Материал: 2019 ESC acute pulmonaryembolism

6 |
|
|
|
|
|
|
ESC Guidelines |
|
|
|
|||||
Table 1 Classes of recommendations |
|
|
|||||
|
|
|
|
|
|
Wording to use |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
recommendationsof |
|
|
Class I |
Evidence and/or general agreement |
Is recommended or is indicated |
|
|
|
|
|
that a given treatment or procedure is |
|
|
||
|
|
|
|
|
|
||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Class II |
|
|
|
|
Classes |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Class IIa |
|
Weight of evidence/opinion is in |
Should be considered |
|
|
|
|
|
|
|
|||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Class IIb |
|
established by evidence/opinion. |
May be considered |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Class III |
Evidence or general agreement that the |
Is not recommended |
|
|
|
|
|
|
given treatment or procedure is not |
|
2019 |
|
|
|
|
|
useful/effective, and in some cases |
|
||
|
|
|
|
may be harmful. |
|
©ESC |
|
|
|
|
|
|
|
|
|
Table 2 Levels of evidence
Level of |
|
Data derived from multiple randomized clinical trials |
evidence A |
|
or meta-analyses. |
|
|
|
Level of |
|
Data derived from a single randomized clinical trial |
evidence B |
|
or large non-randomized studies. |
|
|
|
Level of |
|
Consensus of opinion of the experts and/or small studies, |
2019 |
evidence C |
|
retrospective studies, registries. |
|
|
|
|
©ESC |
determination and the implementation of preventive, diagnostic or therapeutic medical strategies. However, the ESC Guidelines do not override in any way whatsoever the individual responsibility of health professionals to make appropriate and accurate decisions in consideration of each patient’s health condition and in consultation with that patient or the patient’s caregiver where appropriate and/or necessary. It is also the health professional’s responsibility to verify the rules and regulations applicable in each country to drugs and devices at the time of prescription.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
2 Introduction
2.1 Why do we need new Guidelines on the diagnosis and management of pulmonary embolism?
This document follows the previous ESC Guidelines focusing on the clinical management of pulmonary embolism (PE), published in 2000, 2008, and 2014. Many recommendations have been retained or their validity has been reinforced; however, new data have extended or
2019 September 13 on guest by 1093/eurheartj/ehz405/5556136.abstract/doi/10-article-com/eurheartj/advance.oup.https://academic from Downloaded

ESC Guidelines |
7 |
|
|
modified our knowledge in respect of the optimal diagnosis, assessment, and treatment of patients with PE. These new aspects have been integrated into previous knowledge to suggest optimal and—whenever possible—objectively validated management strategies for patients with suspected or confirmed PE. To limit the length of the printed text, additional information, tables, figures, and references are available as supplementary data on the ESC website (www.escardio.org).
These Guidelines focus on the diagnosis and management of acute PE in adult patients. For further details specifically related to the diagnosis and management of deep vein thrombosis (DVT), the reader is referred to the joint consensus document of the ESC Working Groups of Aorta and Peripheral Vascular Diseases, and Pulmonary Circulation and Right Ventricular Function.1
2.2 What is new in the 2019 Guidelines?
2.2.1 New/revised concepts in 2019
Diagnosis
D-dimer cut-off values adjusted for age or clinical probability can be used as an alternative to the fixed cut-off value.
Updated information is provided on the radiation dosage when using CTPA and a lung scan to diagnose PE (Table 6).
Risk assessment
A clear definition of haemodynamic instability and high-risk PE is provided (Table 4).
Assessment of PE severity and early PE-related risk is recommended, in addition to comorbidity/aggravating conditions and overall death risk.
A clear word of caution that RV dysfunction may be present, and affect early outcomes, in patients at ‘low risk’ based on clinical risk scores.
Treatment in the acute phase
Thoroughly revised section on haemodynamic and respiratory support for high-risk PE (Section 6.1).
A dedicated management algorithm is proposed for high-risk PE (Supplementary Figure 1).
NOACs are recommended as the first choice for anticoagulation treatment in a patient eligible for NOACs; VKAs are an alternative to NOACs.
The risk-adjusted management algorithm (Figure 6) was revised to take into consideration clinical PE severity, aggravating conditions/ comorbidity, and the presence of RV dysfunction.
Chronic treatment after the first 3 months
Risk factors for VTE recurrence have been classified according to high, intermediate, or low recurrence risk (Table 11).
Potential indications for extended anticoagulation are discussed, including the presence of a minor transient or reversible risk factor for the index PE, any persisting risk factor, or no identifiable risk factor.
Terminology such as ‘provoked’ vs. ‘unprovoked’ PE/VTE is no longer supported by the Guidelines, as it is potentially misleading and not helpful for decision-making regarding the duration of anticoagulation.
Continued
VTE recurrence scores are presented and discussed in parallel with bleeding scores for patients on anticoagulation treatment (Supplementary Tables 13 and 14 respectively).
A reduced dose of apixaban or rivaroxaban for extended anticoagulation should be considered after the first 6 months of treatment.
PE in cancer
Edoxaban or rivaroxaban should be considered as an alternative to LMWH, with a word of caution for patients with gastrointestinal cancer due to the increased bleeding risk with NOACs.
PE in pregnancy
A dedicated diagnostic algorithm is proposed for suspected PE in pregnancy (Figure 7).
Updated information is provided on radiation absorption related to procedures used for diagnosing PE in pregnancy (Table 12).
Long-term sequelae
An integrated model of patient care after PE is proposed to ensure optimal transition from hospital to community care.
Recommendations on patient care have been extended to the entire spectrum of post-PE symptoms and functional limitation, not only  CTEPH.
CTEPH.
A new comprehensive algorithm is proposed for patient follow-up after acute PE (Figure 8).
CTEPH = Chronic thromboembolic pulmonary hypertension; CTPA = computed tomography pulmonary angiography; LMWH = low-molecular weight heparin; NOAC(s) = non-vitamin K antagonist oral anticoagulant(s); PE = pulmonary embolism; RV = right ventricular; VKA(s) = vitamin K antagonist(s); VTE = venous thromboembolism.
2.2.2 Changes in recommendations 2014 19
|
Recommendations |
2014 |
2019 |
|
|
|
|
|
|
|
|
|
Rescue thrombolytic therapy is recommended for |
IIa |
I |
|
|
|
patients who deteriorate haemodynamically. |
|
|
||
|
|
|
|
|
|
|
Surgical embolectomy or catheter-directed |
|
|
|
|
|
treatment should be considered as alternatives |
IIb |
IIa |
|
|
|
to rescue thrombolytic therapy for patients |
|
|
||
|
|
|
|
|
|
|
who deteriorate haemodynamically. |
|
|
|
|
|
D-dimer measurement and clinical prediction |
|
|
|
|
|
rules should be considered to rule out PE during |
IIb |
IIa |
|
|
|
pregnancy or the post-partum period. |
|
|
|
|
|
Further evaluation may be considered for asymp- |
|
|
|
|
|
tomatic PE survivors at increased risk for |
III |
IIb |
|
|
|
CTEPH. |
|
|
|
|
|
|
|
|
|
|
CTEPH = Chronic thromboembolic pulmonary hypertension; PE = pulmonary embolism.
Coloured columns indicate classes of recommendation (see Table 1 for colour coding).
2019 September 13 on guest by 1093/eurheartj/ehz405/5556136.abstract/doi/10-article-com/eurheartj/advance.oup.https://academic from Downloaded

8 |
ESC Guidelines |
|
|
2.2.3 Main new recommendations 2019
|
Diagnosis |
|
|
|
|
|
|
|
A D-dimer test, using an age-adjusted cut-off or |
|
|
|
adapted to clinical probability, should be considered |
IIa |
|
|
as an alternative to the fixed cut-off level. |
|
|
|
If a positive proximal CUS is used to confirm PE, risk |
|
|
|
assessment should be considered to guide |
IIa |
|
|
management. |
|
|
|
V/Q SPECT may be considered for PE diagnosis. |
IIb |
|
|
Risk assessment |
|
|
|
Assessment of the RV by imaging or laboratory bio- |
|
|
|
markers should be considered, even in the presence |
IIa |
|
|
of a low PESI or a sPESI of 0. |
|
|
|
Validated scores combining clinical, imaging, and labo- |
|
|
|
ratory prognostic factors may be considered to fur- |
IIb |
|
|
ther stratify PE severity. |
|
|
|
Treatment in the acute phase |
|
|
|
When oral anticoagulation is initiated in a patient with |
|
|
|
PE who is eligible for a NOAC (apixaban, dabigatran, |
I |
|
|
edoxaban, or rivaroxaban), a NOAC is the recom- |
||
|
|
|
|
|
mended form of anticoagulant treatment. |
|
|
|
Set-up of multidisciplinary teams for management of |
|
|
|
high-risk and selected cases of intermediate-risk PE |
IIa |
|
|
should be considered, depending on the resources |
||
|
|
|
|
|
and expertise available in each hospital. |
|
|
|
ECMO may be considered, in combination with surgi- |
|
|
|
cal embolectomy or catheter-directed treatment, in |
IIb |
|
|
refractory circulatory collapse or cardiac arrest. |
|
|
|
Chronic treatment and prevention of recurrence |
|
|
|
Indefinite treatment with a VKA is recommended for |
I |
|
|
patients with antiphospholipid antibody syndrome. |
||
|
|
|
|
|
Extended anticoagulation should be considered for |
|
|
|
patients with no identifiable risk factor for the index |
IIa |
|
|
PE event. |
|
|
|
Extended anticoagulation should be considered for |
|
|
|
patients with a persistent risk factor other than anti- |
IIa |
|
|
phospholipid antibody syndrome. |
|
|
|
Extended anticoagulation should be considered for |
|
|
|
patients with a minor transient/reversible risk factor |
IIa |
|
|
for the index PE event. |
|
|
|
|
|
|
|
A reduced dose of apixaban or rivaroxaban should be |
IIa |
|
|
considered after the first 6 months. |
||
|
|
|
|
|
PE in cancer |
|
|
|
Edoxaban or rivaroxaban should be considered as an |
|
|
|
alternative to LMWH, with the exception of patients |
IIa |
|
|
with gastrointestinal cancer. |
|
|
|
PE in pregnancy |
|
|
|
Amniotic fluid embolism should be considered in a |
|
|
|
pregnant or post-partum woman, with unexplained |
IIa |
|
|
haemodynamic instability or respiratory deteriora- |
||
|
|
|
|
|
tion, and disseminated intravascular coagulation. |
|
|
|
|
Continued |
|
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
Thrombolysis or surgical embolectomy should be
IIa
considered for pregnant women with high-risk PE.
NOACs are not recommended during pregnancy or
III
lactation.
Post-PE care and long-term sequelae
Routine clinical evaluation is recommended 3 6 |
I |
|
months after acute PE. |
|
|
|
|
|
An integrated model of care is recommended after |
|
|
acute PE to ensure optimal transition from hospital to |
I |
|
ambulatory care. |
|
|
It is recommended that symptomatic patients with |
|
|
mismatched perfusion defects on a V/Q scan >3 |
|
|
months after acute PE are referred to a pulmonary |
|
|
hypertension/CTEPH expert centre, taking into |
I |
|
account the results of echocardiography, natriu- |
|
|
retic peptide, and/or cardiopulmonary exercise |
|
|
testing. |
|
|
|
|
|
CPET = cardiopulmonary exercise testing; CTEPH = Chronic thromboembolic pulmonary hypertension; CUS = compression ultrasonography; ECMO = extracorporeal membrane oxygenation; LMWH = low-molecular weight heparin; NOAC(s) = non-vitamin K antagonist oral anticoagulant(s); PE = pulmonary embolism; PESI = Pulmonary Embolism Severity Index; RV = right ventricular; SPECT = single-photon emission computed tomography; sPESI = simplified Pulmonary Embolism Severity Index; VKA(s) = vitamin K antagonist(s); V/Q = ventilation/perfusion (lung scintigraphy).
Coloured columns indicate classes of recommendation (see Table 1 for colour coding).
3 General considerations
3.1 Epidemiology
Venous thromboembolism (VTE), clinically presenting as DVT or PE, is globally the third most frequent acute cardiovascular syndrome behind myocardial infarction and stroke.2 In epidemiological studies, annual incidence rates for PE range from 39 115 per 100 000 population; for DVT, incidence rates range from 53 162 per 100 000 population.3,4 Cross-sectional data show that the incidence of VTE is almost eight times higher in individuals aged >80 years than in the fifth decade of life.3 In parallel, longitudinal studies have revealed a rising tendency in annual PE incidence rates4 7 over time. Together with the substantial hospitalassociated, preventable, and indirect annual expenditures for VTE (an estimated total of up to e8.5 billion in the European Union),8 these data demonstrate the importance of PE and DVT in ageing populations in Europe and other areas of the world. They further suggest that VTE will increasingly pose a burden on health systems worldwide in the years to come.
PE may cause <300 000 deaths per year in the US, ranking high
among the causes of cardiovascular mortality.3 In six European countries with a total population of 454.4 million, more than 370 000 deaths were related to VTE in 2004, as estimated on the basis of an epidemiological model.9 Of these patients, 34% died suddenly or within a few hours of the acute event, before therapy could be initiated or take effect. Of the other patients, death resulted from acute PE that was diagnosed after death in 59% and only 7% of patients who died early were correctly diagnosed with PE before death.9
2019 September 13 on guest by 1093/eurheartj/ehz405/5556136.abstract/doi/10-article-com/eurheartj/advance.oup.https://academic from Downloaded
ESC Guidelines |
9 |
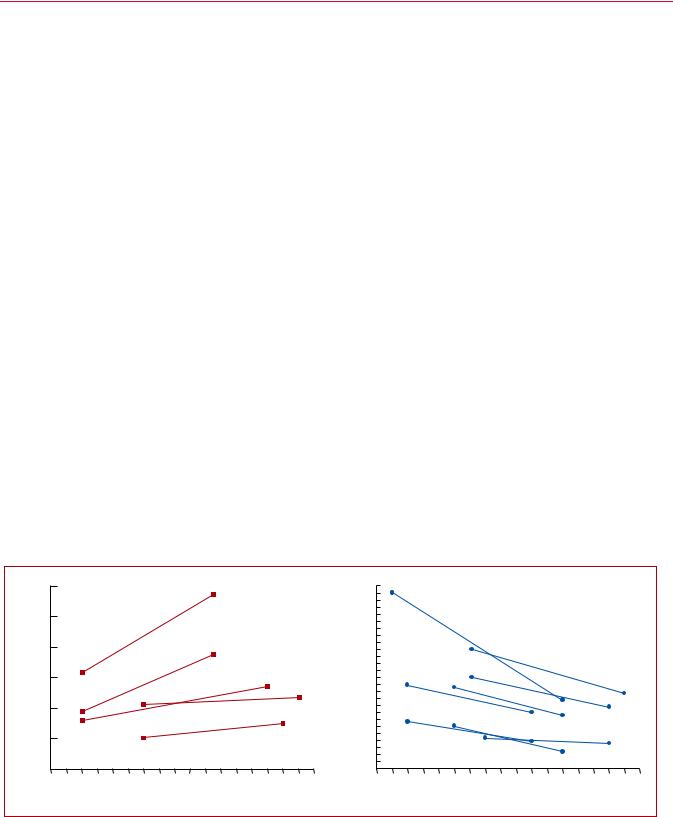
Time trend analyses in European, Asian, and North American populations suggest that case fatality rates of acute PE may be decreasing.4 7,10,11 Increased use of more effective therapies and interventions, and possibly better adherence to guidelines, has most likely exerted a significant positive effect on the prognosis of PE in recent years. However, there is also a tendency towards overdiagnosis of (subsegmental or even non-existent) PE in the modern era,14 and this might in turn lead to a false drop in case fatality rates by inflating the denominator, i.e. the total number of PE cases.
Figure 1 summarizes the existing data on global trends in PE, highlighting increasing incidence rates in parallel with decreasing case fatality rates over an 15 year period.
In children, studies have reported an annual incidence of VTE of between 53 57 per 100 000 among hospitalized patients,19,20 and between 1.4 4.9 per 100 000 in the community overall.21,22
3.2 Predisposing factors
There is an extensive collection of predisposing environmental and genetic factors for VTE; a list of predisposing (risk) factors is shown in Table 3. VTE is considered to be a consequence of the interaction between patient-related—usually permanent—risk factors and set- ting-related—usually temporary—risk factors. Since categorization of temporary and permanent risk factors for VTE is important for assessing the risk of recurrence, and consequently for decisionmaking on chronic anticoagulation, it is discussed in more detail in section 8 of these Guidelines.
Major trauma, surgery, lower-limb fractures and joint replacements, and spinal cord injury are strong provoking factors for VTE.23,24 Cancer is a well-recognized predisposing factor for VTE. The risk of VTE varies with different types of cancer;25,26 pancreatic cancer, haematological malignancies, lung cancer, gastric cancer, and brain cancer carry the highest risk.27,28 Moreover, cancer
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
is a strong risk factor for all-cause mortality following an episode of VTE.29
Oestrogen-containing oral contraceptive agents are associated with an elevated VTE risk, and contraceptive use is the most frequent VTE risk factor in women of reproductive age.30 32 More specifically, combined oral contraceptives (containing both an oestrogen and a progestogen) are associated with an approximately twoto six-fold increase in VTE risk over baseline.32,33 In general, the absolute VTE risk remains low in the majority of the >100 million combined oral contraceptive users worldwide;34 however, VTE risk factors, including severe inherited thrombophilia (discussed in section 8),35 increase this risk. Third-generation combined oral contraceptives, containing progestogens such as desogestrel or gestodene, are associated with a higher VTE risk than the second-generation combined oral contraceptives, which contain progestogens such as levonorgestrel or norgestrel.36,37 On the other hand, hormone-releasing intrauterine devices and some progesterone-only pills (used at contraceptive doses) are not associated with a significant increase in VTE risk;33,38 consequently, and following counselling and full risk assessment, these options are often proposed to women with a personal or strong family history of VTE.
In post-menopausal women who receive hormone replacement therapy, the risk of VTE varies widely depending on the formulation used.39
Infection is a common trigger for VTE.23,40,41 Blood transfusion and erythropoiesis-stimulating agents are also associated with an increased risk of VTE.23,42
In children, PE is usually associated with DVT and is rarely unprovoked. Serious chronic medical conditions and central venous lines are considered likely triggers of PE.43
VTE may be viewed as part of the cardiovascular disease continuum, and common risk factors—such as cigarette smoking, obesity, hypercholesterolaemia, hypertension, and diabetes
|
|
120 |
|
|
|
|
|
|
|
|
|
|
|
26 |
Chinaa, 17 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
%) |
24 |
|
|
|
|
|
|
|
|
||
|
of PE diagnoses / 100.000 inhabitants) |
100 |
|
|
|
|
|
|
|
|
|
|
in-hospital deaths / 100 PE diagnoses; |
22 |
|
|
|
|
|
|
|
|
|
|
Annual Incidence Rate |
|
|
|
|
|
|
|
|
|
|
|
20 |
|
|
|
|
|
|
|
|
|
|
||
80 |
|
|
|
|
|
|
|
|
|
Case Finality Rate |
18 |
|
|
|
|
|
|
|
|
|
|
|||
|
|
|
|
|
|
|
|
|
|
|
|
Italya, 6 |
|
|
|
|
|
|
|
|||||
|
|
|
|
|
|
|
|
|
|
16 |
|
|
|
|
|
|
|
|
|
|||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||
60 |
|
|
|
|
|
|
|
|
|
14 |
USb, 14 |
Spaina, 5 |
|
|
|
|
|
|
||||||
USb,14 |
|
|
|
|
|
|
|
12 |
USb, 16 |
|
|
|
|
|
|
|
||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||
|
USa, 14 |
Italya, 6 |
|
|
|
|
|
10 |
|
|
|
|
|
|
|
|
|
|
||||||
40 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||||||
|
|
|
|
|
|
|
|
|
08 |
|
|
|
|
|
|
|
|
|
|
|||||
|
|
|
|
|
|
|
|
|
|
|
|
USa, 16 |
|
|
|
|
|
|
|
|||||
|
Australiaa, 15 |
|
|
|
|
|
|
|
06 |
|
|
|
|
|
|
|
|
|
||||||
|
|
|
|
|
|
|
|
US |
a, 14 |
|
|
|
|
|
|
|
|
|||||||
|
(number |
20 |
|
|
|
|
|
|
|
|
|
|
(number of |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Spaina, 5 |
|
|
|
|
|
|
04 |
|
|
|
USa,11 |
|
|
|
|
|
|
||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||||||
|
|
|
|
|
|
|
|
|
|
02 |
|
|
|
|
|
|
|
|
|
|||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||
|
0 |
|
|
|
|
|
|
|
|
|
|
0 |
|
|
|
|
|
|
|
|
|
|
||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||
|
|
|
1997 |
1999 |
2001 |
2003 |
2005 |
2007 |
2009 |
2011 |
2013 |
|
|
|
1997 |
1999 |
2001 |
2003 |
2005 |
2007 |
2009 |
2011 |
2013 |
2019 |
|
|
|
|
|
|
Year |
|
|
|
|
|
|
|
|
|
|
|
|
Year |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
©ESC |
||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Figure 1 Trends in annual incidence rates (left panel) and case fatality rates (right panel) of pulmonary embolism around the world, based on data |
||||||||||||||||||||||||
retrieved from various references.5,6,11,14 17 Reproduced with permission from JACC 2016;67:976-90. PE = pulmonary embolism; US = United States. aPE listed as principal diagnosis.
bAny listed code for PE was considered.
2019 September 13 on guest by 1093/eurheartj/ehz405/5556136.abstract/doi/10-article-com/eurheartj/advance.oup.https://academic from Downloaded

10 |
ESC Guidelines |
|
|
Table 3 Predisposing factors for venous thromboembolism (data modified from Rogers et al.23 and Anderson and Spencer24)
Strong risk factors (OR > 10)
Fracture of lower limb
Hospitalization for heart failure or atrial fibrillation/flutter (within previous 3 months)
Hip or knee replacement
Major trauma
Myocardial infarction (within previous 3 months)
Previous VTE
Spinal cord injury
Moderate risk factors (OR 2 9)
Arthroscopic knee surgery
Autoimmune diseases
Blood transfusion
Central venous lines
Intravenous catheters and leads
Chemotherapy
Congestive heart failure or respiratory failure
Erythropoiesis-stimulating agents
Hormone replacement therapy (depends on formulation)
In vitro fertilization
Oral contraceptive therapy
Post-partum period
Infection (specifically pneumonia, urinary tract infection, and HIV)
Inflammatory bowel disease
Cancer (highest risk in metastatic disease)
Paralytic stroke
Superficial vein thrombosis
Thrombophilia
Weak risk factors (OR < 2)
Bed rest >3 days
Diabetes mellitus
Arterial hypertension
Immobility due to sitting (e.g. prolonged car or air travel)
Increasing age
Laparoscopic surgery (e.g. cholecystectomy)
Obesity
Pregnancy
Varicose veins
HIV = human immunodeficiency virus; OR = odds ratio; VTE = venous thromboembolism.
mellitus44 47—are shared with arterial disease, notably atherosclerosis.48 51 However, this may be an indirect association mediated, at least in part, by the complications of coronary artery disease and, in the case of smoking, cancer.52,53 Myocardial infarction and heart failure increase the risk of PE.54,55 Conversely, patients with VTE have an increased risk of subsequent myocardial infarction and stroke, or peripheral arterial embolization.56
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
3.3 Pathophysiology and determinants of outcome
Acute PE interferes with both circulation and gas exchange. Right ventricular (RV) failure due to acute pressure overload is considered the primary cause of death in severe PE. Pulmonary artery pressure (PAP) increases if >30 50% of the total cross-sectional area of the pulmonary arterial bed is occluded by thromboemboli.57 PE-induced vasoconstriction, mediated by the release of thromboxane A2 and serotonin, contributes to the initial increase in pulmonary vascular resistance (PVR) after PE.58 Anatomical obstruction and hypoxic vasoconstriction in the affected lung area lead to an increase in PVR, and a proportional decrease in arterial compliance.59
The abrupt increase in PVR results in RV dilation, which alters the contractile properties of the RV myocardium via the Frank Starling mechanism. The increase in RV pressure and volume leads to an increase in wall tension and myocyte stretch. The contraction time of the RV is prolonged, while neurohumoral activation leads to inotropic and chronotropic stimulation. Together with systemic vasoconstriction, these compensatory mechanisms increase PAP, improving flow through the obstructed pulmonary vascular bed and thus temporarily stabilizing systemic blood pressure (BP). However, the extent of immediate adaptation is limited, as a nonpreconditioned, thin-walled RV is unable to generate a mean PAP >40 mmHg.
Prolongation of RV contraction time into early diastole in the left ventricle (LV) leads to leftward bowing of the interventricular septum.60 The desynchronization of the ventricles may be exacerbated by the development of right bundle branch block. As a result, LV filling is impeded in early diastole, and this may lead to a reduction in the cardiac output (CO), and contribute to systemic hypotension and haemodynamic instability.61
As described above, excessive neurohumoral activation in PE can be the result of both abnormal RV wall tension and circulatory shock. The finding of massive infiltrates of inflammatory cells in the RV myocardia of patients who died within 48 h of acute PE may be explained by high levels of epinephrine released as a result of the PE-induced ‘myocarditis’.62 This inflammatory response might explain the secondary haemodynamic destabilization that sometimes occurs 24 48 h after acute PE, although early recurrence of PE may be an alternative explanation in some of these cases.
Finally, the association between elevated circulating levels of biomarkers of myocardial injury and an adverse early outcome indicates that RV ischaemia is of pathophysiological significance in the acute phase of PE.63,64 Although RV infarction is uncommon after PE, it is likely that the imbalance between oxygen supply and demand can result in damage to cardiomyocytes, and further reduce contractile forces. Systemic hypotension is a critical element in this process, leading to impairment of the coronary driving pressure to the overloaded RV.
The detrimental effects of acute PE on the RV myocardium and the circulation are summarized in Figure 2.
Respiratory failure in PE is predominantly a consequence of haemodynamic disturbances.66 Low CO results in desaturation of the mixed venous blood. Zones of reduced flow in obstructed
2019 September 13 on guest by 1093/eurheartj/ehz405/5556136.abstract/doi/10-article-com/eurheartj/advance.oup.https://academic from Downloaded