Материал: Bovine Viral Diarrhea Virus Diagnosis, Management, and Control
36 |
BVDV: Diagnosis, Management, and Control |
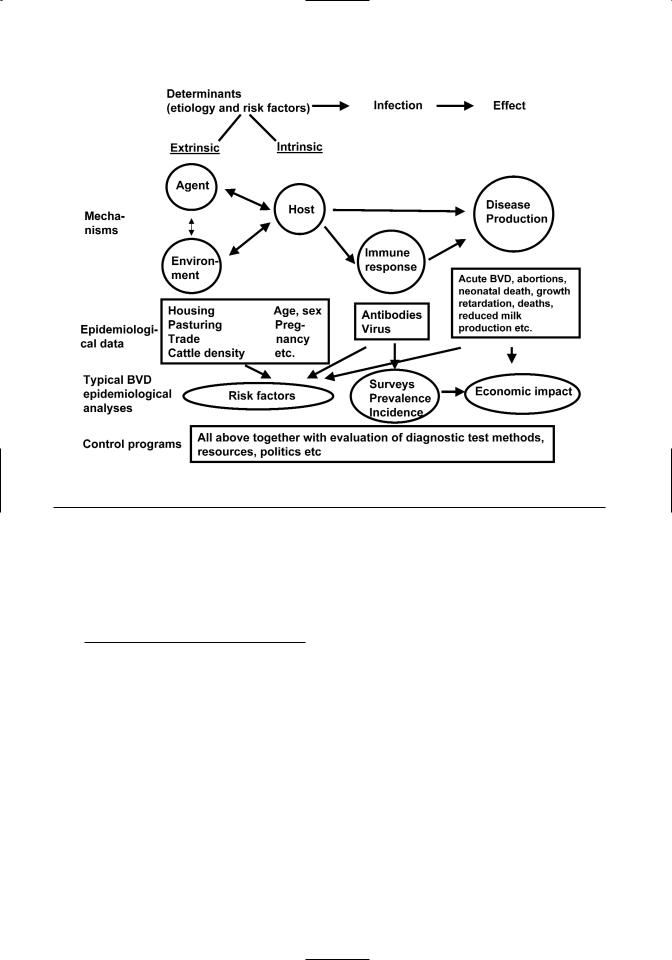
Figure 2.1. An epidemiological framework for the necessary data for describing BVDV occurrence, risk factors, and effect.
PREVALENCE OF BVDV INFECTIONS
The prevalence of a disease or infection (or any other condition) is defined as the proportion of the population with the disease at a given point in time:
= Number of diseased or infected animals at time t
p
Number of animals in the sample at time t
The prevalence can be interpreted as the probability that a randomly selected animal has the disease at time t. Thus, the prevalence of BVDV antibody carriers is the proportion of animals having antibodies, no matter when they were acquired. The advantage of a prevalence study is that animals need to be sampled only once. The disadvantage is that it is not known when the antibodies were acquired, or how. However, for PI animals we know that they were infected in the first trimester of fetal life (although viremic animals need to be retested after 2–3 weeks, it is still considered a prevalence study), and hence prevalence studies on the occurrence of PI animals are very useful.
Diagnostic methods for the detection of BVDV
and its antibodies are covered in detail in Chapter 12. In the context of describing occurrence, it is important to evaluate prevalence studies in relation to the sensitivity and specificity of the diagnostic tests. If, for example, a serologic test has revealed a prevalence of 45% antibody carriers (apparent prevalence, AP) and the test is known to have sensitivity (Se) of 97% and a specificity (Sp) of 88%, the true prevalence (TP) can be calculated from this formula:
TP = |
AP + Sp −1 |
= |
0.45 |
+ 0.88 −1 |
= 0.39 |
|
|
|
|||
|
Sp + Se −1 0.88 |
+ 0.97 −1 |
|||
where AP denotes apparent prevalence (the prevalence of test-positive animals). This means that the true prevalence is 39%. These calculations are made with the assumption that all serologically positive animals arose via natural infection. Antibodies titers that are due to vaccination will throw off these calculations.
For the detection of both virus and antibodies in individual animals, the sensitivities and specificities are usually very high and often close to 100%. For detection of antibodies, the Se and Sp of different

Risk Assessment |
37 |
ELISA tests as compared to serum neutralization test have often been higher than 90% (Cho et al., 1991: Se = 97.6% and Sp = 100%; Rønsholt et al., 1997: Se = 96.5% and Sp = 97.5%; Canal et al., 1998: Se = 97.5% and Sp = 99.2%; Kramps et al., 1999: Se = 98% and Sp = 99%). Comparison of BVDV antigen ELISA with conventional tissue culture isolation technique has also shown high sensitivity and specificity (Rønsholt et al., 1997: Se = 97.9% and Sp = 99.5%).
Many studies give estimates on the prevalence of infected herds. Here it is important to know that the definition of an infected herd can vary substantially and that the sensitivity and specificity can show higher variation and will often be known with a lower certainty than at the individual animal level. Correct classification of herds into being truly infected or noninfected has important implications for observational studies on epidemiological aspects— e.g., prevalence, incidence, identification of risk factors, and quantification of the effect of BVDV infection on disease and production.
The term herd test or herd diagnosis is based on testing samples from more than one animal. The sample can consist of separate samples from individual animals (e.g., blood samples) or it can be a pooled sample from several animals (e.g., pooled blood samples or a bulk tank milk sample). If the samples consist of individual animals with a sample size of n, and the herd is defined as positive if there is at least one positive animal in the herd, the herd sensitivity (HSe) and herd specificity (HSp) can be calculated from the prevalence of test-positive animals (AP) and from the specificity at animal level (Sp) as follows:
HSe = 1 − (1 − AP)n
and
HSp = Spn
From the formulas it can be seen that increasing the sample size will result in increased herd sensitivity but decreased herd specificity. Furthermore, a high prevalence would increase herd sensitivity but would not affect herd specificity. These formulas are true only when definition of a herd is based on one or more animals being positive (i.e., the critical number is 1). Sometimes, a herd is defined positive only if a higher number of animals are positive and in these cases other approaches are needed (Martin et al., 1992). In many published epidemiological studies the herd prevalence is given based on at least
one animal being positive for either antibody or virus.
The herd diagnoses for the presence of PI animals are often based indirectly on an interpretation of serological test results of screening young stock, from antibody reaction in bulk tank milk or combinations hereof. The true status should then preferably be based on examination of all individual animals. In 26 herds where blood testing of all animals was used as the gold standard, the HSe and HSp of using a small screening sample of young stock was calculated as 0.93 and 1.0, respectively (Houe, 1999). Others have reported HSe and HSp of 0.66 and 1.0, respectively, using serologic results from unvaccinated heifers among 14 dairy herds (Pillars et al., 2002). The reason for a low HSe can be that the PI animals are young (Houe, 1992b; Houe, 1999; Pillars et al., 2002) or it has even occurred that by chance the PI animals were selected for serologic testing giving negative results (Pillars et al., 2002). Therefore repeating the serologic testing of young stock a few months later and also having them tested for virus will increase HSe significantly.
The antibody level in bulk tank milk has shown good correlation with the prevalence of antibody carriers in a herd (Niskanen, 1993; Houe, 1994; Beaudeau et al., 2001). Using a screening sample of young stock as a gold standard, a blocking ELISA measuring antibody levels in bulk tank milk was evaluated. Among 352 dairy herds the bulk tank milk antibody test had a sensitivity of 1 and a specificity of 0.62 at a blocking percentage (cutoff) of 50% (Houe, 1999). Furthermore, at an estimated herd prevalence of 26%, the positive and negative predictive values were 0.48 and 1.00, respectively. This means that the bulk tank milk test is very good at detecting herds with true PI animals. On the other hand, many herds without PI animals may also have high antibody levels (e.g., due to a PI animal moved from the herd before the test), and therefore the test result is false positive. In some areas, there may be poor predictive values of antibody detection among a few young stock or antibody level in bulk milk for herd diagnosis because of high prevalence of infection and large variation of antibody-positive animals in herds without PI animals (Zimmer et al., 2002).
Many studies use an increase in bulk tank milk antibody level as an indication of a recent infection in the herd (Niskanen et al., 1995). Also PCR on bulk milk has been used to directly detect virus excretion in the herd (Drew et al., 1999). Unfortunately, there are no international standards for clas-

38 |
BVDV: Diagnosis, Management, and Control |
sification of herd infection status, and one should be careful when comparing studies that use different definitions for an infected herd.
EPIDEMIOLOGICAL STUDIES FOR
ESTIMATION OF PREVALENCE
Due to the development of faster and cheaper diagnostic methods the number of prevalence studies has increased significantly in recent years. When comparing different studies one should be aware of differences in study design, study period, sampling frame, sampling method, sampling units, sample size, and the exact test variable being measured.
Tables 2.1–2.3 summarize a number of prevalence studies including some keyword information on sampling frame and sampling methods. These studies were selected to represent a broad variety of different geographical regions. Another criterion for selection was that the studies contained sufficient information on study design and sampling strategies to be judged fairly representative for the study area. Attempts were made to include both older and newer prevalence studies. Studies in countries with national control and eradication programs are from before or at the beginning of the eradication programs.
The sampling frame in the tables specifies whether samples have been taken from certain regions in the country and whether there is information on the type of enterprise (dairy herds, beef herds, breeding herds, or artificial insemination centers). The exact sampling methods are often rather complicated and for details the reader is referred to the cited publications. The tables include information on the major criteria for sampling such as the randomness and representativeness of the samples and whether only certain age groups are sampled. The sampling has often been done by initially selecting herds (primary sampling units) followed by sampling of individual animals (secondary sampling units). But there are also examples where animals are the primary sampling unit (e.g., sampling from abattoirs) or where herds are sampled without further sampling of animals (e.g., testing for antibody level in bulk milk, as shown in Table 2.3).
The variables behind the prevalence measures when testing individual animals are either antibodies (Table 2.1) or virus (Table 2.2). The same study can, therefore, be represented in both tables if animals are tested for both antibodies and virus. The prevalence can be given both at the animal level as well as at the herd level (here defined as the prevalence of herds with any antibody or virus-positive animal). In addition, many studies aim directly at
obtaining a herd infection status by serological testing of a few young stock, testing the bulk tank milk for antibody or virus, or by various combinations of these methods. These studies provide prevalence estimates of herds with any antibody carriers or presence of any PI animals (or rather herds being suspected of having PI animals). As these studies are often based on different interpretations of antibody level in bulk tank milk, they are presented separately in Table 2.3 (some keywords of the interpretation from the studies are given in the table). Altogether, the following six prevalence measures are presented in Tables 2.1–2.3:
1.Prevalence of antibody-positive animals
2.Prevalence of virus-positive animals
3.Prevalence of herds with at least one antibody carrier
4.Prevalence of herds with at least one viruspositive animal
5.Prevalence of herds with antibody-positive animals as detected from screening samples (spot tests) and bulk milk
6.Prevalence of herds with high probability of virus-positive animals as detected from screening samples (spot tests), antibody level in bulk milk, or PCR on bulk milk
One of the difficulties of interpreting serological results is the very different use of vaccines. When there were clear indications of the use of vaccine, either in the country or in the study herds, this information is provided in the tables. One should also be careful interpreting the prevalence of infected herds if not all animals in the herd are tested.
Due to the various sampling strategies and interpretation of test results, formal statistical analyses of Tables 2.1–2.3, estimation of confidence intervals (CI) of the true occurrence, and testing for differences between different areas are not straightforward. Instead, many different studies are outlined in the tables in order to provide the reader with a spectrum of results from different areas, emphasizing some selected trends. The reader should be aware of the sampling methods when comparing prevalence. If, for example, only younger animals were sampled for the detection of PI animals, the prevalence would be higher than if sampling was done among all animals (simply because PI animals die faster than other animals).
Considering the prevalence of antibody carriers at the animal level, there seems to be a continuum of prevalence levels from a minimum of 12% to a maximum of 89% (Table 2.1). However, if all the pre-

39
Table 2.1. Epidemiological studies for estimation of prevalence of animals with antibodies against BVDV
|
|
|
Sampling Method |
Sample Size |
Prevalence |
|
|
|||
|
|
|
|
|
|
|
|
|
|
|
Country/ |
Study |
|
|
|
|
|
Herd |
Animal |
|
|
Region |
Period |
Sampling Frame |
Herds |
Animals |
Herds |
Animals |
Level (%) |
Level (%) |
Vaccination |
Reference |
|
|
|
|
|
|
|
|
|
|
|
Europe |
|
|
|
|
|
|
|
|
|
|
Belgium |
. . . |
Southern Belgium |
42.5% of herds had |
All animals in herd |
61 |
9685 |
100 |
66 |
Some vaccination |
Schreiber et al., |
|
|
Belgian White Blue |
prior diagnosis or |
|
|
|
|
|
(not considered |
1999 |
|
|
and Friesian |
were suspicious |
|
|
|
|
|
important) |
|
|
|
Holstein |
|
|
|
|
|
|
|
|
Denmark |
1988 |
Jutland in Denmark |
Representative |
All animals in herd |
19 |
2570 |
100 |
64 |
No vaccination |
Houe and Mey- |
|
|
Dairy herds |
NPE |
|
|
|
|
|
|
ling, 1991 |
Netherlands . . . |
. . . |
. . . |
Random among ani- |
>100 |
1798 |
— |
65 |
. . . |
Kramps et al., |
|
|
|
|
|
mals in BHV1a |
|
|
|
|
|
1999 |
|
|
|
|
diagnostics and |
|
|
|
|
|
|
|
|
|
|
field trial |
|
|
|
|
|
|
Norway |
1984–1986 |
Wide geographic |
Geographic repre- |
Random |
187 |
1133 |
28 |
19 |
No vaccination |
Løken et al., 1991 |
|
|
representation |
sentative, NPE |
>2 years |
|
|
|
|
|
|
|
|
Dairy herds |
|
|
|
|
|
|
|
|
Poland |
. . . |
Bulls at artificial |
— |
>6 months old |
— |
175 |
— |
86 |
. . . |
Polak and |
|
|
insemination |
|
|
|
|
|
|
|
Zmudzinski, |
|
|
centers |
|
|
|
|
|
|
|
1999 |
Scotland |
1992–1993 |
South West Scotland |
. . . |
Random |
78 |
109 |
— |
78 |
. . . |
McGowan and |
|
|
Breeding bulls |
|
|
|
|
|
|
|
Murray, 1999 |
|
|
on dairy, beef of |
|
|
|
|
|
|
|
|
|
|
mixed farms |
|
|
|
|
|
|
|
|
|
|
(5 bulls from |
|
|
|
|
|
|
|
|
|
|
dealers) |
|
|
|
|
|
|
|
|
Slovenia |
1996 |
5 regions |
. . . |
All animals in herd |
274 |
6892 |
. . . |
17 |
. . . |
Grom et al., 1999 |
|
|
Breeding herds |
|
|
|
|
|
|
|
|
Spain |
1997 |
Asturias region |
Random/stratified |
20 herds: all animals |
28 |
529 |
86 |
21 |
No vaccination |
Mainer-Jaime et |
|
|
Dairy herds |
NPE |
8 herds: Random, |
|
|
|
|
|
al., 2001 |
|
|
|
|
>1-year-old |
|
|
|
|
|
|
Sweden |
. . . |
11 counties in dif- |
NPE |
Breeding heifers |
114 |
711 |
— |
41 |
No vaccination |
Alenius et al., |
|
|
ferent parts of |
|
|
|
|
|
|
|
1986 |
|
|
Sweden |
|
|
|
|
|
|
|
|
Sweden |
1987 |
County of Koppar- |
Random |
All lactating cows |
15 |
413 |
73 |
46 |
No vaccination |
Niskanen et al., |
|
|
berg |
|
|
|
|
|
|
|
1991 |
|
|
Dairy herds |
|
|
|
|
|
|
|
(continued) |

40
Table 2.1. Epidemiological studies for estimation of prevalence of animals with antibodies against BVDV (continued)
|
|
|
Sampling Method |
Sample Size |
Prevalence |
|
|
|||
|
|
|
|
|
|
|
|
|
|
|
Country/ |
Study |
|
|
|
|
|
Herd |
Animal |
|
|
Region |
Period |
Sampling Frame |
Herds |
Animals |
Herds |
Animals |
Level (%) |
Level (%) |
Vaccination |
Reference |
|
|
|
|
|
|
|
|
|
|
|
Switzerland |
1994–1995 Canton of St. Gallen |
Random |
Cows and heifers |
95 |
2892 |
100 |
84 |
. . . |
Braun et al., 1997 |
|
|
|
Dairy herds |
|
(All) |
|
|
|
|
|
|
Switzerland |
1995 |
Canton of St. |
Invited by cantonal |
Animals prior to |
149 |
990 |
— |
63 |
. . . |
Braun et al., 1998 |
|
|
Gallen, 7 Alpine |
veterinary officer |
pasture, 98% |
|
|
|
|
|
|
|
|
pastures |
|
were replacement |
|
|
|
|
|
|
|
|
Swiss Braunvieh |
|
cattle NPE |
|
|
|
|
|
|
|
|
cattle |
|
|
|
|
|
|
|
|
|
|
Dairy herds |
|
|
|
|
|
|
|
|
Switzerland |
1993–1994 |
Dairy herds |
Random (at least |
All cows |
113 |
1635 |
99 |
72 |
. . . |
Stärk et al., 1997 |
|
|
|
5 cows) |
|
|
|
|
|
|
|
United |
1974–1975 |
England and Wales |
3 herds in each |
12 per herd repre- |
133 |
1593 |
— |
62 |
. . . |
Harkness et al., |
Kingdom |
|
|
county |
senting a range |
|
|
|
|
|
1978 |
|
|
|
|
of ages |
|
|
|
|
|
|
United |
1985–1986 |
England and Wales |
— |
Submissions of |
— |
18,759 |
— |
65 |
. . . |
Edwards et al., |
Kingdom |
|
|
|
more than 10 |
|
|
|
|
|
1987 |
|
|
|
|
samples to Cen- |
|
|
|
|
|
|
|
|
|
|
tral Veterinary |
|
|
|
|
|
|
|
|
|
|
Laboratory |
|
|
|
|
|
|
North, Central and South America |
|
|
|
|
|
|
|
|
||
Canada |
. . . |
Saskatchewan and |
Several sources: Brucellosis certification, |
295 |
1745 |
(67)b |
41 |
Some vaccination. |
Durham and |
|
|
|
Alberta |
bull test station, research station and |
|
|
|
|
One-third of |
Hassard, 1990 |
|
|
|
Several breeds |
others |
|
|
|
|
|
vaccinated had |
|
|
|
Especially beef |
|
|
|
|
|
|
antibodies |
|
Canada |
1990–1991 |
Northwestern |
Random |
Systematic random |
15 |
311 |
93 |
53 |
Only nonvacc. |
Ganaba et al., |
|
|
Quebec, beef |
>25 breeding |
among pregnant. |
|
|
|
|
within 1 year |
1995 |
|
|
herds (cow calf) |
females |
12–25 cows per |
|
|
|
|
included here |
|
|
|
|
|
herd |
|
|
|
|
|
|
Canada |
. . . |
3 Maritime |
Stratified, two stage |
5 cows or 5 heifers |
89 |
445 |
(66) |
38 |
Tested animals |
VanLeeuwen et al., |
|
|
provinces: New |
random sample |
>6 months old |
|
|
|
|
not vaccinated |
2001 |
|
|
Brunswick, Nova |
|
|
|
|
|
|
|
|
|
|
Scotia, Prince |
|
|
|
|
|
|
|
|
|
|
Edward Island |
|
|
|
|
|
|
|
|
|
|
Dairy herds |
|
|
|
|
|
|
|
|