Материал: 2019 ESC acute pulmonaryembolism
16 |
ESC Guidelines |
|
|
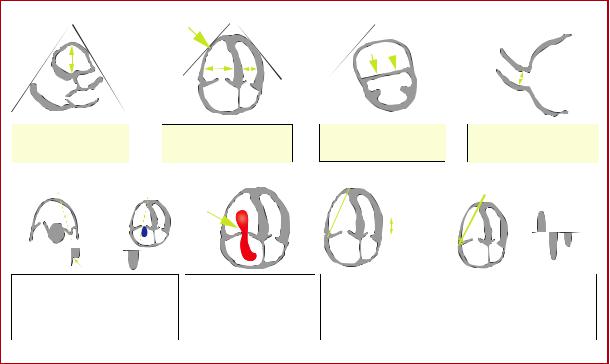
RV
Ao
LV
LA
A. Enlarged right ventricle, parasternal long axis view
RV
RA

AcT <60 ms
”notch”
RV LV
RA
B. Dilated RV with basal RV/LV ratio >1.0, and McConnell sign (arrow), four chamber view
RiHTh
RV
TRPG
<60 mmHg
RV 
IVC
RA
LV
C. Flattened intraventricle septum (arrows) parasternal short axis view
M-Mode
 TAPSE
TAPSE
<16 mm
D. Distended inferior vena cava with diminished inspiratory collapsibility, subcostal view
Tissue Doppler Imaging
S’
E’ A’
S’ <9.5/s
E. 60/60 sign: coexistence of |
|
F. Right heart mobile thrombus |
|
G. Decreased tricuspid annular |
|
H. Decreased peak systolic (S’) |
|
acceleration time of pulmonary ejection |
|
detected in right heart cavities |
|
plane systolic excursion (TAPSE) |
|
velocity of tricuspid annulus |
|
<60 ms and midsystolic “notch” with |
|
(arrow) |
|
measured with M-Mode |
|
(<9.5 cm/s) |
|
mildy elevated (<60 mmHg) peak systolic |
|
|
|
(<16 mm) |
|
|
|
gradient at the tricuspic valve |
|
|
|
|
|
|
©ESC 2019 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Figure 3 Graphic representation of transthoracic echocardiographic parameters in the assessment of right ventricular pressure overload. A0 = peak late diastolic (during atrial contraction) velocity of tricuspid annulus by tissue Doppler imaging; AcT = right ventricular outflow Doppler acceleration time; Ao = aorta; E0 = peak early diastolic velocity of tricuspid annulus by tissue Doppler imaging; IVC = inferior vena cava; LA = left atrium; LV = left ventricle; RA = right atrium; RiHTh = right heart thrombus (or thrombi); RV = right ventricle/ventricular; S0 = peak systolic velocity of tricuspid annulus by tissue Doppler imaging; TAPSE = tricuspid annular plane systolic excursion; TRPG = tricuspid valve peak systolic gradient.
Echocardiographic examination is not mandatory as part of the routine diagnostic workup in haemodynamically stable patients with suspected PE,124 although it may be useful in the differential diagnosis of acute dyspnoea. This is in contrast to suspected high-risk PE, in which the absence of echocardiographic signs of RV overload or dysfunction practically excludes PE as the cause of haemodynamic instability. In the latter case, echocardiography may be of further help in the differential diagnosis of the cause of shock, by detecting pericardial tamponade, acute valvular dysfunction, severe global or regional LV dysfunction, aortic dissection, or hypovolaemia.152 Conversely, in a haemodynamically compromised patient with suspected PE, unequivocal signs of RV pressure overload, especially with more specific echocardiographic findings (60/60 sign, McConnell sign, or rightheart thrombi), justify emergency reperfusion treatment for PE if immediate CT angiography is not feasible in a patient with high clinical probability and no other obvious causes for RV pressure overload.152
Mobile right-heart thrombi are detected by TTE or transoesophageal echocardiography (TOE), or by CT angiography, in <4% of unselected patients with PE.153 155 Their prevalence may reach 18% among PE patients in the intensive care setting.156 Mobile right-heart thrombi essentially confirm the diagnosis of PE and are associated with high early mortality, especially in patients with RV dysfunction.155,157 159
In some patients with suspected acute PE, echocardiography may detect increased RV wall thickness or tricuspid insufficiency jet velocity beyond values compatible with acute RV pressure overload (>3.8 m/s or a tricuspid valve peak systolic gradient >60 mmHg).160 In these
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
cases, chronic thromboembolic (or other) pulmonary hypertension (PH) should be included in the differential diagnosis.
4.10 Compression ultrasonography
In the majority of cases, PE originates from DVT in a lower limb, and only rarely from upper-limb DVT (mostly following venous catheterization). In a study using venography, DVT was found in 70% of patients with proven PE.161 Nowadays, lower-limb CUS has largely replaced venography for diagnosing DVT. CUS has a sensitivity >90% and a specificity of
95% for proximal symptomatic DVT.162,163 CUS shows a DVT in 30 50% of patients with PE,162 164 and finding a proximal DVT in patients suspected of having PE is considered sufficient to warrant anticoagulant treatment without further testing.165 However, patients in whom PE is indirectly confirmed by the presence of a proximal DVT should undergo risk assessment for PE severity and the risk of early death.
In the setting of suspected PE, CUS can be limited to a simple fourpoint examination (bilateral groin and popliteal fossa). The only validated diagnostic criterion for DVT is incomplete compressibility of the vein, which indicates the presence of a clot, whereas flow measurements are unreliable. A positive proximal CUS result has a high positive predictive value for PE. The high diagnostic specificity (96%) along with a low sensitivity (41%) of CUS in this setting was shown by a recent meta-analysis.165,166 CUS is a useful procedure in the diagnostic strategy of patients with CT contraindications. The probability of a positive proximal CUS in suspected PE is higher in patients with signs and symptoms related to the leg veins than in asymptomatic patients.162,163
2019 September 13 on guest by 1093/eurheartj/ehz405/5556136.abstract/doi/10-article-com/eurheartj/advance.oup.https://academic from Downloaded

|
ESC Guidelines |
|
17 |
|
|
||
|
|
|
|
|
|
|
|
|
4.11 Recommendations for diagnosis |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Recommendations |
Classa |
Levelb |
|
|
|
|
|
|
|
|
|
|
|
|
|
Suspected PE with haemodynamic instability |
|
|
|
|
|
|
|
In suspected high-risk PE, as indicated by the presence of haemodynamic instability, bedside echocardiography or emer- |
I |
C |
|
|
|
|
|
gency CTPA (depending on availability and clinical circumstances) is recommended for diagnosis.169 |
|
|
|
||
|
|
|
|
|
|
|
|
|
|
It is recommended that i.v. anticoagulation with UFH, including a weight-adjusted bolus injection, be initiated without delay |
I |
C |
|
|
|
|
|
in patients with suspected high-risk PE. |
|
|
|
||
|
|
|
|
|
|
|
|
|
|
Suspected PE without haemodynamic instability |
|
|
|
|
|
|
|
The use of validated criteria for diagnosing PE is recommended.12 |
I |
B |
|
|
|
|
|
Initiation of anticoagulation is recommended without delay in patients with high or intermediate clinical probability of PE |
I |
C |
|
|
|
|
|
while diagnostic workup is in progress. |
|
|
|
||
|
|
|
|
|
|
|
|
|
|
Clinical evaluation |
|
|
|
|
|
|
|
It is recommended that the diagnostic strategy be based on clinical probability, assessed either by clinical judgement or by |
I |
A |
|
|
|
|
|
a validated prediction rule.89,91,92,103,134,170 172 |
|
|
|
||
|
|
|
|
|
|
|
|
|
|
D-dimer |
|
|
|
|
|
|
|
Plasma D-dimer measurement, preferably using a highly sensitive assay, is recommended in outpatients/emergency depart- |
|
|
|
|
|
|
|
ment patients with low or intermediate clinical probability, or those that are PE-unlikely, to reduce the need for unneces- |
I |
A |
|
|
|
|
|
sary imaging and irradiation.101 103,122,164,171,173,174 |
|
|
|
|
|
|
|
As an alternative to the fixed D-dimer cut-off, a negative D-dimer test using an age-adjusted cut-off (age 10 mg/L, in |
|
|
|
|
|
|
|
patients aged >50 years) should be considered for excluding PE in patients with low or intermediate clinical probability, |
IIa |
B |
|
|
|
|
|
or those that are PE-unlikely.106 |
|
|
|
|
|
|
|
As an alternative to the fixed or age-adjusted D-dimer cut-off, D-dimer levels adapted to clinical probabilityc should be |
IIa |
B |
|
|
|
|
|
considered to exclude PE.107 |
|
|
|
||
|
|
|
|
|
|
|
|
|
|
D-dimer measurement is not recommended in patients with high clinical probability, as a normal result does not safely |
III |
A |
|
|
|
|
|
exclude PE, even when using a highly sensitive assay.175,176 |
|
|
|
||
|
|
|
|
|
|
|
|
|
|
CTPA |
|
|
|
|
|
|
|
It is recommended to reject the diagnosis of PE (without further testing) if CTPA is normal in a patient with low or inter- |
I |
A |
|
|
|
|
|
mediate clinical probability, or who is PE-unlikely.101,122,164,171 |
|
|
|
||
|
|
|
|
|
|
|
|
|
|
It is recommended to accept the diagnosis of PE (without further testing) if CTPA shows a segmental or more proximal |
I |
B |
|
|
|
|
|
filling defect in a patient with intermediate or high clinical probability.115 |
|
|
|
||
|
|
|
|
|
|
|
|
|
|
It should be considered to reject the diagnosis of PE (without further testing) if CTPA is normal in a patient with high clini- |
IIa |
B |
|
|
|
|
|
cal probability or who is PE-likely.171 |
|
|
|
||
|
|
|
|
|
|
|
|
|
|
Further imaging tests to confirm PE may be considered in cases of isolated subsegmental filling defects.115 |
IIb |
C |
|
|
|
|
|
CT venography is not recommended as an adjunct to CTPA.115,164 |
III |
B |
|
|
|
|
|
V/Q scintigraphy |
|
|
|
|
|
|
|
It is recommended to reject the diagnosis of PE (without further testing) if the perfusion lung scan is normal.75,122,134,174 |
I |
A |
|
|
|
|
|
It should be considered to accept that the diagnosis of PE (without further testing) if the V/Q scan yields high probability |
IIa |
B |
|
|
|
|
|
for PE.134 |
|
|
|
||
|
|
|
|
|
|
|
|
|
|
A non-diagnostic V/Q scan should be considered as exclusion of PE when combined with a negative proximal CUS in |
IIa |
B |
|
|
|
|
|
patients with low clinical probability, or who are PE-unlikely.75,122,174 |
|
|
|
||
|
|
|
|
|
|
|
|
|
|
|
|
Continued |
|
||
2019 September 13 on guest by 1093/eurheartj/ehz405/5556136.abstract/doi/10-article-com/eurheartj/advance.oup.https://academic from Downloaded

18 |
ESC Guidelines |
||||
|
|
|
|
|
|
|
|
|
|
|
|
|
V/Q SPECT |
|
|
|
|
|
V/Q SPECT may be considered for PE diagnosis.121,126 128 |
IIbd |
B |
|
|
|
Lower-limb CUS |
|
|
|
|
|
It is recommended to accept the diagnosis of VTE (and PE) if a CUS shows a proximal DVT in a patient with clinical suspi- |
I |
A |
|
|
|
cion of PE.164,165 |
|
|
||
|
|
|
|
|
|
|
If CUS shows only a distal DVT, further testing should be considered to confirm PE.177 |
IIa |
B |
|
|
|
If a positive proximal CUS is used to confirm PE, assessment of PE severity should be considered to permit risk-adjusted |
IIa |
C |
|
|
|
management.178,179 |
|
|
||
|
|
|
|
|
|
|
MRA |
|
|
|
|
|
MRA is not recommended for ruling out PE.139,140 |
III |
A |
|
|
CT = computed tomographic; CTPA = computed tomography pulmonary angiography/angiogram; CUS = compression ultrasonography; DVT = deep vein thrombosis; i.v. = intravenous; MRA = magnetic resonance angiography; PE = pulmonary embolism; SPECT = single-photon emission computed tomography; UFH = unfractionated heparin; V/Q = ventilation/perfusion (lung scintigraphy); VTE = venous thromboembolism.
aClass of recommendation. bLevel of evidence.
cD-dimer cut-off levels adapted to clinical probability according to the YEARS model (signs of DVT, haemoptysis, and whether an alternative diagnosis is less likely than PE) may be used. According to this model, PE is excluded in patients without clinical items and D-dimer levels <1000 mg/L, or in patients with one or more clinical items and D-dimer levels <500 mg/L.107
dLow level of recommendation in view of the limitations summarized in Table 5.
In patients admitted to the emergency department with haemodynamic instability and suspicion of PE, a combination of venous ultrasound with cardiac ultrasound may further increase specificity. Conversely, an echocardiogram without signs of RV dysfunction and a normal venous ultrasound excluded PE with a high (96%) negative predictive value in one study.167
For further details on the diagnosis and management of DVT, the reader is referred to the joint consensus document of the ESC Working Groups of Aorta and Peripheral Vascular Diseases, and Pulmonary Circulation and Right Ventricular Function.1
4.12 Computed tomography venography
When using CTPA, it is possible to image the deep veins of the legs during the same acquisition.115 However, this approach has not been widely validated and the added value of venous imaging is limited.164 Moreover, using CT venography is associated with increased radiation doses.168
5 Assessment of pulmonary embolism severity and the risk of early death
Risk stratification of patients with acute PE is mandatory for determining the appropriate therapeutic management approach. As described in section 3.3, initial risk stratification is based on clinical symptoms and signs of haemodynamic instability (Table 4), which indicate a high risk of early death. In the large remaining group of patients with PE who present without haemodynamic instability, further (advanced) risk stratification requires the assessment of two sets of prognostic criteria: (i) clinical, imaging, and laboratory indicators of PE severity, mostly related to the presence of RV dysfunction; and (ii) presence of comorbidity and any other aggravating conditions that may adversely affect early prognosis.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
5.1 Clinical parameters of pulmonary embolism severity
Acute RV failure, defined as a rapidly progressive syndrome with systemic congestion resulting from impaired RV filling and/or reduced RV flow output,68 is a critical determinant of outcome in acute PE. Tachycardia, low systolic BP, respiratory insufficiency (tachypnoea and/or low SaO2), and syncope, alone or in combination, have been associated with an unfavourable short-term prognosis in acute PE.
5.2 Imaging of right ventricular size and function
5.2.1 Echocardiography
Echocardiographic parameters used to stratify the early risk of patients with PE are graphically presented in Figure 3, and their prognostic values are summarized in Supplementary Data Table 3. Of these, an RV/LV diameter ratio >1.0 and a TAPSE <16 mm are the findings for which an association with unfavourable prognosis has most frequently been reported.148
Overall, evidence for RV dysfunction on echocardiography is found in >25% of unselected patients with acute PE.145 Systematic reviews and meta-analyses have suggested that RV dysfunction on echocardiography is associated with an elevated risk of short-term mortality in patients who appear haemodynamically stable at presentation,180,181 but its overall positive predictive value for PE-related death was low (<10%) in a metaanalysis.180 This weakness is partly related to the fact that echocardiographic parameters have proved difficult to standardize.148,180 Nevertheless, echocardiographic assessment of the morphology and function of the RV is widely recognized as a valuable tool for the prognostic assessment of normotensive patients with acute PE in clinical practice.
In addition to RV dysfunction, echocardiography can identify right- to-left shunt through a patent foramen ovale and the presence of right heart thrombi, both of which are associated with increased
2019 September 13 on guest by 1093/eurheartj/ehz405/5556136.abstract/doi/10-article-com/eurheartj/advance.oup.https://academic from Downloaded

ESC Guidelines |
19 |
mortality in patients with acute PE.67,158 A patent foramen ovale also increases the risk of ischaemic stroke due to paradoxical embolism in patients with acute PE and RV dysfunction.182,183
5.2.2 Computed tomographic pulmonary angiography
CTPA parameters used to stratify the early risk of patients with PE are summarized in Supplementary Data Table 3. Fourchamber views of the heart by CT angiography can detect RV enlargement (RV end-diastolic diameter and RV/LV ratio measured in the transverse or four-chamber view) as an indicator of RV dysfunction. The prognostic value of an enlarged RV is supported by the results of a prospective multicentre cohort study in 457 patients.184 In that study, RV enlargement (defined as an RV/LV ratio >0.9) was an independent predictor of an adverse inhospital outcome, both in the overall population with PE [hazard
ratio (HR) 3.5, 95% CI 1.6 7.7] and in haemodynamically stable patients (HR 3.8, 95% CI 1.3 10.9).184 A meta-analysis of 49
studies investigating >13 000 patients with PE confirmed that an increased RV/LV ratio of >1.0 on CT was associated with a 2.5- fold increased risk for all-cause mortality [odds ratio (OR) 2.5,
95% CI 1.8 3.5], and with a five-fold risk for PE-related mortality (OR 5.0, 95% CI 2.7 9.2).185
Mild RV dilation (RV/LV slightly above 0.9) on CT is a frequent finding (>50% of haemodynamically stable PE patients186), but it probably has minor prognostic significance. However, increasing RV/ LV diameter ratios are associated with rising prognostic specificity,187,188 even in patients considered to be at ‘low’ risk on the basis of clinical criteria.186 Thus, RV/LV ratios > 1.0 (instead of 0.9) on CT angiography may be more appropriate to indicate poor prognosis.
Apart from RV size and the RV/LV ratio, CT may provide further prognostic information based on volumetric analysis of the heart chambers189 191 and assessment of contrast reflux to the inferior vena cava (IVC).185,192,193
5.3 Laboratory biomarkers
5.3.1 Markers of myocardial injury
Elevated plasma troponin concentrations on admission may be associated with a worse prognosis in the acute phase of PE. Cardiac troponin I or T elevation are defined as concentrations above the normal limits, and thresholds depend on the assay used; an overview of the cut-off values has been provided by a meta-analysis.194 Of patients with acute PE, between 30 (using conventional assays)194,195 and 60% (using highsensitivity assays)196,197 have elevated cardiac troponin I or T concentrations. A meta-analysis showed that elevated troponin concentrations were associated with an increased risk of mortality, both in unselected patients (OR 5.2, 95% CI 3.3 8.4) and in those who were haemodynamically stable at presentation (OR 5.9, 95% CI 2.7 13.0).195
On their own, increased circulating levels of cardiac troponins have relatively low specificity and positive predictive value for early mortality in normotensive patients with acute PE. However, when interpreted in combination with clinical and imaging findings, they may improve the identification of an elevated PE-related risk and the further prognostic stratification of such patients (Supplementary Data Table 4). At the other end of the severity spectrum, high-sensitivity troponin assays possess a high negative predictive value in the setting of acute PE.197 For example, in a
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
prospective multicentre cohort of 526 normotensive patients, high-sensitivity troponin T concentrations <14 pg/mL had a negative predictive value of 98% for excluding an adverse in-hospital clinical outcome.63 Age-adjusted high-sensitivity troponin T cutoff values (>14 pg/mL for patients aged <75 years and >45 pg/mL for those >75 years) may further improve the negative predictive
value of this biomarker.196
Heart-type fatty acid-binding protein (H-FABP), an early and sensitive marker of myocardial injury, provides prognostic information in acute PE, both in unselected198,199 and normotensive patients.200,201 In a meta-analysis investigating 1680 patients with PE, H-FABP concentrations >6 ng/mL were associated with an adverse short-term
outcome (OR 17.7, 95% CI 6.0 51.9) and all-cause mortality (OR 32.9, 95% CI 8.8 123.2).202
5.3.2 Markers of right ventricular dysfunction
RV pressure overload due to acute PE is associated with increased myocardial stretch, which leads to the release of B-type natriuretic peptide (BNP) and N-terminal (NT)-proBNP. Thus, the plasma levels of natriuretic peptides reflect the severity of RV dysfunction and haemodynamic compromise in acute PE.203 A meta-analysis found that 51% of 1132 unselected patients with acute PE had elevated BNP or NT-proBNP concentrations on admission; these patients had a 10% risk of early death (95% CI 8.0 13%) and a 23% (95% CI 20 26%) risk of an adverse clinical outcome.204
Similar to cardiac troponins (see above), elevated BNP or NTproBNP concentrations possess low specificity and positive predictive value (for early mortality) in normotensive patients with PE,205 but low levels of BNP or NT-proBNP are capable of excluding an unfavourable early clinical outcome, with high sensitivity and a negative predictive value.180 In this regard, an NT-proBNP cut-off value <500 pg/mL was used to select patients for home treatment in a multicentre management study.206 If emphasis is placed on increasing the prognostic specificity for an adverse early outcome, higher cut-off values >600 pg/mL might be more appropriate.207
5.3.3Other laboratory biomarkers
Lactate is a marker of imbalance between tissue oxygen supply and demand, and consequently of severe PE with overt or imminent haemodynamic compromise. Elevated arterial plasma levels >2 mmol/L
predict PE-related complications, both in unselected208 and in initially normotensive209,210 PE patients.
Elevated serum creatinine levels and a decreased (calculated) glomerular filtration rate are related to 30 day all-cause mortality in acute PE.211 Elevated neutrophil gelatinase-associated lipocalin and cystatin C, both indicating acute kidney injury, are also of prognostic value.212
A recent meta-analysis investigating 18 616 patients with acute PE
found that hyponatraemia predicted in-hospital mortality (OR 5.6, 95% CI 3.4 9.1).213
Vasopressin is released upon endogenous stress, hypotension, and low CO. Its surrogate marker, copeptin, has been reported to be useful for risk stratification of patients with acute PE.214,215 In a singlecentre derivation study investigating 268 normotensive PE patients, copeptin levels >24 pmol/L were associated with a 5.4-fold (95% CI
1.7 17.6) increased risk of an adverse outcome.216 These results
2019 September 13 on guest by 1093/eurheartj/ehz405/5556136.abstract/doi/10-article-com/eurheartj/advance.oup.https://academic from Downloaded
20 |
ESC Guidelines |
|
|
were confirmed in 843 normotensive PE patients prospectively included in three European cohorts.217
5.4 Combined parameters and scores for assessment of pulmonary embolism severity
In patients who present without haemodynamic instability, individual baseline findings may not suffice to determine and further classify PE severity and PE-related early risk when used as stand-alone parameters. As a result, various combinations of the clinical, imaging, and laboratory parameters described above have been used to build prognostic scores, which permit a (semi)quantitative assessment of early PE-related risk of death. Of these, the Bova218 221 and the H- FABP (or high-sensitivity troponin T), Syncope, Tachycardia (FAST) scores219,222,223 have been validated in cohort studies (see Supplementary Data Table 4). However, their implications for patient management remain unclear. To date, only a combination of RV dysfunction on an echocardiogram (or CTPA) with a positive cardiac troponin test has directly been tested as a guide for early therapeutic decisions (anticoagulation plus reperfusion treatment vs. anticoagulation alone) in a large randomized controlled trial (RCT) of PE patients presenting without haemodynamic instability.224
5.5 Integration of aggravating conditions and comorbidity into risk assessment of acute pulmonary embolism
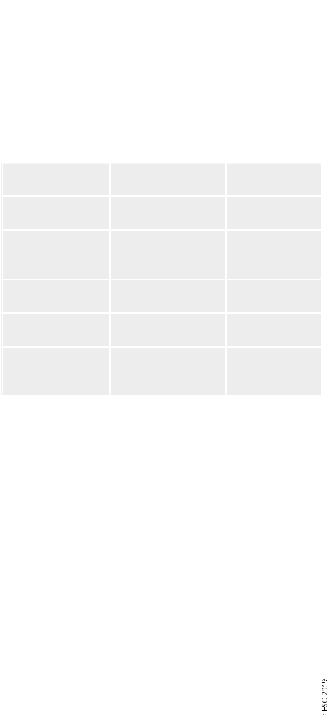
In addition to the clinical, imaging, and laboratory findings, which are directly linked to PE severity and PE-related early death, baseline parameters related to aggravating conditions and comorbidity are necessary to assess a patient’s overall mortality risk and early outcome. Of the clinical scores integrating PE severity and comorbidity, the Pulmonary Embolism Severity Index (PESI) (Table 7) is the one that has been most extensively validated to date.225 228 The principal strength of the PESI lies in the reliable identification of patients at low risk for 30 day mortality (PESI classes I and II). One randomized trial employed a low PESI as the principal inclusion criterion for home treatment of acute PE.178
In view of the complexity of the original PESI, which includes 11 differently weighed variables, a simplified version (sPESI; Table 7) has been developed and validated.229 231 As with the original version of the PESI, the strength of the sPESI lies in the reliable identification of patients at low risk for 30 day mortality. The prognostic performance of the sPESI has been confirmed in observational cohort studies,227,228 although this index has not yet been prospectively used to guide therapeutic management of low-risk PE patients.
The diagnosis of concomitant DVT has been identified as an adverse prognostic factor, being independently associated with death within the first 3 months after acute PE.232 In a meta-analysis investigating 8859 patients with PE, the presence of concomitant DVT was confirmed as a predictor of 30 day all-cause mortality (OR 1.9, 95% CI 1.5 2.4), although it did not predict PE-related adverse outcomes at 90 days.233 Thus, concomitant DVT can be regarded as an indicator of significant comorbidity in acute PE.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
Table 7 Original and simplified Pulmonary Embolism Severity Index
|
Parameter |
|
Original |
Simplified |
|
|
|
version226 |
version229 |
|
Age |
|
Age in years |
1 point (if age >80 |
|
|
|
|
years) |
|
Male sex |
|
þ10 points |
|
|
Cancer |
|
þ30 points |
1 point |
|
Chronic heart |
|
þ10 points |
|
|
failure |
|
|
1 point |
|
|
|
|
|
|
Chronic pulmonary |
|
þ10 points |
|
|
|
|
||
|
disease |
|
|
|
|
Pulse rate >110 |
|
þ20 points |
1 point |
|
b.p.m. |
|
|
|
|
Systolic BP <100 |
|
þ30 points |
1 point |
|
mmHg |
|
|
|
|
Respiratory rate |
|
þ20 points |
|
|
>30 breaths per |
|
|
|
|
min |
|
|
|
|
Temperature |
|
þ20 points |
|
|
<36 C |
|
|
|
|
Altered mental |
|
þ60 points |
|
|
status |
|
|
|
|
Arterial oxyhaemo- |
|
þ20 points |
1 point |
|
globin saturation |
|
|
|
<90% |
|
|
|
|
|
|
|
|
|
|
|
|
Risk strataa |
|
|
|
|
Class I: 65 points |
0 points 5 30 day |
|
|
|
very low 30 day mor- |
mortality risk 1.0% |
|
|
|
tality risk (0 1.6%) |
(95% CI 0.0 2.1%) |
|
|
|
Class II: 66 85 |
|
|
|
|
points |
|
|
|
|
low mortality risk |
|
|
|
(1.7 3.5%) |
1 point(s) 5 30 |
|
|
|
|
Class III: 86 105 |
|
|
|
|
points |
day mortality risk |
|
|
|
moderate mortality |
10.9% (95% CI |
|
|
|
risk (3.2 7.1%) |
8.5 13.2%) |
|
|
|
Class IV: 106 125 |
|
|
|
|
points |
|
|
|
|
high mortality risk |
|
|
|
(4.0 11.4%) |
|
|
|
|
|
Class V: >125 |
|
|
|
|
points |
|
|
|
|
very high mortality |
|
|
|
|
risk (10.0 24.5%) |
|
BP = blood pressure; b.p.m. = beats per minute; CI = confidence interval. aBased on the sum of points.
5.6 Prognostic assessment strategy
The classification of PE severity and the risk of early (in-hospital or 30 day) death is summarized in Table 8. Risk assessment of acute PE begins upon suspicion of the disease and initiation of the diagnostic workup. At this early stage, it is critical to identify patients with (suspected) high-risk PE. This clinical setting necessitates an emergency
2019 September 13 on guest by 1093/eurheartj/ehz405/5556136.abstract/doi/10-article-com/eurheartj/advance.oup.https://academic from Downloaded