Материал: 2019 ESC acute pulmonaryembolism

ESC Guidelines |
41 |
10 Long-term sequelae of pulmonary embolism
The patency of the pulmonary arterial bed is restored in the majority of PE survivors within the first few months following the acute episode; therefore, no routine follow-up CTPA imaging is needed in such patients treated for PE.427 However, in other patients, thrombi become persistent and organized, which in rare cases may result in CTEPH, a potentially life-threatening obstructing vasculopathy. The rarity of this condition is in contrast to the relatively large number of patients who report persisting dyspnoea or poor physical performance over several months after acute PE. Thus, the aims of an efficient follow-up strategy after PE should be to: (i) provide appropriate care (exercise rehabilitation, treatment of comorbidity, behavioural education, and modification of risk factors) to patients with persisting symptoms, and (ii) ensure early detection of CTEPH to refer the patient for further diagnostic workup and specific treatment.
10.1 Persisting symptoms and functional limitation after pulmonary embolism
Cohort studies conducted over the past decade (summarized in Klok et al.428) have revealed that persisting or deteriorating dyspnoea, and poor physical performance, are frequently present 6 months to 3 years after an acute PE episode. The proportion of patients claiming that their health status is worse at 6 month follow-up than it was at the time of PE diagnosis varies widely, ranging between 20 and 75%.429 431 The following baseline parameters and findings could be identified as predictors of exertional dyspnoea at long-term followup after PE: advanced age, cardiac or pulmonary comorbidity, higher body mass index, and history of smoking;429 higher systolic PAP and RV dysfunction at diagnosis;430,432,433 and residual pulmonary vascular obstruction at discharge.434
More recently, a prospective cohort study enrolled 100 patients at five Canadian hospitals between 2010 and 2013, and followed them over 1 year.435 As many as 47% of the patients had reduced maximal aerobic capacity, defined as peak oxygen consumption <80% of the predicted value on cardiopulmonary exercise testing (CPET). This functional outcome was associated with significantly worse generic health-related quality of life and dyspnoea scores, as well as with a significantly reduced 6 min walk distance.435 Independent predictors of reduced functional exercise capacity and quality of life over time included female sex, higher body mass index, history of lung disease, higher pulmonary artery systolic pressures on the 10 day echocardiogram, and higher main pulmonary artery diameter on the baseline CTPA.436 Of note, pulmonary function tests and echocardiographic results at follow-up were largely within normal limits, both in patients with and without reduced maximal aerobic capacity.435 Lack of an association between exercise impairment, and persistent RV dilation or dysfunction, was also reported by a study of 20 survivors of massive or submassive PE.437
Taken together, older and more recent cohort studies have suggested that muscle deconditioning, particularly in the presence of excess body weight and cardiopulmonary comorbidity, is largely responsible for the frequently reported dyspnoea and signs of exercise limitation after acute PE. This also means that, at least in the majority of cases, poor physical performance after PE does not
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
appear to be attributable to ‘large’ residual thrombi, or persisting/ progressive PH and RV dysfunction. Ongoing prospective studies in large numbers of patients may help to better identify predictors of functional and/or haemodynamic impairment after acute PE, and their possible implications for shaping follow-up programmes.438
As mentioned in section 6, it remains unclear whether early reperfusion treatment, notably thrombolysis, has an impact on clinical symptoms, functional limitation, or persistent (or new-onset) PH at long-term follow-up after PE. Consequently, prevention of long-term PE sequelae is, at present, no justification for thrombolytic treatment in the acute phase of PE.
10.2 Chronic thromboembolic pulmonary hypertension
10.2.1 Epidemiology, pathophysiology, and natural history
CTEPH is a disease caused by the persistent obstruction of pulmonary arteries by organized thrombi, leading to flow redistribution and secondary remodelling of the pulmonary microvascular bed. CTEPH has been reported with a cumulative incidence of between 0.1 and 9.1% in the first 2 years after a symptomatic PE event; the large margin of error is due to referral bias, the paucity of early symptoms, and the difficulty of differentiating acute PE from symptoms of preexisting CTEPH.439,440 A prospective, multicentre, observational screening survey for the detection of CTEPH included patients with acute PE from 11 centres in Switzerland, from March 2009 to November 2016. Screening for possible CTEPH was performed at 6, 12, and 24 months using a stepwise algorithm that included a phonebased dyspnoea survey, TTE, right heart catheterization, and radiological confirmation of CTEPH. Of 508 patients assessed for CTEPH screening over 2 years, CTEPH incidence following PE was 3.7 per 1000 patient-years, with a 2 year cumulative incidence of 0.79%.441 In Germany, the incidence of CTEPH in 2016 was estimated at 5.7 per million adult population.442
The hallmark of CTEPH is fibrotic transformation of a pulmonary arterial thrombus, causing fixed mechanical obstruction of pulmonary arteries and leading to overflow of the open pulmonary arterial bed. Together with collateral supply from systemic arteries downstream of pulmonary arterial occlusions, this contributes to microvascular remodelling causing a progressive increase in PVR.443 Owing to this complex pathophysiology, there is no clear correlation between the degree of mechanical obstruction found at
imaging and haemodynamics, which can deteriorate in the absence of recurrent PE.444
Two historical trials assessed survival in patients with CTEPH before the availability of surgical treatment. In both studies, mean PAP >30 mmHg was related to poor survival, similar to that reported for idiopathic pulmonary arterial hypertension.445,446
The most frequently cited risk factors and predisposing conditions for CTEPH are shown in Table 13. In an international registry, a history of acute PE was reported by 75% of patients.447 Associated conditions and comorbidities included thrombophilic disorders, particularly antiphospholipid antibody syndrome and high coagulation factor VIII levels, cancer, a history of splenectomy, inflammatory bowel disease, ventriculo-atrial shunts, and infection of chronic i.v. lines and devices such as implantable pacemakers.
2019 September 13 on guest by 1093/eurheartj/ehz405/5556136.abstract/doi/10-article-com/eurheartj/advance.oup.https://academic from Downloaded

42 |
ESC Guidelines |
|
|
Table 13 Risk factors and predisposing conditions for chronic thromboembolic pulmonary hypertension447 449 |
|
Findings related to the acute PE event |
Concomitant chronic diseases and conditions predisposing to |
(obtained at PE diagnosis) |
CTEPH (documented at PE diagnosis or at 3 6 month follow-up) |
Previous episodes of PE or DVT |
Ventriculo-atrial shunts |
Large pulmonary arterial thrombi on CTPA |
Infected chronic i.v. lines or pacemakers |
Echocardiographic signs of PH/RV dysfunctiona |
History of splenectomy |
CTPA findings suggestive of pre-existing chronic |
Thrombophilic disorders, particularly antiphospholipid antibody syndrome and |
thromboembolic diseaseb |
high coagulation factor VIII levels |
|
Non-O blood group |
|
Hypothyroidism treated with thyroid hormones |
|
History of cancer |
|
Myeloproliferative disorders |
|
Inflammatory bowel disease |
|
Chronic osteomyelitis |
CTEPH = Chronic thromboembolic pulmonary hypertension; CTPA = computed tomographic pulmonary angiography; DVT = deep vein thrombosis; i.v. = intravenous; LV = left ventricular; PE = pulmonary embolism; PH = pulmonary hypertension; RV = right ventricular.
aEchocardiographic criteria of RV dysfunction are graphically presented in Figure 3, and their prognostic value summarized in Supplementary Data Table 3. On CTPA (fourchamber views of the heart), RV dysfunction is defined as RV/LV diameter ratio >1.0.
bDirect and indirect vascular signs, as well as lung parenchymal findings, are summarized in Supplementary Data Table 2.
10.2.2 Clinical presentation and diagnosis
Diagnosing CTEPH is difficult. Algorithms for predicting450 or ruling out CTEPH451,452 are limited by a lack of specificity. The clinical characteristics of patients enrolled in an international CTEPH registry have shown that the median age at diagnosis is 63 years and that both sexes are equally affected; paediatric cases are rare.447 Clinical symptoms and signs are non-specific or absent in early CTEPH, with signs of right heart failure only becoming evident in advanced disease. Thus, early diagnosis remains a challenge in CTEPH, with a median time of 14 months between symptom onset and diagnosis in expert centres.453 When present, the clinical symptoms of CTEPH may resemble those of acute PE or of pulmonary arterial hypertension; in the latter context, oedema and haemoptysis occur more often in CTEPH, while syncope is more common in pulmonary arterial hypertension.453
The diagnosis of CTEPH is based on findings obtained after at least 3 months of effective anticoagulation, to distinguish this condition from acute PE. The diagnosis requires a mean PAP of >25 mmHg along with a pulmonary arterial wedge pressure of <15 mmHg, documented at right heart catheterization in a patient with mismatched perfusion defects on V/Q lung scan. Specific diagnostic signs for CTEPH on multidetector CT angiography or conventional pulmonary cineangiography include ring-like stenoses, webs, slits, and chronic total occlusions.289
Some patients may present with normal pulmonary haemodynamics at rest despite symptomatic disease. If other causes of exercise limitation are excluded, these patients are considered as having chronic thromboembolic disease (CTED). Identification of patients with chronic thromboembolism without PH, who may have an indication for surgical or interventional treatment, requires particular expertise and should be done in CTEPH referral centres. Among 1019 patients who were submitted to pulmonary endarterectomy (PEA) in a UK referral centre, 42 patients did not have pulmonary
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
hypertension at rest but showed functional improvement after the operation.454
Planar V/Q lung scan is a suitable first-line imaging modality for CTEPH as it has 96 97% sensitivity and 90 95% specificity for the diagnosis.455 SPECT seems less sensitive than planar V/Q scanning if assessed at a level of individual segmental arteries, but it is unlikely to miss clinically relevant CTEPH in an individual patient. In contrast to CTEPH, abnormal mismatched perfusion defects sometimes found in pulmonary arterial hypertension and pulmonary veno-occlusive disease typically have a non-segmental pattern.
CTPA is gaining ground as a diagnostic modality in CTEPH,456 but it should not be used as a stand-alone test to exclude the disease.455 Newer diagnostic tests include dual-energy CT, which allows the simultaneous assessment of patency of the pulmonary arteries and of lung perfusion, probably at a cost of some increase in radiation delivered to the patient. Magnetic resonance imaging of the pulmonary vasculature is still considered inferior to CT.457 Cone-beam CT,458 angioscopy,459 intravascular ultrasound, and optical coherence tomography are more suitable for the characterization of lesions during interventional treatment than for diagnosis. High-resolution CT scan of the chest may assist in the differential diagnosis of CTEPH, showing emphysema, bronchial, or interstitial lung disease, as well as infarcts, and vascular and thoracic wall malformations. Perfusion inequalities manifesting as a mosaic parenchymal pattern are frequently found in CTEPH, but may also be observed in <12% of patients with other causes of PH. Differential diagnosis of CTEPH should also include pulmonary arteritis, pulmonary angiosarcoma, tumour embolism, parasites (hydatid cyst), foreign body embolism, and congenital or acquired pulmonary artery stenoses.289
10.2.3 Surgical treatment
Surgical PEA is the treatment of choice for operable CTEPH. In contrast to surgical embolectomy for acute PE, treatment of CTEPH
2019 September 13 on guest by 1093/eurheartj/ehz405/5556136.abstract/doi/10-article-com/eurheartj/advance.oup.https://academic from Downloaded

ESC Guidelines |
43 |
necessitates a true bilateral endarterectomy through the medial layer of the pulmonary arteries. It requires deep hypothermia and intermittent circulatory arrest, without a need for cerebral perfusion.460,461 In-hospital mortality is currently as low as 4.7%462 and is even lower in high-volume single centres.463 The majority of patients experience substantial relief from symptoms and near-normalization of haemodynamics.461 464 Owing to the complexity of both the surgical technique and peri-procedural management, PEA is performed in specialized centres. Eligibility for surgery requires a decision taken during a dedicated meeting of a multidisciplinary CTEPH team including experienced surgeons for PEA, interventional radiologists or cardiologists, radiologists experienced in pulmonary vascular imaging, and clinicians with expertise in PH. The CTEPH team should confirm the diagnosis, assess the surgical accessibility of chronic postthrombotic obstructions (‘surgical operability’), and consider the risks related to comorbidities (‘medical operability’). The operability of patients with CTEPH is determined by multiple factors that cannot easily be standardized. These are related to the suitability of the patient, the expertise of the surgical team, and available resources. General criteria include pre-operative New York Heart Association (NYHA) functional class and the surgical accessibility of thrombi in the main, lobar, or segmental pulmonary arteries.462 Advanced age per se is no contraindication for surgery. There is no haemodynamic threshold or measure of RV dysfunction that can be considered to preclude PEA.
Data from the international CTEPH registry, set up in 27 centres to evaluate the long-term outcome and outcome predictors in 679
operated and not-operated patients, showed estimated survival at 3 years of 89% in operated and 70% in not-operated patients.465
Mortality was associated with NYHA functional class, RA pressure, and a history of cancer.465 In this prospective registry, the long-term prognosis of operated patients was better than the outcome of notoperated patients.465 Additional correlates of mortality were bridging therapy with pulmonary vasodilators, post-operative PH, surgical complications, and additional cardiac procedures in operated patients, and comorbidities such as coronary disease, left heart failure, and chronic obstructive pulmonary disease in not-operated patients.465 A recent report identified mean PAP >38 mmHg and
PVR >425 dyn*s*cm-5 as determinants of poor prognosis in survivors
of surgical treatment for CTEPH.466
Post-operative ECMO is recommended as the standard of care in PEA centres.461 Early post-operative reperfusion oedema may require veno-arterial ECMO, and severe persistent PH may be bridged to emergency lung transplantation with ECMO. After PEA, patients should be followed in CTEPH centres to exclude persistent or recurrent PH, with at least one haemodynamic assessment to be considered at 6 12 months after the intervention.
10.2.4 Balloon pulmonary angioplasty
Over the past decade, balloon pulmonary angioplasty (BPA) has emerged as an effective treatment for technically inoperable CTEPH. It allows dilatation of obstructions down to subsegmental vessels, which are inaccessible to surgery. BPA is a stepwise procedure requiring several (usually 4 10) separate sessions. This is necessary to engage all under-perfused lung segments, while limiting the contrast burden and radiation delivered per session. Navigation in distal
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
pulmonary arteries requires particular expertise, as the complexity and individual variability of the pulmonary arterial tree greatly exceeds that of other vascular beds. Complications include wireand balloon-induced injury, which may result in intrapulmonary bleeding, haemoptysis, and reperfusion lung injury. Usually, bleeding resolves spontaneously, but sometimes it has to be controlled by transient balloon inflation proximal to the site of perforation; in rare cases it requires embolization. Mild hypoxaemia is frequent and can be controlled by oxygen delivery. Mechanical ventilation or ECMO is rarely needed.
The largest published registry to date included 249 patients with a mean age of 61.5 years, who were treated with BPA between 2004 and 2013 in seven Japanese centres.467 Mean PAP decreased from 43 to 24 mmHg after terminating BPA sessions, and this result was maintained in 196 patients who underwent follow-up right heart catheterization. Complications occurred in 36% of the patients, including pulmonary injury (18%), haemoptysis (14%), and pulmonary artery
perforation (2.9%). After BPA, 30 day mortality was 2.6% and overall survival was 97% at 1 year.467
While most of the BPA procedures are performed in technically inoperable patients, this method has also been used for sequential treatment for PH persisting after PEA. Few ‘rescue’ BPA interventions performed in unstable patients remaining on ECMO after PEA were ineffective.468
10.2.5 Pharmacological treatment
Optimal medical treatment for CTEPH consists of anticoagulants, as well as diuretics and oxygen in cases of heart failure or hypoxaemia. Lifelong oral anticoagulation with VKAs is recommended, and also after successful PEA or BPA. No data exist on the efficacy and safety of NOACs.
Pulmonary microvascular disease in CTEPH has provided the rationale for also testing drugs that have been approved for pulmonary arterial hypertension for this indication. Based on available data, medical treatment of CTEPH with targeted therapy is now justified for technically inoperable patients,469,470 as well as for patients with PH persisting after PEA.469 To date, the only drug approved for inoperable CTEPH or persistent/recurrent PH after PEA is riociguat, an oral stimulator of soluble guanylate cyclase.469 In a prospective randomized trial of 261 patients with inoperable CTEPH or persistent/recurrent PH after PEA, treatment with riociguat significantly increased 6 min walking distance and reduced PVR.469 In a similar population of 157 patients, the dual endothelin antagonist bosentan showed a positive effect on haemodynamics, but no improvement was observed in exercise capacity and the primary outcome was not met.471 Another dual endothelin antagonist, macitentan, was found to significantly improve PVR and 6 min walking distance compared to placebo in a phase II trial focusing on inoperable patients with CTEPH.470 Currently, riociguat is being tested in trials addressing its efficacy and safety: (i) as bridging therapy for patients scheduled to undergo PEA (NCT 03273257) and (ii) in comparison to BPA (NCT 02634203).
Overall, the effects on clinical worsening of drugs tested with RCTs in patients with CTEPH have not yet been clarified. Furthermore, no data exist on medical treatment in technically operable patients with prohibitive comorbidities or those refusing surgery.
2019 September 13 on guest by 1093/eurheartj/ehz405/5556136.abstract/doi/10-article-com/eurheartj/advance.oup.https://academic from Downloaded
44 |
ESC Guidelines |
Off-label combination of drugs approved for pulmonary arterial hypertension has been proposed for CTEPH patients presenting with severe haemodynamic compromise, but only limited prospective data are available to date.470
Medical therapy is not indicated in symptomatic survivors of acute PE with documented post-thrombotic obstructions but an absence of PH at right heart catheterization at rest (CTED).
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
10.3 Strategies for patient follow-up after pulmonary embolism
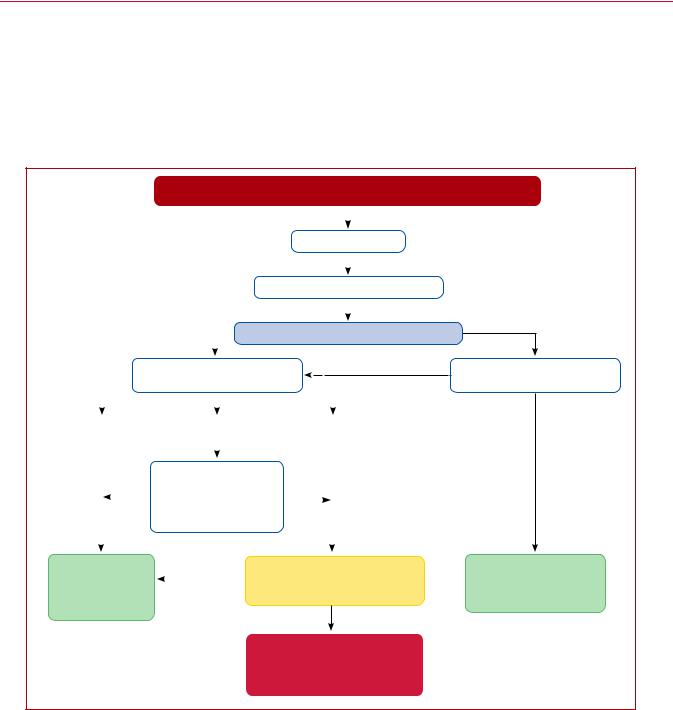
Figure 8 displays a proposed follow-up strategy for survivors of acute PE following discharge from hospital. Evaluation of the patients 3 6 months after the acute PE episode is recommended to assess the persistence (or new onset) and severity of dyspnoea or functional limitation, and to check for possible signs of VTE recurrence, cancer,
|
|
|
|
|
|
|
|
|
|
DIAGNOSIS OF ACUTE PE |
|||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||
|
|
|
|
|
|
|
|
|
|
|
Anticoagulate |
||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||
|
|
|
|
|
|
|
|
|
|
FOLLOW-UP AT 3–6 MONTHSa |
|||||
|
|
|
|
|
|
|
|
|
|
|
|
||||
|
|
|
|
Yes |
|
|
|
Dyspnoea and/or functional limitationb? |
|||||||
|
|
|
|
|
|
||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
≥1 present: |
|
|
|
|
|
TTE: |
|
|
|
may consider TTE |
|||||||
|
|
Determine probability of PHc |
|||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Low |
|
Intermediate |
|
|
|
High |
|||||||||
|
|
|
|
|
|
|
|
≥1 |
|
|
|
||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||
|
|
None |
|
CONSIDER: |
|
|
|
|
|
||||||
|
|
present |
1) Elevated NT-proBNP |
|
present |
|
|
|
|||||||
|
|
|
|
|
|
|
|
|
|||||||
|
|
|
2) Risk factors for CTEPHd |
|
|
|
|
|
|||||||
|
|
|
|
|
|
|
|||||||||
|
|
|
3) Abnormal CPET resultse |
|
|
|
|||||||||
|
|
|
|
No |
|
|
|
|
|
|
|
|
|||
Seek alternative |
|
|
|
|
V/Q SCAN: |
||||||||||
|
|
|
|
|
|
|
|
||||||||
causes of dyspnoeaf |
|
|
|
|
|
|
Mismatched perfusion defects? |
||||||||
|
|
|
|
|
|
||||||||||
and/or |
|
|
|
|
|
|
|||||||||
common causes of PH
Yes
Refer to PH/CTEPH expert centre for further diagnostic work-up
No
ASSESS:
Risk factors for CTEPHd
None present
Focus on anticoagulation and secondary prophylaxis; advise to return if symptoms appear
©ESC 2019
Figure 8 Follow-up strategy and diagnostic workup for long-term sequelae of pulmonary embolism. CPET = cardiopulmonary exercise testing; CTEPH = chronic thromboembolic pulmonary hypertension; NT-proBNP = N-terminal pro B-type natriuretic peptide;
PE = pulmonary embolism; PH = pulmonary hypertension; TTE = transthoracic echocardiography/echocardiogram; V/Q = ventilation/perfusion (lung scintigraphy).
aAssess the persistence (or new onset) and severity of dyspnoea or functional limitation, and also check for possible signs of VTE recurrence, cancer, or bleeding complications of anticoagulation.
bThe Medical Research Council scale can be used to standardize the evaluation of dyspnoea;160 alternatively, the World Health Organization functional class can be determined (Supplementary Data Table16).289
cAs defined by the ESC/ERS guidelines on the diagnosis and treatment of Pulmonary Hypertension (Supplementary Data Tables 17 and 18).289 dRisk factors and predisposing conditions for CTEPH are listed in Table 13.
eCardiopulmonary exercise testing, if appropriate expertise and resources are available on site; abnormal results include, among others, reduced maximal aerobic capacity (peak oxygen consumption), reduced ventilatory equivalent for carbon dioxide, and reduced end-tidal carbon dioxide pressure.
fConsider CPET in the diagnostic work-up.
2019 September 13 on guest by 1093/eurheartj/ehz405/5556136.abstract/doi/10-article-com/eurheartj/advance.oup.https://academic from Downloaded

ESC Guidelines |
45 |
|
|
or bleeding complications of anticoagulation. The severity of dyspnoea can be assessed using the Medical Research Council scale;160 alternatively, the World Health Organization functional class can be determined (Supplementary Data Table 16).289
In patients complaining of persisting dyspnoea and poor physical performance, TTE should be considered as the next step to assess the probability of (chronic) PH and thus possible CTEPH. The criteria and levels of PH probability are defined by current ESC Guidelines,289 and are listed in Supplementary Data Tables 17 and 18. Patients with a high echocardiographic probability of PH, or those with intermediate probability combined with elevated NTproBNP levels or risk factors/predisposing conditions for CTEPH, such as those listed in Table 13, should be considered for a V/Q scan.
If mismatched perfusion defects are found on the V/Q scan, referral to a PH or CTEPH expert centre for further diagnostic workup is indicated. If, on the other hand, the V/Q scan is normal and the patient’s symptoms remain unexplained, CPET may be performed. By providing evidence of reduced maximal aerobic capacity, CPET supports the need for further follow-up visits and helps to identify candidates for pulmonary rehabilitation, exercise, or weight-reduction programmes.435,436 CPET may also be helpful in patients with suspected CTEPH and coexisting left heart and/or respiratory disease; in such cases, it can help to establish the main limiting factor and thus set priorities for the treatment strategy.472
For patients who report as free of dyspnoea or functional limitation at 3 6-month follow-up after acute PE but have risk factors/predisposing conditions for CTEPH (Table 13), further follow-up visits may be scheduled and the patient must be advised to return if symptoms appear. Alternatively, TTE may be considered to assess the probability of PH (Figure 8).
Apart from the recommended screening and diagnostic measures, an integrated model of patient care after PE should be provided, taking into consideration the infrastructure and possibilities offered by each country’s health system. The model should include appropriately qualified nurses, interdisciplinary working with physicians in the care of both in-hospital and ambulatory PE patients, standardized treatment protocols adapted to the capacities of each hospital, and bidirectional referral pathways between general practice and the hospital. Such models ensure smooth transitions between hospital specialists and practitioners; provide continuity, and easy access to care along with information and education; and respect the patients’ preferences, and those of their families and social environment. In this context, nurse-led care models to deliver follow-up have been shown to be effective after acute coronary syndrome,473 in primary care-based management of chronic diseases,474 and in community based self-management initiatives.475 A recently published study investigated the care of 42 patients followed at a pulmonary arterial hypertension (PAH)/CTEPH nurse-led outpatient clinic and showed positive results.476 During patient follow-up visits, appropriately qualified nurses screen for signs and symptoms indicating VTE recurrence or complications of treatment, and assess adherence to medication. Nurses work collaboratively with patients using behavioural frameworks and motivational interviewing, to identify and modify associated risk factors (smoking cessation, diet, physical activity, and exercise). In addition, they promote self-management skills such as
10.4 Recommendations for follow-up after acute pulmonary embolism
|
Recommendations |
Classa |
Levelb |
|
|
|
|
|
|
|
|
|
Routine clinical evaluationc of patients 3 6 |
|
|
|
|
|
months after the acute PE episode is |
I |
B |
|
|
|
recommended.288,352,353,437 |
|
|
|
|
|
An integrated model of patient care after PE |
|
|
|
|
|
(involving hospital specialists, appropriately |
|
|
|
|
|
qualified nurses, and primary care physicians) |
I |
C |
|
|
|
is recommended to ensure optimal transition |
|
|
|
|
|
from hospital to community care. |
|
|
|
|
|
In symptomatic patients with mismatched per- |
|
|
|
|
|
fusion defects persisting on V/Q scand beyond |
|
|
|
|
|
3 months after acute PE, referral to a PH/ |
|
|
|
|
|
CTEPH expert centre is recommended, after |
I |
C |
|
|
|
taking into account the results of echocardiog- |
|
|
|
|
|
raphy, natriuretic peptide levels, and/or |
|
|
|
|
|
CPET.477 |
|
|
|
|
|
Further diagnostic evaluatione should be con- |
|
|
|
|
|
sidered in patients with persistent or new- |
IIa |
C |
|
|
|
onset dyspnoea/exercise limitation after PE. |
|
|
|
|
|
Further diagnostic evaluatione may be consid- |
|
|
|
|
|
ered in asymptomatic patients with risk factors |
IIb |
C |
|
|
|
for CTEPH.f 447 449,478 |
|
|
|
|
CPET = cardiopulmonary exercise testing; CT = computed tomography; CTEPH = Chronic thromboembolic pulmonary hypertension; PE = pulmonary embolism; PH = pulmonary hypertension; V/Q = ventilation/perfusion (lung scintigraphy). aClass of recommendation.
bLevel of evidence.
cFor symptoms suggesting recurrence, bleeding, malignancy, or persistent or new-onset exercise limitation, and to decide on extension of anticoagulant treatment.
dAlternatively, dual-energy CT may be used, if appropriate expertise and resources are available on-site.
eAs proposed in the algorithm shown in Figure 8.
fRisk factors and predisposing conditions for CTEPH are listed in Table 13.
. |
the use of compression stockings, safe increase in mobility, increased |
. |
|
. |
|
. |
awareness of signs of recurrence, or complications. |
. |
|
. |
|
. |
|
. |
|
. |
|
. |
|
. |
|
. |
|
. |
|
. |
11 Non-thrombotic pulmonary |
. |
|
. |
|
. |
|
. |
embolism |
.. |
|
. |
|
. |
|
. |
This section is included in the Supplementary Data available online |
. |
|
. |
|
. |
on the EHJ and ESC websites (www.escardio.org/guidelines). |
. |
|
. |
|
. |
|
. |
|
. |
|
. |
|
. |
|
. |
|
. |
|
. |
12 Key messages |
. |
|
. |
|
. |
|
. |
|
. |
The ESC Task Force has selected 10 simple key messages and rules |
. |
|
. |
|
. |
to guide physicians in the diagnosis and management of PE: |
. |
|
. |
|
. |
|
. |
|
. |
(1) In patients presenting with haemodynamic instability, perform bed- |
. |
|
. |
|
. |
side TTE as a fast, immediate step to differentiate suspected high- |
. |
|
. |
|
. |
risk PE from other acute life-threatening situations. |
. |
2019 September 13 on guest by 1093/eurheartj/ehz405/5556136.abstract/doi/10-article-com/eurheartj/advance.oup.https://academic from Downloaded