical embolectomy or percutaneous catheter-directed treatment should be reserved for patients who develop signs of haemodynamic instability. In the PEITHO trial, the mean time between randomization and death or haemodynamic
decompensation was 1.79 ± 1.60 days in the placebo (heparin-only) arm.179 Therefore, it appears reasonable to leave patients with inter-
mediate-high-risk PE on LMWH anticoagulation over the first 2 - 3 days and ensure that they remain stable before switching to oral anticoagulation. As mentioned in the previous section, the specifications concerning the increased initial dose of apixaban or rivaroxaban, or the minimum overall period of heparin anticoagulation before switching to dabigatran or edoxaban, must be followed.
Suggestions for the anticoagulation and overall management of acute PE in specific clinical situations, for which conclusive evidence is lacking, are presented in Supplementary Data Table 9.
7.2.3Management of low-risk pulmonary embolism: triage for early discharge and home treatment
As a general rule, early discharge of a patient with acute PE and continuation of anticoagulant treatment at home should be considered if three sets of criteria are fulfilled: (i) the risk of early PE-related death or serious complications is low (section 5); (ii) there is no serious comorbidity or aggravating condition(s) (see section 5) that would mandate hospitalization; and (iii) proper outpatient care and anticoagulant treatment can be provided, considering the patient’s (anticipated) compliance, and the possibilities offered by the healthcare system and social infrastructure.
Randomized trials and prospective management cohort studies that investigated the feasibility and safety of early discharge, and home treatment, of PE adhered to these principles, even though
2019 September 13 on guest by 1093/eurheartj/ehz405/5556136.abstract/doi/10-article-com/eurheartj/advance.oup.https://academic from Downloaded