Материал: Bovine Viral Diarrhea Virus Diagnosis, Management, and Control
Vaccines |
211 |
of the vaccines in use from the 1960s through the 1990s to confer protection against BVDV 2 has not been established. The vaccines in use since the 1960s had been, in retrospect, BVDV 1a cp strains, such as the Singer, NADL, and C24V (Oregon) strains. Questions then arose as to whether to accept or prove that BVDV 1 vaccines protected against BVDV 2 when used according to the label or whether BVDV 2 strains should be added to the BVDV 1a vaccines.
VACCINE STRAINS
Virus strains being used in various BVDV vaccines are shown in Table 13.1. In 2002, the genotype BVDV 2 was further divided into subgenotypes, BVDV 2a and BVDV 2b (Flores et al., 2002). Selected BVDV strains have been subtyped (J.F. Ridpath, personal communication, 2003) and those are reflected in the Table 13.1. The vaccines in the U.S. primarily contain BVDV 1a cp strains, although some vaccines do contain BVDV 2 cp strains (refer to Table 13.1). All but four vaccines contain cp biotypes; only two killed and two MLV vaccines contain ncp strains. Of the latter vaccines, one contains a ncp strain, WRL, referred to as BVDV 1 without any reference to the subtype, and another contains a BVDV 1 ncp strain (GL 760) and a subtype is not described. One killed vaccine contains
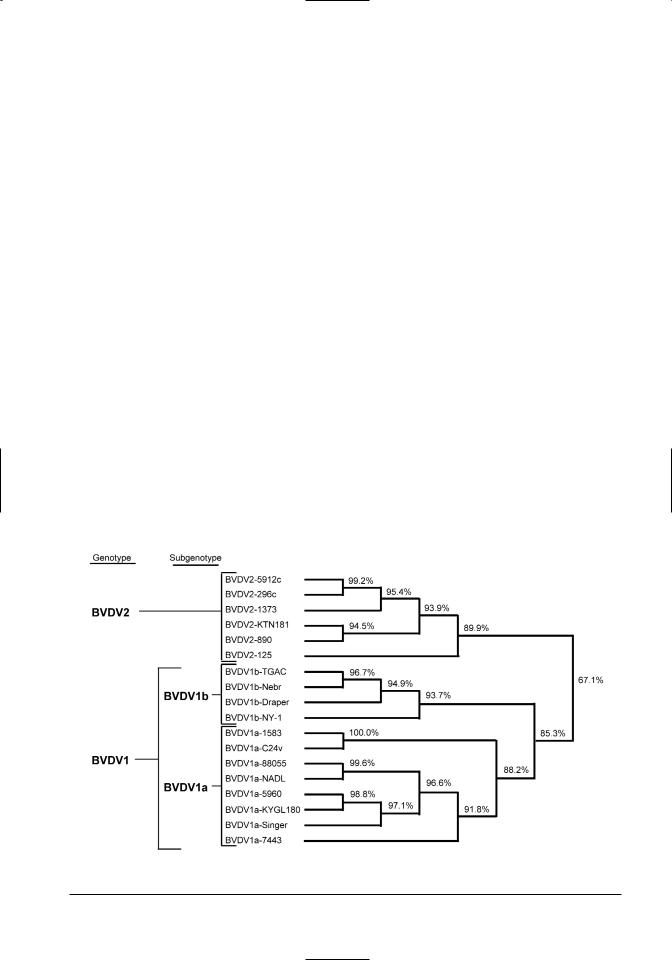
strain 6309, a BVDV 1 ncp strain whose subtype is not specified. Only one killed vaccine contains ncp BVDV 1b. A dendrogram representing genetic relatedness for reference BVDV strains, including many vaccinal strains, is shown in Figure 13.1. There are several companies in the U.S. that manufacture vaccines or single/multiple immunogens that are marketed by other companies. Thus, vaccines sold under different names may have identical immunogens.
Isolation of BVDV 1b and BVDV 2 from clinic cases cited above demonstrates the need for BVDV vaccines that confer a broad spectrum of complete immunity to these multiple BVDV subtypes. The addition of BVDV 2 immunogen to either MLV or killed BVDV 1a vaccines has been the subject of marketing efforts by certain companies; some other companies, rather than including type 2 immunogens in their vaccine, have submitted data that satisfied licensing requirements allowing a label claim for cross protection against BVDV 2.
MLV VACCINES
The advantages and disadvantages of MLV BVDV vaccines are similar to those of other MLV vaccines. The MLV vaccines require lesser amounts of virus than do killed vaccines because the vaccine replicates in the host to build immunogenic mass. In general, MLV vaccines require only one dose for initial
Figure 13.1. Dendogram representing the relatedness of nucleotide sequences from reference BVDV 1a, BVDV 1b, and BVDV 2 strains.

212 |
BVDV: Diagnosis, Management, and Control |
immunization but do require more rigid handling procedures because the vaccine virus is susceptible to inactivation by chemicals and/or exposure to higher temperatures. Upon administration of MLV vaccine, the BVDV vaccinal strains replicate in the susceptible bovine, resulting in viremia (Cortese et al., 1997; Fulton et al., 2003c; Grooms et al., 1998). The duration of viremia is between 3 and 7 days after which the virus is cleared as the calves develop antibodies (Fulton et al., 2003c). Thus vaccinal strains must be differentiated from field strains if BVDV is isolated from calves within 2 weeks after MLV vaccination. Live virus in MLV vaccine may also cause immunosuppressive effects on leukocyte function of vaccinated cattle (Roth and Kaberle, 1983), potentially rendering them more susceptible to other infections. Another concern is the effect of live vaccine virus on the reproductive tract, because BVDV virus or antigen was detected in the ovaries of heifers receiving an MLV BVDV vaccine up to 30 days prior to testing (Grooms et al., 1998).
A major concern for MLV BVDV use was the observation of a postvaccinal disease following MLV vaccination. Within 1–4 weeks after MLV vaccination, a mucosal disease (MD)–like syndrome occurred in cattle (Bittle, 1968; McKercher et al., 1968; Peter et al., 1967; Rosner, 1968; Lambert, 1968). Postvaccinal MD may result when an animal persistently infected with a ncp virus is exposed to the cp BVDV included in the vaccine. However, vaccination of PI animals with an MLV vaccine will not invariably cause MD. There have been multiple studies whereby PI calves have been given MLV BVDV vaccines with cp strains and no MD resulted (Bolin et al., 1985b; Bolin et al., 1988; Fulton et al., 2003b). Those studies used cp strains that were antigenically different from the PI strains, and the calves developed antibodies to the cp strain in the vaccines. Thus PI animals may not be cleared from herds by vaccination, and failure to develop antibody titers following vaccinations is not a good method to screen for PI animals.
In addition, postvaccinal disease can result from infection with adventitious ncp field strains of BVDV introduced via contaminated fetal bovine serum or cell lines used in vaccine manufacturing. The ncp BVDV is a frequent contaminant of fetal bovine serum (Bolin and Ridpath, 1998; Studer et al., 2002). An example of BVDV contamination of an MLV vaccine occurred when vaccination with a BHV-1 marker vaccine with BVDV 2 contamination caused severe disease beginning 6 days after vaccination with high morbidity on 11 of 12 farms
(Barkema et al., 2001). Signs included nasal discharge, fever, and diarrhea. Necropsy revealed erosions and ulcers of mucosa of the digestive tract.
The ability of BVDV to infect the fetus resulting in abortions, stillbirths, and development defects has caused MLV BVDV vaccines to be contraindicated in pregnant cattle (Orban et al., 1983; Liess et al., 1984). In one study, MLV vaccine containing a BVDV 1a cp strain (C24V) was used in pregnant heifers resulting in fetal infection and disease. In addition there were ncp strains isolated from affected calves diagnosed as PI. The source of the ncp strains was not identified; however, a cp vaccine containing ncp strains is possible. Another study indicated that cp challenge inoculums may give rise to ncp strains (Done et al., 1980). Pregnant heifers were inoculated with a pool of BVDV cp strains (10), and transplacental infections occurred with all ncp strains being isolated, but no cp isolates.
Potentially a vaccine containing a ncp BVDV contaminant could induce a PI calf if the fetus were exposed between days 42–125 of gestation (McClurkin et al., 1984). Fetuses exposed in the last trimester of pregnancy could survive, but may have antibody titers in the precolostral serums after calving. Data suggest that MD was induced after recombination between a ncp BVDV 2 strain with a BVDV 1a cp NADL vaccine strain (Ridpath and Bolin, 1995). The cp strains can cause fetal infections; however, experimental inoculation of pregnant heifers with a cp strain resulted in no PI calves (Brownlie et al., 1989). The potential for a replicating vaccine virus or a contaminating ncp to be transmitted to susceptible contacts must be addressed for any MLV vaccine. Although an MLV BVDV may replicate in a susceptible animal causing viremia, virus may or not be shed. More importantly, there must be sufficient virus shed to infect contacts as indicated by viremia and/or seroconversion. In a recent study (Fulton et al., 2003c) calves vaccinated with each of three BVDV 1a MLV vaccines developed a transient viremia that was cleared after antibodies were induced; however, there was no transmission of the virus to susceptible contact calves in the same pen. The use of ear notch immunohistochemistry (IHC) for BVDV antigen did not detect IHC positives in a preliminary study testing calves after they received MLV BVDV vaccine (Dubois et al., 2000).
KILLED VACCINES
Killed vaccines also have advantages and disadvantages. From a production cost standpoint, killed vac-

Vaccines |
213 |
cines are expensive because larger amounts of virus are required to prepare each dose of the vaccine as compared to MLV vaccines and there is the added cost of adjuvants. The process of virus inactivation for the production of killed vaccine is likely to also inactivate possible contaminants if any; however, this is not guaranteed unless the final product is tested for replicating virus. Killed BVDV vaccines are generally safer in the pregnant cow, and some vaccination programs advocate killed BVDV vaccines during pregnancy. One disadvantage might be that two doses are generally required for the initial immunization. Both MLV and killed BVDV vaccines have induced antibodies to a wide range of BVDV subtypes, usually resulting in higher antibodies to the specific BVDV subtype(s) in the respective vaccine (Fulton et al., 1997; Fulton and Burge, 2000).
The duration of antibody titers to various BVDV strains was found to vary among different studies. Cortese et al. (1998a) reported that cattle receiving an MLV BVDV 1a (NADL) vaccine induced antibodies to numerous BVDV 1 and BVDV 2 strains detectable through 18 months after vaccination. In other studies, there was a decline in BVDV antibodies by 140 days after initial vaccination (Fulton et al., 1995; Fulton and Burge, 2000). Revaccination at day 140 with either killed or MLV vaccine did induce increased antibodies in calves, especially those with low antibody titers (Fulton et al., 1995 and Fulton and Burge, 2000). This rapid anamnestic response points out that, while antibody titers may decline or disappear, an improved immune response remains in effect. From this standpoint, the true duration of immunity cannot be determined by just measuring serum antibody levels but should rather be determined by challenge with virulent virus.
PROTECTION BASED ON DISEASE FORM
The efficacy of a BVDV vaccine should be measured by three different methods (Van Oirschot et al., 1999). The first is experimental vaccination under controlled conditions followed by direct experimental viral challenge and observation of clinical signs. Secondly, there should be transmission studies to determine whether the vaccine prevents or reduces the transmission of the challenge virus. Finally, field trials of the vaccine are needed to determine protection/cost benefits under production conditions and natural exposure. To truly help dairy and beef cattle clinicians and producers make rational decisions on appropriate vaccine use, the
studies outlined above should go beyond the minimal licensure requirements.
Recently the USDA APHIS CVB began requiring data on file to support label claims. In an ideal setting, methods should be available for veterinarians and the public to analyze the documentation for approved and marketed vaccines. All too frequently, the public’s only access to vaccine efficacy information is from marketing materials and advertisements for vaccines. Use of peer-reviewed publications would be an appropriate means to that end for public information. Intellectual property issues should be honored, but the experimental design, results, statistical analysis, and interpretation (discussion) should be available to allow producers and veterinarians to make informed decisions.
MECHANISM OF PROTECTION FOR BVDV
Cattle are capable of mounting both humoral (antibody) and cell-mediated (T-cell) immune responses to BVDV. These responses occur after vaccination or field/natural infection of susceptible, seronegative cattle. As cited above, the vaccines induce antibodies to multiple BVDV subtypes, but antibody titers are generally higher to the vaccine strain and to strains belonging to the same genotype or subgenotype. Numerous vaccine efficacy/immunogenicity studies in which antibody titers have been measured are available in published literature. Unfortunately, in many cases, the authors do not identify the BVDV subtype in either the vaccine, natural infection, or challenge strain. The challenge strain used in the laboratory for a virus neutralization test is also not always known. Our current understanding of antigenic diversity among genotypes and subgenotypes calls for more attention to subtypes in future immunity studies.
The fact that antibodies provide protection is clear from studies that demonstrate passive protection against BVDV challenge in calves fed colostrum containing BVDV antibodies (Howard et. al., 1989; Bolin and Ridpath, 1995). Resistance either to disease severity or viral infection/shedding was dependent upon BVDV antibody titers in the sera. Cortese et al. (1998) showed that calves fed colostrum with BVDV antibodies were protected against experimental BVDV. However, high concentrations of maternally derived BVDV antibodies have been shown to block a protective response to MLV BVDV 1a NADL vaccine (Ellis et al., 2001). Kirkpatrick et al. (2001) reported that dairy calves fed colostrum with BVDV antibodies had low BVDV antibody titers of

214 |
BVDV: Diagnosis, Management, and Control |
1:4–1:16 and did not seroconvert to either BVDV 1a or BVDV 2 after vaccination with an MLV BVDV 1a and BVDV 2 vaccine.
Postnatal calves develop T-cell immune responses after vaccination or infection. It is assumed that these calves can develop concurrent humoral antibody and T-cell responses. Trying to determine which arm of the acquired immunity (humoral or cell-mediated) is more important in protection is difficult because in the intact, healthy susceptible animal, it is hard to immobilize one arm of the immune system to determine its role in protection. Beer et al. (1997) demonstrated that immunized and experimentally exposed calves responded with positive cytotoxic T-cell responses, along with high neutralizing antibody titers. More recently, Endsley et al. (2002) reported that calves developed antigenspecific T-cell responses when given either a BVDV 1 MLV or BVDV 1 and BVDV 2 MLV vaccine. Information on humoral antibody responses to BVDV 1 and BVDV 2 was not provided. Using a variety of tests for T-cell immunity and antibodies, the protective immune mechanism was not apparent in calves receiving an MLV BVDV 1 vaccine and subsequently challenged with heterologous BVDV 2 (Cortese et al., 1998b).
Passively acquired BVDV antibodies prevented clinical disease in colostrum-fed calves exposed to virulent BVDV (Ridpath et al., 2003). The serum antibody titers decayed at the same rate as in unchallenged colostrum-fed calves. These same colostrums-fed calves previously challenged were still protected from clinical disease after the serum antibodies had decayed to undetectable levels. A protective response was thus mounted in calves with passive immunity but which was not reflected by antibody titers. According to the authors, cellmediated immunity may have a role in preventing postnatal disease.
EFFICACY STUDIES
In a review of published studies for both postnatal calves and fetal infection/disease, van Oirschot et al. (1999) cited numerous trials in both sheep and cattle for the prevention of fetal infections and trials in cattle for preventing postnatal infections. In that review, numerous reports were cited indicating the use of sheep as a model for BVDV vaccine development and evaluation. Although sheep are an active, inexpensive BVDV model, this chapter focuses on studies of current North American vaccines in cattle. Selected other studies will be included that address vaccine technology and experimental design, which
have worldwide relevance. Until recently, the impact of BVDV antigenic diversity on vaccines and challenge models had not been addressed. In addition, only recently efforts have been made to study vaccine protection against fetal infections. One of the few early studies was done by Duffel et al. (1984) who reported that immunity to BVDV was induced in heifers prior to breeding, and that their fetuses were protected after challenge at day 100 of gestation (Duffel et al., 1984).
This chapter updates previous published studies with the current designation of subtypes. However, it should be realized that a large database of all known BVDV strains with respect to specific subtype designations does not exist. Experimental studies are available that have evaluated both killed and MLV vaccines. The biotype/genotype in the vaccines until recently was confined to the BVDV 1a cp strains. Likewise, challenge strains have varied, as have experimental challenge methods and parameters for evaluation of clinical illness. In general, the experimental challenge has been relatively soon after the last vaccine dose, usually 14–28 days, and the duration of immunity for the postnatal protection and fetal protection studies has not always and not uniformly been performed. The general consensus for postnatal protection is that there is incomplete protection against clinical signs/disease. Brock and Chase (2000) described an experimental protocol to evaluate fetal protection in cattle by BVDV vaccines.
PROTECTION AGAINST POSTNATAL
INFECTION/DISEASE
A combination killed BVDV vaccine containing a BVDV 1a cp strain and a BVDV 1 ncp strain (subtype not designated in Table 13.1) given in two doses induced BVDV antibodies, and vaccinates were protected against clinical illness when compared to controls. The challenge virus, BVDV 1b ncp New York (NY-1) strain, was given 14 days after the second dose. The antibody titers of vaccinates at the time of challenge ranged between 1:16 and 1:128 (mean = 1:42.7). The strain used in neutralization assay was not identified. The protection was not complete because some vaccinates had illness (Talens et al., 1989).
Calves given a MLV vaccine containing the BVDV 1a NADL strain were challenged with BVDV 1b ncp NY-1 strain on postvaccination day 27. None of the vaccinated calves had clinical signs in the 14day postchallenge period nor did they have rectal temperatures above 39.7°C (Phillips et al., 1975). With the evident need for protection against BVDV

Vaccines |
215 |
Table 13.1. Strains and types of bovine viral diarrhea virus (BVDV) in modified-live virus (MLV) and killed vaccines.
Vaccine Type |
|
|
|
and Trade Name |
Name of Strain |
Genotype/Biotype |
Company |
|
|
|
|
MLV |
|
1 ncp1 |
|
Arsenal 4.1 |
GL 760 |
Novartis |
|
Express 5 |
Singer |
1a cp |
Boehringer Ingelheim Vet Medica |
|
296 |
2a cp2 |
|
BoviShield 4 |
NADL |
1a cp |
Pfizer Animal Health |
Pyramid 4 |
Singer |
1a cp |
Fort Dodge Animal Health |
Reliant 4 |
NADL |
1a cp |
Merial |
Frontier 4 Plus |
C24V |
1a cp |
Intervet |
|
296 |
2a cp2 |
|
Titanium 5 |
C24V |
1a cp |
Agrilabs |
|
296 |
2a cp2 |
|
Jencine 4 |
WRL |
1 ncp |
Schering-Plough Animal Health |
Herd Vac 3 |
Singer |
1a cp |
Biocor Animal Health |
Bovine Viral |
|
|
|
Diarrhea Vaccine |
C24V |
1a cp |
Colorado Serum Co. |
Killed |
|
|
|
Elite 4 |
Singer |
1a cp |
Boehringer Ingelheim Vet Medica |
Horizon 4 Plus |
C24 V |
1a cp |
Intervet |
|
125C |
2a cp |
|
Master Guard 5 |
C24 V |
1a cp |
Agrilabs |
|
125C |
2a cp |
|
Respishield 4 |
Singer |
1a cp |
Merial |
Triangle 4 + type II |
Singer |
1a cp |
Fort Dodge Animal Health |
|
5912 |
2a cp |
|
CattleMaster 4 |
5960 |
1a cp |
Pfizer Animal Health |
|
6309 |
1 ncp |
|
ViraShield 5 |
KY22 |
1a cp |
Grand Laboratories |
|
TN 131 |
2 ncp |
|
Surround 4 |
Singer |
1a cp |
Biocor Animal Health |
|
NY |
1b ncp |
|
cp = cytopathic; ncp = noncytopathic 1Information provided by the company.
2Based on sequencing information by Dr. J. F. Ridpath, USDA, ARS, NADC.
2 strains, there have been published studies where BVDV 1-vaccinated cattle have been protected when challenged by BVDV 2. Cortese et al. (1998b) reported a study in which a BVDV 1a NADL vaccinal strain was given to seronegative 10–14-day-old dairy calves and the calves then challenged intranasally with a virulent BVDV 2 24515 ncp strain 21 days later. The antibody titers at the time of challenge
ranged from negative to 1:66 (mean = 1:15) to BVDV 1a NADL and negative to 1:6 (mean = 1:1) to BVDV 2 125C. Vaccinated calves were protected against clinical signs and viremia in the 14-day postchallenge observation period.
A related study by Ellis et al. (2001) indicated that the MLV BVDV 1a NADL vaccinal strain was given to 10–14-day-old or 4-month-old seronegative