Материал: 2019 ESC acute pulmonaryembolism
ESC Guidelines |
21 |
|
|
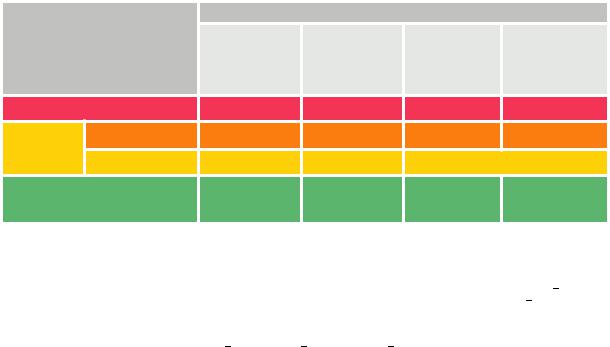
Table 8 Classification of pulmonary embolism severity and the risk of early (in-hospital or 30 day) death
Early mortality risk |
|
Indicators of risk |
|
|
||
|
Haemodynamic |
Clinical parameters |
RV dysfunction on |
Elevated cardiac |
|
|
|
instabilitya |
of PE severity and/ |
TTE or CTPAb |
troponin levelsc |
|
|
|
|
or comorbidity: |
|
|
|
|
|
|
PESI class III–V or |
|
|
|
|
|
|
sPESI ≥1 |
|
|
|
|
High |
+ |
(+)d |
+ |
(+) |
|
|
Intermediate–high |
- |
+e |
+ |
+ |
|
|
Intermediate |
- |
+e |
|
|
|
|
Intermediate–low |
One (or none) positive |
|
||||
Low |
- |
- |
- |
Assesment optional; |
2019 |
|
if assessed, negative |
©ESC |
|||||
|
|
|
|
|||
|
|
|
|
|
||
BP = blood pressure; CTPA = computed tomography pulmonary angiography; H-FABP = heart-type fatty acid-binding protein; NT-proBNP = N-terminal pro B-type natriuretic peptide; PE = pulmonary embolism; PESI = Pulmonary Embolism Severity Index; RV = right ventricular; sPESI = simplified Pulmonary Embolism Severity Index; TTE = transthoracic echocardiogram.
aOne of the following clinical presentations (Table 4): cardiac arrest, obstructive shock (systolic BP <90 mmHg or vasopressors required to achieve a BP >90 mmHg despite an adequate filling status, in combination with end-organ hypoperfusion), or persistent hypotension (systolic BP <90 mmHg or a systolic BP drop >40 mmHg for >15 min, not caused by new-onset arrhythmia, hypovolaemia, or sepsis).
bPrognostically relevant imaging (TTE or CTPA) findings in patients with acute PE, and the corresponding cut-off levels, are graphically presented in Figure 3, and their prognostic value is summarized in Supplementary Data Table 3.
cElevation of further laboratory biomarkers, such as NT-proBNP >600 ng/L, H-FABP >6 ng/mL, or copeptin >24 pmol/L, may provide additional prognostic information. These markers have been validated in cohort studies but they have not yet been used to guide treatment decisions in randomized controlled trials.
dHaemodynamic instability, combined with PE confirmation on CTPA and/or evidence of RV dysfunction on TTE, is sufficient to classify a patient into the high-risk PE category. In these cases, neither calculation of the PESI nor measurement of troponins or other cardiac biomarkers is necessary.
eSigns of RV dysfunction on TTE (or CTPA) or elevated cardiac biomarker levels may be present, despite a calculated PESI of I II or an sPESI of 0.234 Until the implications of such discrepancies for the management of PE are fully understood, these patients should be classified into the intermediate-risk category.
diagnostic algorithm (Figure 4) and immediate referral for reperfusion treatment, as explained in section 7, and displayed in Figure 6 and Supplementary Data Figure 1. Testing for laboratory biomarkers such as cardiac troponins or natriuretic peptides is not necessary for immediate therapeutic decisions in patients with high-risk PE.
In the absence of haemodynamic instability at presentation, further risk stratification of PE is recommended, as it has implications for early discharge vs. hospitalization or monitoring of the patient (explained in section 7). Table 8 provides an overview of the clinical, imaging, and laboratory parameters used to distinguish intermediateand low-risk PE. The PESI is—in its original or simplified form—the most extensively validated and most broadly used clinical score to date, as it integrates baseline indicators of the severity of the acute PE episode with aggravating conditions and the comorbidity of the patient. Overall, a PESI of class I II or an sPESI of 0 is a reliable predictor of low-risk PE.
In addition to clinical parameters, patients in the intermediate-risk group who display evidence of both RV dysfunction (on echocardiography or CTPA) and elevated cardiac biomarker levels in the circulation (particularly a positive cardiac troponin test) are classified into the intermediate-high-risk category. As will be discussed in more detail in section 7, close monitoring is recommended in these cases to permit the early detection of haemodynamic decompensation or collapse, and consequently the need for rescue reperfusion therapy.179 Patients in whom the RV appears normal on echocardiography or
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
CTPA, and/or who have normal cardiac biomarker levels, belong to the intermediate-low-risk category. As an alternative approach, use of further prognostic scores combining clinical, imaging, and laboratory parameters may be considered to semi-quantitatively assess the severity of the PE episode, and distinguish intermediate-high-risk and intermediate-low-risk PE. Supplementary Data Table 4 lists the scores most frequently investigated for this purpose in observational (cohort) studies; however, none of them has been used in RCTs to date.
A recent meta-analysis included 21 cohort studies with a total of 3295 patients with ‘low-risk’ PE based on a PESI of I II or an sPESI of 0.234 Overall, 34% (95% CI 30 39%) of them were reported to have signs of RV dysfunction on echocardiography or CTPA. Data on early mortality were provided in seven studies (1597 patients) and revealed an OR of 4.19 (95% CI 1.39 12.58) for death from any cause in the presence of RV dysfunction; elevated cardiac troponin levels were associated with a comparable magnitude of risk elevation.234 Early all-cause mortality rates (1.8% for RV dysfunction and 3.8% for elevated troponin levels234) were in the lower range of those previously reported for patients with intermediate-risk PE.235 Until the clinical implications of such discrepancies are clarified, patients with signs of RV dysfunction or elevated cardiac biomarkers, despite a low PESI or an sPESI of 0, should be classified into the intermediate-low-risk category.
2019 September 13 on guest by 1093/eurheartj/ehz405/5556136.abstract/doi/10-article-com/eurheartj/advance.oup.https://academic from Downloaded

22 |
ESC Guidelines |
|||||
|
|
|
|
|
|
|
|
5.7 Recommendations for prognostic assessment |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Recommendations |
Classa |
Levelb |
|
|
|
|
|
|
|
|
|
|
|
Initial risk stratification of suspected or confirmed PE, based on the presence of haemodynamic instability, is recom- |
I |
B |
|
|
|
|
mended to identify patients at high risk of early mortality.218,219,235 |
|
|
||
|
|
|
|
|
|
|
|
|
In patients without haemodynamic instability, further stratification of patients with acute PE into intermediateand low- |
I |
B |
|
|
|
|
risk categories is recommended.179,218,219,235 |
|
|
||
|
|
|
|
|
|
|
|
|
In patients without haemodynamic instability, use of clinical prediction rules integrating PE severity and comorbidity, pref- |
IIa |
B |
|
|
|
|
erably the PESI or sPESI, should be considered for risk assessment in the acute phase of PE.178,226,229 |
|
|
||
|
|
|
|
|
|
|
|
|
Assessment of the RV by imaging methodsc or laboratory biomarkersd should be considered, even in the presence of a |
IIa |
B |
|
|
|
|
low PESI or a negative sPESI.234 |
|
|
||
|
|
|
|
|
|
|
|
|
In patients without haemodynamic instability, use of validated scores combining clinical, imaging, and laboratory PE-related |
IIb |
C |
|
|
|
|
prognostic factors may be considered to further stratify the severity of the acute PE episode.218 223 |
|
|
||
|
|
|
|
|
|
|
PE = pulmonary embolism; PESI = Pulmonary Embolism Severity Index; RV = right ventricle; sPESI = simplified Pulmonary Embolism Severity Index. aClass of recommendation.
bLevel of evidence.
cTransthoracic echocardiography or computed tomography pulmonary angiography. dCardiac troponins or natriuretic peptides.
6 Treatment in the acute phase
6.1 Haemodynamic and respiratory support
6.1.1 Oxygen therapy and ventilation
Hypoxaemia is one of the features of severe PE, and is mostly due to the mismatch between ventilation and perfusion. Administration of supplemental oxygen is indicated in patients with PE and SaO2 <90%. Severe hypoxaemia/respiratory failure that is refractory to conventional oxygen supplementation could be explained by right-to-left shunt through a patent foramen ovale or atrial septal defect.67 Further oxygenation techniques should also be considered, including high-flow oxygen (i.e. a high-flow nasal cannula)236,237 and mechanical ventilation (non-invasive or invasive) in cases of extreme instability (i.e. cardiac arrest), taking into consideration that correction of hypoxaemia will not be possible without simultaneous pulmonary reperfusion.
Patients with RV failure are frequently hypotensive or are highly susceptible to the development of severe hypotension during induction of anaesthesia, intubation, and positive-pressure ventilation. Consequently, intubation should be performed only if the patient is unable to tolerate or cope with non-invasive ventilation. When feasible, non-invasive ventilation or oxygenation through a high-flow nasal cannula should be preferred; if mechanical ventilation is used, care should be taken to limit its adverse haemodynamic effects. In particular, positive intrathoracic pressure induced by mechanical ventilation may reduce venous return and worsen low CO due to RV failure in patients with high-risk PE; therefore, positive end-expiratory pressure should be applied with caution. Tidal volumes of approximately 6 mL/kg lean body weight should be used in an attempt to keep the end-inspiratory plateau pressure <30 cm H2O. If intubation is needed, anaesthetic drugs more prone to cause hypotension should be avoided for induction.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
6.1.2 Pharmacological treatment of acute right ventricular failure
Acute RV failure with resulting low systemic output is the leading cause of death in patients with high-risk PE. The principles of acute right heart failure management have been reviewed in a statement from the Heart Failure Association and the Working Group on Pulmonary Circulation and Right Ventricular Function of the ESC.68 An overview of the current treatment options for acute RV failure is provided in Table 9.
If the central venous pressure is low, modest (<500 mL) fluid challenge can be used as it may increase the cardiac index in patients with acute PE.238 However, volume loading has the potential to overdistend the RV and ultimately cause a reduction in systemic CO.239 Experimental studies suggest that aggressive volume expansion is of no benefit and may even worsen RV function.240 Cautious volume loading may be appropriate if low arterial pressure is combined with an absence of elevated filling pressures. Assessment of central venous pressure by ultrasound imaging of the IVC (a small and/or collapsible IVC in the setting of acute high-risk PE indicates low volume status) or, alternatively, by central venous pressure monitoring may help guide volume loading. If signs of elevated central venous pressure are observed, further volume loading should be withheld.
Use of vasopressors is often necessary, in parallel with (or while waiting for) pharmacological, surgical, or interventional reperfusion treatment. Norepinephrine can improve systemic haemodynamics by bringing about an improvement in ventricular systolic interaction and coronary perfusion, without causing a change in PVR.240 Its use should be limited to patients in cardiogenic shock. Based on the results of a small series, the use of dobutamine may be considered for patients with PE, a low cardiac index, and normal BP; however, raising the cardiac index may aggravate the ventilation/perfusion mismatch by further redistributing flow from (partly) obstructed to unobstructed vessels.241 Although experimental data suggest that
2019 September 13 on guest by 1093/eurheartj/ehz405/5556136.abstract/doi/10-article-com/eurheartj/advance.oup.https://academic from Downloaded

ESC Guidelines |
|
23 |
|
||
Table 9 Treatment of right ventricular failure in acute high-risk pulmonary embolism |
||
|
|
|
Strategy |
Properties and use |
Caveats |
Volume optimization |
|
|
Cautious volume loading, saline, or Ringer’s |
Consider in patients with normal low central |
Volume loading can over-distend the RV, wor- |
lactate, <500 mL over 15 30 min |
venous pressure (due, for example, to con- |
sen ventricular interdependence, and reduce |
|
comitant hypovolaemia) |
CO239 |
Vasopressors and inotropes |
|
|
Norepinephrine, 0.2 1.0 mg/kg/mina 240 |
Increases RV inotropy and systemic BP, pro- |
Excessive vasoconstriction may worsen tissue |
|
motes positive ventricular interactions, and |
perfusion |
|
restores coronary perfusion gradient |
|
Dobutamine, 2 20 mg/kg/min241 |
Increases RV inotropy, lowers filling pressures |
May aggravate arterial hypotension if used |
|
|
alone, without a vasopressor; may trigger or |
|
|
aggravate arrhythmias |
Mechanical circulatory support |
|
|
Veno arterial ECMO/extracorporeal life |
Rapid short-term support combined with |
Complications with use over longer periods |
support251,252,258 |
oxygenator |
(>5 10 days), including bleeding and infec- |
|
|
tions; no clinical benefit unless combined with |
|
|
surgical embolectomy; requires an experienced |
|
|
team |
|
|
|
CO = cardiac output; BP = blood pressure; ECMO = extracorporeal membrane oxygenation; RV = right ventricle/ventricular. aEpinephrine is used in cardiac arrest.
levosimendan may restore RV pulmonary arterial coupling in acute PE by combining pulmonary vasodilation with an increase in RV contractility,242 no evidence of clinical benefit is available.
Vasodilators decrease PAP and PVR, but may worsen hypotension and systemic hypoperfusion due to their lack of specificity for the pulmonary vasculature after systemic [intravenous (i.v.)] administration. Although small clinical studies have suggested that inhalation of nitric oxide may improve the haemodynamic status and gas exchange of patients with PE,243 245 no evidence for its clinical efficacy or safety is available to date.246
6.1.3 Mechanical circulatory support and oxygenation
The temporary use of mechanical cardiopulmonary support, mostly with veno arterial extracorporeal membrane oxygenation (ECMO), may be helpful in patients with high-risk PE, and circulatory collapse or cardiac arrest. Survival of critically ill patients has been described in a number of case series,247 252 but no RCTs testing the efficacy and safety of these devices in the setting of high-risk PE have been conducted to date. Use of ECMO is associated with a high incidence of complications, even when used for short periods, and the results depend on the experience of the centre as well as patient selection. The increased risk of bleeding related to the need for vascular access should be considered, particularly in patients undergoing thrombolysis. At present, the use of ECMO as a stand-alone technique with anticoagulation is controversial247,252 and additional therapies, such as surgical embolectomy, have to be considered.
A few cases suggesting good outcomes with use of the ImpellaVR catheter in patients in shock caused by acute PE have been reported.253,254
6.1.4 Advanced life support in cardiac arrest
Acute PE is part of the differential diagnosis of cardiac arrest with non-shockable rhythm against a background of pulseless electrical
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
activity. In cardiac arrest presumably caused by acute PE, current guidelines for advanced life support should be followed.255,256 The decision to treat for acute PE must be taken early, when a good outcome is still possible. Thrombolytic therapy should be considered; once a thrombolytic drug is administered, cardiopulmonary resuscitation should be continued for at least 60 90 min before terminating resuscitation attempts.257
6.2 Initial anticoagulation
6.2.1 Parenteral anticoagulation
In patients with high or intermediate clinical probability of PE (see section 4), anticoagulation should be initiated while awaiting the results of diagnostic tests. This is usually done with subcutaneous, weightadjusted low-molecular weight heparin (LMWH) or fondaparinux (Supplementary Data Table 5), or i.v. unfractionated heparin (UFH). Based on pharmacokinetic data (Supplementary Data Table 6),259 an equally rapid anticoagulant effect can also be achieved with a nonvitamin K antagonist oral anticoagulant (NOAC), and phase III clinical trials have demonstrated the non-inferior efficacy of a single-oral drug anticoagulation strategy using higher doses of apixaban for 7 days or rivaroxaban for 3 weeks.259 261
LMWH and fondaparinux are preferred over UFH for initial anticoagulation in PE, as they carry a lower risk of inducing major bleeding and heparin-induced thrombocytopenia.262 265 Neither LMWH nor fondaparinux need routine monitoring of anti-Xa levels. Use of UFH is nowadays largely restricted to patients with overt haemodynamic instability or imminent haemodynamic decompensation in whom primary reperfusion treatment will be necessary. UFH is also recommended for patients with serious renal impairment [creatinine clearance (CrCl) <30 mL/min] or severe obesity. If LMWH is prescribed in patients with CrCl 15 - 30 mL/min, an adapted dosing scheme should be used. The
2019 September 13 on guest by 1093/eurheartj/ehz405/5556136.abstract/doi/10-article-com/eurheartj/advance.oup.https://academic from Downloaded

24 |
ESC Guidelines |
dosing of UFH is adjusted based on the activated partial thromboplastin time (Supplementary Data Table 7).266
6.2.2Non-vitamin K antagonist oral anticoagulants
NOACs are small molecules that directly inhibit one activated coagulation factor, which is thrombin for dabigatran and factor Xa for apixaban, edoxaban, and rivaroxaban. The characteristics of NOACs used in the treatment of acute PE are summarized in Supplementary Data Table 6. Owing to their predictable bioavailability and pharmacokinetics, NOACs can be given at fixed doses without routine laboratory monitoring. Compared with vitamin K antagonists (VKAs),
there are fewer interactions when NOACs are given concomitantly with other drugs.259 In the phase III VTE trials, the dosages of dabiga-
tran, rivaroxaban, and apixaban were not reduced in patients with mild moderate renal dysfunction (CrCl between 30 60 mL/min), whereas edoxaban was given at a 30 mg dose in these patients. Patients with CrCl <25 mL/min were excluded from the trials testing apixaban, whereas patients with CrCl <30 mL/min were excluded from those investigating rivaroxaban, edoxaban, and dabigatran (Supplementary Data Table 8).
Phase III trials on the treatment of acute VTE (Supplementary Data Table 8), as well as those on extended treatment beyond the first 6 months (see section 8), demonstrated the non-inferiority of NOACs compared with the combination of LMWH with VKA for the prevention of symptomatic or lethal VTE recurrence, along with significantly reduced rates of major bleeding.267 The different drug regimens tested in these trials are displayed in Supplementary Data Table 8. In a meta-analysis, the incidence rate of the primary efficacy outcome was
2.0% for NOAC-treated patients and 2.2% for VKA-treated patients [relative risk (RR) 0.88, 95% CI 0.74 1.05].268 Major bleeding occurred in 1.1% of NOAC-treated patients and 1.7% of VKAtreated patients for an RR of 0.60 (95% CI 0.41 0.88). Compared with VKA-treated patients, critical site major bleeding occurred less frequently in NOAC-treated patients (RR 0.38, 95% CI 0.23 0.62); in particular, there was a significant reduction in intracranial bleeding (RR 0.37, 95% CI 0.21 0.68) and in fatal bleeding (RR 0.36, 95% CI 0.15 0.87) with NOACs compared with VKAs.268
Suggestions for the anticoagulation management of PE in specific clinical situations, for which conclusive evidence is lacking, are presented in Supplementary Data Table 9.
Practical guidance for clinicians regarding the handling of NOACs
and the management of emergency situations related to their use are regularly updated by the European Heart Rhythm Association.259
6.2.3Vitamin K antagonists
VKAs have been the gold standard in oral anticoagulation for more than 50 years. When VKAs are used, anticoagulation with UFH, LMWH, or fondaparinux should be continued in parallel with the oral anticoagulant for >5 days and until the international normalized ratio (INR) value has been 2.0 3.0 for 2 consecutive days. Warfarin may be started at a dose of 10 mg in younger (e.g. aged <60 years) otherwise healthy patients and at a dose <5 mg in older patients.269 The daily dose is adjusted according to the INR over the next 5 7 days, aiming for an INR level of 2.0 3.0. Pharmacogenetic testing may increase the precision of warfarin dosing.270,271 When used in addition to clinical parameters, pharmacogenetic testing improves
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
anticoagulation control and may be associated with a reduced risk of bleeding, but does not reduce the risk of thromboembolic events or mortality.272
The implementation of a structured anticoagulant service (most commonly, anticoagulant clinics) appears to be associated with increased time in the therapeutic range and improved clinical outcome, compared with control of anticoagulation by the general practitioner.273,274 Finally, in patients who are selected and appropriately trained, self-monitoring of VKA is associated with fewer thromboembolic events and increased time in the therapeutic range compared with usual care.275
6.3 Reperfusion treatment
6.3.1 Systemic thrombolysis
Thrombolytic therapy leads to faster improvements in pulmonary obstruction, PAP, and PVR in patients with PE, compared with UFH alone; these improvements are accompanied by a reduction in RV dilation on echocardiography.276 279 The greatest benefit is observed when treatment is initiated within 48 h of symptom onset, but thrombolysis can still be useful in patients who have had symptoms for 6 14 days.280 Unsuccessful thrombolysis, as judged by persistent clinical instability and unchanged RV dysfunction on echocardiography after 36 h, has been reported in 8% of high-risk PE patients.281
A meta-analysis of thrombolysis trials that included (but were not confined to) patients with high-risk PE, defined mainly as the presence of cardiogenic shock, indicated a significant reduction in the combined outcome of mortality and recurrent PE (Supplementary Data Table 10). This was achieved with a 9.9% rate of severe bleeding and a 1.7% rate of intracranial haemorrhage.282
In normotensive patients with intermediate-risk PE, defined as the presence of RV dysfunction and elevated troponin levels, the impact of thrombolytic treatment was investigated in the Pulmonary Embolism Thrombolysis (PEITHO) trial.179 Thrombolytic therapy was associated with a significant reduction in the risk of haemodynamic decompensation or collapse, but this was paralleled by an increased risk of severe extracranial and intracranial bleeding.179 In the PEITHO trial, 30 day death rates were low in both treatment groups, although meta-analyses have suggested a reduction in PErelated and overall mortality of as much as 50 60% following throm-
bolytic treatment in the intermediate-risk category (Supplementary Data Table 10).282,283
The approved regimens and doses of thrombolytic agents for PE, as well as the contraindications to this type of treatment, are shown in Table 10. Accelerated i.v. administration of recombinant tissuetype plasminogen activator (rtPA; 100 mg over 2 h) is preferable to prolonged infusions of first-generation thrombolytic agents (streptokinase and urokinase). Preliminary reports on the efficacy and safety of reduced-dose rtPA284,285 need confirmation by solid evidence before any recommendations can be made in this regard. UFH may be administered during continuous infusion of alteplase, but should be discontinued during infusion of streptokinase or urokinase.65 Reteplase,286 desmoteplase,287 or tenecteplase179,278,279 have also been investigated; at present, none of these agents are approved for use in acute PE.
It remains unclear whether early thrombolysis for (intermediateor high-risk) acute PE has an impact on clinical symptoms, functional
2019 September 13 on guest by 1093/eurheartj/ehz405/5556136.abstract/doi/10-article-com/eurheartj/advance.oup.https://academic from Downloaded

ESC Guidelines |
|
25 |
|
|
|
||
Table 10 Thrombolytic regimens, doses, and contraindications |
|
||
|
|
|
|
Molecule |
Regimen |
Contraindications to fibrinolysis |
|
rtPA |
100 mg over 2 h |
Absolute |
|
|
|
History of haemorrhagic stroke or stroke of unknown origin |
|
|
0.6 mg/kg over 15 min (maximum dose 50 mg)a |
||
Streptokinase |
250 000 IU as a loading dose over 30 min, followed by |
Ischaemic stroke in previous 6 months |
|
|
|||
|
100 000 IU/h over 12 24 h |
Central nervous system neoplasm |
|
|
Major trauma, surgery, or head injury in previous 3 weeks |
||
|
Accelerated regimen: 1.5 million IU over 2 h |
||
|
Bleeding diathesis |
||
Urokinase |
4400 IU/kg as a loading dose over 10 min, followed by |
||
Active bleeding |
|||
|
4400 IU/kg/h over 12 24 h |
||
|
Relative |
||
|
Accelerated regimen: 3 million IU over 2 h |
Transient ischaemic attack in previous 6 months |
|
|
|
||
|
|
Oral anticoagulation |
|
|
|
Pregnancy or first post-partum week |
|
|
|
Non-compressible puncture sites |
|
|
|
Traumatic resuscitation |
|
|
|
Refractory hypertension (systolic BP >180 mmHg) |
|
|
|
Advanced liver disease |
|
|
|
Infective endocarditis |
|
|
|
Active peptic ulcer |
|
|
|
|
|
BP = blood pressure; IU = international units; rtPA, recombinant tissue-type plasminogen activator.
aThis is the accelerated regimen for rtPA in pulmonary embolism; it is not officially approved, but it is sometimes used in extreme haemodynamic instability such as cardiac arrest.
limitation, or CTEPH at long-term follow-up. A small randomized trial of 83 patients suggested that thrombolysis might improve functional capacity at 3 months compared with anticoagulation alone.278 In the PEITHO trial,179 mild persisting symptoms, mainly dyspnoea, were present in 33% of the patients at long-term (at 41.6 ± 15.7 months) clinical follow-up.288 However, the majority of patients (85% in the tenecteplase arm and 96% in the placebo arm) had a low or intermediate probability—based on the ESC Guidelines defini- tion289—of persisting or new-onset PH at echocardiographic followup.288 Consequently, the findings of this study do not support a role for thrombolysis with the aim of preventing long-term sequelae (section 10) after intermediate-risk PE, although they are limited by the fact that clinical follow-up was available for only 62% of the study population.
6.3.2 Percutaneous catheter-directed treatment
Mechanical reperfusion is based on the insertion of a catheter into the pulmonary arteries via the femoral route. Different types of catheters (summarized in Supplementary Data Table 11) are used for mechanical fragmentation, thrombus aspiration, or more commonly a pharmacomechanical approach combining mechanical or ultrasound fragmentation of the thrombus with in situ reduced-dose thrombolysis.
Most knowledge about catheter-based embolectomy is derived from registries and pooled results from case series.290,291 The overall procedural success rates (defined as haemodynamic stabilization, correction of hypoxia, and survival to hospital discharge) of percutaneous catheter-based therapies reported in these studies have reached 87%;292 however, these results may be subject to publication bias. One RCT compared conventional heparin-based
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
treatment and a catheter-based therapy combining ultrasoundbased clot fragmentation with low-dose in situ thrombolysis in 59 patients with intermediate-risk PE. In that study, ultrasoundassisted thrombolysis was associated with a larger decrease in the RV/LV diameter ratio at 24 h, without an increased risk of bleeding.293 Data from two prospective cohort studies294,295 and a registry,296 with a total of 352 patients, support the improvement in RV function, lung perfusion, and PAP in patients with intermediate- or high-risk PE using this technique. Intracranial haemorrhage was rare, although the rate of Global Utilization of Streptokinase and Tissue Plasminogen Activator for Occluded Coronary Arteries (GUSTO) severe and moderate bleeding complications was 10% in one of these cohorts.294 These results should be interpreted with caution, considering the relatively small numbers of patients treated, the lack of studies directly comparing catheterdirected with systemic thrombolytic therapy, and the lack of data from RCTs on clinical efficacy outcomes.
6.3.3 Surgical embolectomy
Surgical embolectomy in acute PE is usually carried out with cardiopulmonary bypass, without aortic cross-clamping and cardioplegic cardiac arrest, followed by incision of the two main pulmonary arteries with the removal or suction of fresh clots. Recent reports have indicated favourable surgical results in highrisk PE, with or without cardiac arrest, and in selected cases of intermediate-risk PE.297 300 Among 174 322 patients hospitalized between 1999 and 2013 with a diagnosis of PE in New York state, survival and recurrence rates were compared between patients who underwent thrombolysis (n = 1854) or surgical embolectomy (n = 257) as first-line therapy.297 Overall, there
2019 September 13 on guest by 1093/eurheartj/ehz405/5556136.abstract/doi/10-article-com/eurheartj/advance.oup.https://academic from Downloaded