Материал: Bovine Viral Diarrhea Virus Diagnosis, Management, and Control

Pathogenesis |
133 |
posure to a cp BVDV strain, and the re-isolated cp BVD is identical to the one the animal was exposed to. Late onset MD occurs months to years after exposure and the re-isolated cp BVDV is a recombination between the persisting ncp and the original cp BVDV. This distinction is in general possible only under experimental conditions, because in most field cases the identity of the original infecting cp BVDV cannot be determined and the time of exposure cannot be pinpointed. Clinical signs in the end phase of early onset and late onset MD are indistinguishable and there are only subtle differences in tissue lesions of animals that have succumbed to the two forms of MD (Moennig et al., 1993; LieblerTenorio et al., 2000).
PATHOGENESIS OF LESIONS IN MUCOSAL
DISEASE
Correlation between viral antigen and tissue lesions
In field cases of MD, BVDV antigen can be demonstrated by immunohistochemistry in numerous organs and tissues (Meyling, 1970; Bielefeldt Ohmann, 1983). The distribution of viral antigen does not correlate well with tissue lesions, since in general no distinction can be made between the antigen of the ncp and cp BVDV. The ncp BVDV present in PI animals in a wide distribution is still found in animals after they have developed MD, but is not associated with tissue lesions (Bielfeldt Ohmann, 1988). Under experimental conditions, it is possible to selectively demonstrate the distribution of cp BVDV as opposed to the persistent ncp BVDV (Liebler et al., 1991). These conditions require that the cp BVDV used for inoculation differs in binding to one or more monoclonal antibodies from the persisting BVDV. Thus it can be recognized selectively by specific monoclonal antibodies in tissue sections using immunohistochemistry. Further studies indicated that the organ distribution patterns for the cp BVDV were identical whether immunohistochemistry for E2 or whether molecular detection of NS3 protein characteristic for the cp BVDV was used (GreiserWilke et al., 1993). This suggests that results obtained by immunohistochemistry reflect the distribution of cp BVDV in the tissues realistically.
Examination of animals with MD revealed that cp BVDV was predominantly detected in sites where tissue destruction was observed (Liebler et al., 1991). The sites included
tissues (gut-associated lymphoid tissues, bronchus-associated lymphoid tissues)
•Thymic cortex
•Altered mucosa of the upper digestive tract, and (iv) crypt epithelia of small and large intestine
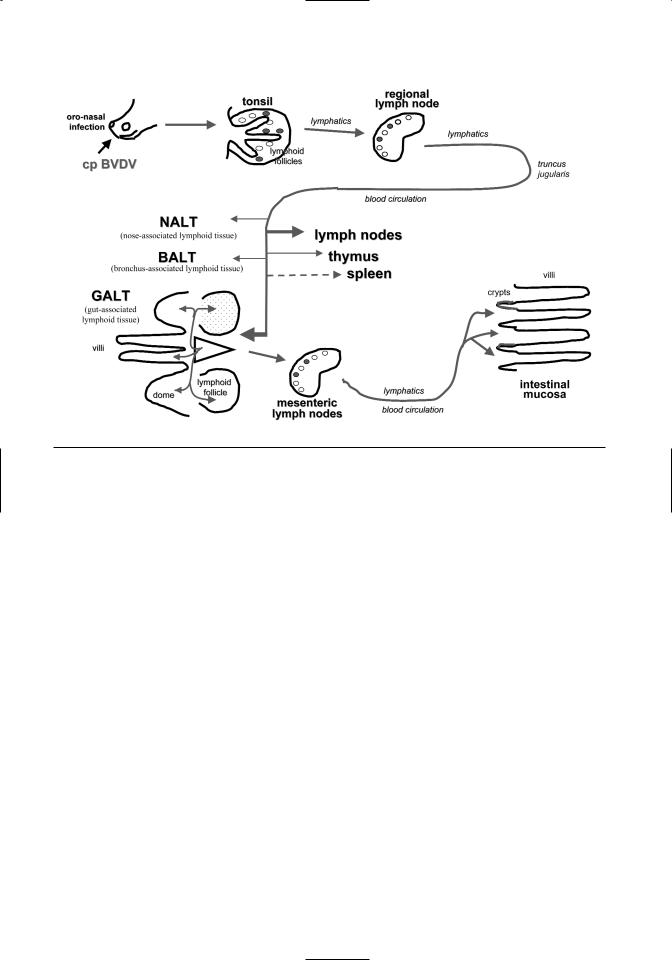
These observations suggest that cp BVDV is not only important for the pathogenesis of MD, but also for the manifestation of MD at mucosal surfaces and in lymphoid tissues. It appears that the replication of cp BVDV is associated with marked lesions. The comparison of tissue lesions and distribution of viral antigen at consecutive times after inoculation of PI cattle with cp BVDV revealed a spreading pattern for the cp BVDV in MD that was similar to the one described for acute BVDV infections either with cp or ncp BVDV (Liebler-Tenorio et al., 1997) (Figure 7.5).
After intranasal inoculation, primary infection of the tonsillar epithelium and virus replication in the tonsil is observed. The cp BVDV then spreads from the tonsils to the regional lymph nodes. With little delay, cp BVDV can also be detected in Peyer’s patches, mucosa-associated lymphoid follicles in the large intestine, and lymphoid follicles of the lung and nasal mucosa. In mucosa-associated lymphoid tissues, the cp viral antigen is primarily found in the lymphoid tissue and only later in the epithelium. This pattern differs from most enteric pathogens. It suggests a hematogeneous spread of cp BVDV to the gastrointestinal and respiratory tract mucosa. cp BVDV spreads to a lesser degree to the peripheral lymph nodes, thymus, and spleen.
Infection of intestinal mucosa occurs initially in close association with mucosa-associated lymphoid tissues by local expansion of the infection. In the progressed phase of MD, cp BVDV is found multifocally and finally diffusely in the intestinal mucosa. The multifocal infection is most likely caused by multifocal immigration of infected lymphocytes into the mucosa. A large number of these cells might be recirculating from Peyer’s patches, since homing is a known phenomenon of the Peyer’s patch lymphocytes. The preferential infection of the intestinal mucosa versus other mucosal surface is most likely influenced by the large amount of mucosa-associated lymphoid tissue in the intestine and the high numbers of recirculating lymphocytes. When cp BVDV is present multifocally or diffusely in the intestinal mucosa, the first signs of diarrhea are observed.
Lesions in lymphoid tissues
•Depleted lymphoid follicles of tonsils, lymph nodes, spleen, mucosa-associated lymphoid
In the early phase of MD the increased frequency of cell death by apoptosis and the changes in prolifer-
134 |
BVDV: Diagnosis, Management, and Control |
Figure 7.5. Spread of cp BVDV in mucosal disease.
ation are key players for the development of tissue lesions both in lymphoid tissues and epithelium. In tonsils, lymph nodes and Peyer´s patches, cp BVDV is predominantly present in the lymphoid follicles. Initially it is associated with follicular dendritic cells (FDCs) and B-lymphocytes. There is a marked reduction in the number of proliferating B-lymphocytes and a marked increase in apoptotic cells. The immature B-lymphocytes in the dark zone of the germinal center, which have a very high proliferative activity, are especially affected (Liebler-Tenorio and Pohlenz, 1997). Transmission electron microscopy reveals a loss of B-lympho- cytes, whereas FDCs often do not have signs of degeneration (Teichmann et al., 2000). If the loss of B-lymphocytes is very abrupt, death of FDCs is observed leaving empty follicular centers. Histologically this presents as cyst formation, a frequent finding in mucosa-associated lymphoid tissues of the intestine, which has led to the descriptive diagnosis of “colitis cystica.” If depletion of lymphocytes develops more slowly, follicles consisting exclusively of FDCs are found.
Cp BVDV was also detected in dendritic cells (DCs) in the interfollicular areas. DCs can be infected with virus and may serve as carriers for viral proteins (Sprecher and Becker, 1993). Since DCs are able to present antigen very effectively to
T-lymphocytes, they may induce a cell-mediated immune response. Further investigation of T-cell subsets in the early phase of MD revealed accumulations of CD4+ T-lymphocytes particularly at sites where lesions develop and it was speculated about possible cytotoxic effects mediated by CD4+ T-lymphocytes (Frink et al., 2002).
Lesions in mucosa-associated lymphoid tissues
Besides changes within the lymphoid follicles, the loss of B-cells also alters the morphology of domes (Liebler et al., 1995). The loss of domes, and consequently of the follicle-associated epithelium, impedes the selective uptake of antigens from the intestinal lumen. Thus MD causes severe impairment of the inductive part of the intestinal mucosal immune system.
The changes in the gut-associated lymphoid tissue cause not only local tissue destruction, but also affect the whole intestinal mucosa (Liebler et al., 1996). In the mucosa of the small and large intestines of animals moribund with MD, the number of IgAand IgM-positive plasma cells in the lamina propria is severely reduced. This is most likely due to the depletion of mucosa-associated lymphoid follicles, which are the sites where precursors of the plasma cells in the lamina propria are generated. The reduced number of plasma cells causes reduced

Pathogenesis |
135 |
production of secretory immunoglobulins essential for the protection of the mucosal surface.
The number of intraepithelial lymphocytes is also reduced (Liebler et al., 1996). Since they are also derived from Peyer´s patches in part, their reduced numbers might reflect decreased production. On the other hand, the altered microenvironment of the epithelium might also influence their presence between the epithelial cells. In conclusion, all parts of the mucosal immune system are severely altered in MD—e.g., FAE, domes, lymphoid follicles, plasma cells in the lamina propria, and T-lymphocytes in the epithelium.
Lesions in mucosa
The intestinal mucosa becomes infected later than the Peyer´s patches and mucosa-associated lymphoid follicles in the large intestine (Liebler-Tenorio et al., 1997). Enterocytes are not infected from the luminal surface as in most enteric viral infections, but from the basolateral surface. Crypt epithelium is particularly affected. Initially there is a coincidence of three phenomena in the crypts; focal infection of a few epithelial cells with cp BVDV, increased number of apoptotic epithelial cells, and increase of epithelial proliferation rate (Liebler-Tenorio and Pohlenz, 1997). The induction of apoptosis in the crypt epithelium appears to play a key role in the destruction of the intestinal mucosa. In affected crypts, the proliferation rate is severely reduced, and there is a reactive increase of proliferating cells in adjacent crypts.
A common finding in moribund animals in the late phase of MD is the multifocal to diffuse infection of the intestinal mucosa with cp BVDV. In the mucosa of these animals, an increasing number of crypts is without proliferating epithelial cells. As soon as the continuous cellular loss of aged cells from the villous tips is not compensated by crypt proliferation, mucosal atrophy and ulceration develop. At this stage of infection, clinical symptoms of diarrhea become apparent. The progressive lesions explain the lack of success in treating apparently sick animals.
CONCLUSIONS
Many pieces of the complex puzzle of BVDV pathogenesis have been put in place over the last decades, but important pieces are still missing. Observations from experimental infections and naturally occuring disease continue to create doubt about some of the concepts of pathogenesis. Currently, advances are being made in elucidating the interaction of BVDV
infection with different components of the specific immune responses (Beer et al., 1997; Rhodes et al., 1999; Collen and Morrison, 2000; Collen et al., 2002), as well as with cells and mediators of the innate immune system (Adler et al., 1994, 1996; Baigent et al., 2002; Ganheim et al., 2003; Peterhans et al., 2003). Another approach focuses on the interaction of BVDV with its target cells—e.g., how BVDV replication influences gene expression in affected cells (Neill and Ridpath, 2003), in which compartments of the cell BVDV proteins are expressed (Grummer et al., 2001); which receptors are used by BVDV to interact with cells (Minocha et al., 1997; Schelp et al., 2000); and how cp strains are able to exert their cp effect on certain, but not all, cells (Perler et al., 2000; Schweizer and Peterhans, 1999, 2001; Grummer et al., 2002; Bendfeldt et al., 2003). Hopefully some of the answers will yield further insight into the pathogenesis of BVDV and allow better control of this economically important disease of ruminants.
REFERENCES
Adler H, Frech B, Meier P, et al.: 1994, Noncytopathic strains of bovine viral diarrhea virus prime bovine bone marrow-derived macrophages for enhanced generation of nitric oxide. Biochem Biophys Res Commun 202:1562–1568.
Adler H, Jungi TW, Pfister H, et al.: 1996, Cytokine regulation by virus infection: Bovine viral diarrhea virus, a flavivirus, downregulates production of tumor necrosis factor alpha in macrophages in vitro. J Virol 70:2650–2653.
Archambault D, Beliveau C, Couture Y, Carman S: 2000, Clinical response and immunomodulation following experimental challenge of calves with type 2 noncytopathogenic bovine viral diarrhea virus. Vet Res 31:215–227.
Archbald LF, Fulton RW, Seger CL, et al.: 1979, Effect of the bovine viral diarrhea (BVD) virus on preimplantation embryos. Theriogenology 11:81–89.
Baigent SJ, Zhang G, Fray MD, et al.: 2002, Inhibition of beta interferon transcription by noncytopathogenic bovine viral diarrhea virus is through an interferon regulatory factor 3-dependent mechanism. J Virol 76:8979–8988.
Bak A, Callesen H, Meyling A, Greve T: 1992, Calves born after embryo transfer from donors persistently infected with BVD virus. Vet Rec 131:37.
Baker JC: 1995, The clinical manifestations of bovine viral diarrhea virus infection. Vet Clin North Am Food Anim Pract 11:425–445.
Barber DM, Nettleton PF, Herring JA: 1985, Disease in a dairy herd associated with the introduction and

136 |
BVDV: Diagnosis, Management, and Control |
spread of bovine virus diarrhea virus. Vet Rec 117:459–464.
Barkema HW, Bartels CJ, van Wuijckhuise L, et al.: 2001, Outbreak of bovine virus diarrhea on Dutch dairy farms induced by a bovine herpesvirus 1 marker vaccine contaminated with bovine virus diarrhea virus type 2. Tijdschr Diergeneeskd
126:158–165.
Baszler TV, Evermann JF, Kaylor PS, et al.: 1995, Diagnosis of naturally occurring bovine viral diarrhea virus infections in ruminants using monoclonal antibody-based immunohistochemistry. Vet Pathol 32:609–618.
Beer M, Wolf G, Pichler J, et al.: 1997, Cytotoxic T- lymphocyte responses in cattle infected with bovine viral diarrhea virus. Vet Microbiol 58:9–22.
Bendfeldt S, Grummer B, Greiser-Wilke I: 2003, No caspase activation but overexpression of Bcl-2 in bovine cells infected with noncytopathic bovine virus diarrhea virus. Vet Microbiol 96:313–326. Bielanski A, Hare WC: 1988, Effect in vitro of bovine viral diarrhea virus on bovine embryos with the
zona pellucida intact, damaged and removed. Vet Res Commun 12:19–24.
Bielanski A, Sapp T, Lutze-Wallace C: 1998, Association of bovine embryos produced by in vitro fertilization with a noncytopathic strain of bovine viral diarrhea virus type II. Theriogenology 49:1231–1238.
Bielefeldt Ohmann H: 1983, Pathogenesis of bovine viral diarrhea-mucosal disease: Distribution and significance of BVDV antigen in diseased calves.
Res Vet Sci 34:5–10.
Bielefeldt Ohmann H: 1984, An oculo-cerebellar syndrome caused by congenital bovine viral diarrhea virus-infection. Acta Vet Scand 25:36–49.
Bielefeldt Ohmann H: 1988, BVD virus antigens in tissues of persistently viremic, clinically normal cattle: Implications for the pathogenesis of clinically fatal disease. Acta Vet Scand 29:77–84.
Binkhorst GJ, Journee DL, Wouda W, Straver PJ, Vos JH: 1983, Neurological disorders, virus persistence and hypomyelination in calves due to intra-uterine infections with bovine virus diarrhea virus. I.
Clinical symptoms and morphological lesions. Vet Q 5:145–155.
Bolin SR, McClurkin AW, Coria MF: 1985a, Effects of bovine viral diarrhea virus on the percentages and absolute numbers of circulating B and T lymphocytes in cattle. Am J Vet Res 46:884–886.
Bolin SR, McClurkin AW, Cutlip RC, Coria MF: 1985b, Severe clinical disease induced in cattle persistently infected with noncytopathic bovine viral diarrhea virus by superinfection with cytopathic bovine viral diarrhea virus. Am J Vet Res 46:573–576.
Bolin SR, Ridpath JF: 1992, Differences in virulence between two noncytopathogenic bovine viral diarrhea viruses in calves. Am J Vet Res 53:2175–2163.
Bolin SR, Ridpath JF: 1998, Prevalence of bovine viral diarrhea virus genotypes and antibodies against these genotypes in fetal bovine serum. J Vet Diagn Invest 10:135–139.
Brock KV, Chase CC: 2000, Development of a fetal challenge method for the evaluation of bovine viral diarrhea virus vaccines. Vet Microbiol 77:209–214.
Brock KV, Grooms DL, Ridpath J, Bolin SR: 1998, Changes in levels of viremia in cattle persistently infected with bovine viral diarrhea virus. J Vet Diagn Invest 10:22–26.
Brodersen BW, Kelling CL: 1998, Effect of concurrent experimentally induced bovine respiratory syncytial virus and bovine viral diarrhea virus infection on respiratory tract and enteric diseases in calves. Am J Vet Res 59:1423–1430.
Brodersen BW, Kelling CL: 1999, Alteration of leukocyte populations in calves concurrently infected with bovine respiratory syncytial virus and bovine viral diarrhea virus. Viral Immunol 12:323–334.
Brown TT, Bistner SI, de Lahunta A, et al.: 1975, Pathogenetic studies of infection of the bovine fetus with bovine viral diarrhea virus. II. Ocular lesions. Vet Pathol 12:394–404.
Brown TT, de Lahunta A, Bistner SI, et al.: 1974, Pathogenetic studies of infection of the bovine fetus with bovine viral diarrhea virus. I. Cerebellar atrophy. Vet Pathol 11:486–505.
Brown TT, de Lahunta A, Scott FW, et al.: 1973, Virus induced congenital anomalies of the bovine fetus. II. Histopathology of cerebellar degeneration (hypoplasia) induced by the virus of bovine viral diarrhea-mucosal disease. Cornell Vet 63:561–578.
Brownlie J, Clarke MC: 1993, Experimental and spontaneous mucosal disease of cattle: A validation of Koch’s postulates in the definition of pathogenesis. Intervirology 35:51–59.
Brownlie J, Clarke MC, Howard CJ: 1984, Experimental production of fatal mucosal disease in cattle. Vet Rec 114:535–536.
Brownlie J, Clarke MC, Howard CJ: 1989, Experimental infection of cattle in early pregnancy with a cytopathic strain of bovine virus diarrhea virus. Res Vet Sci 46:307–311.
Bruschke CJ, Haghparast A, Hoek A, et al.: 1998b, The immune response of cattle, persistently infected with noncytopathic BVDV, after superinfection with antigenically semi-homologous cytopathic BVDV. Vet Immunol Immunopathol
62:37–50.
Bruschke CJ, Weerdmeester K, Van Oirschot JT, Van Rijn PA: 1998a, Distribution of bovine virus diar-

Pathogenesis |
137 |
rhea virus in tissues and white blood cells of cattle during acute infection. Vet Microbiol 64:23–32.
Buckner R: 1997, Diabetes mellitus in a cow persistently infected with bovine viral diarrhea virus.
Tierarztl Prax 25:351–352.
Carman S, van Dreumel T, Ridpath J, et al.: 1998, Severe acute bovine viral diarrhea in Ontario, 1993–1995. J Vet Diagn Invest 10:27–35.
Casaro APE, Kendrick JW, Kennedy PC: 1971, Response of the bovine fetus to bovine viral diarrheamucosal disease virus. Am J Vet Res 32:1543–1562.
Castrucci G, Osburn BI, Ferrari M, Traldi V: 1992, An experimental contribution to the study of the pathogenesis of bovine viral diarrhea virus infection.
Comp Immunol Microbiol Infect Dis 15:163–169. Charleston B, Brackenbury LS, Carr BV, et al.: 2002, Alpha/beta and gamma interferons are induced by infection with noncytopathic bovine viral diarrhea
virus in vivo. J Virol 76:923–927. Charleston B, Fray MD, Baigent S, Carr BV,
Morrison WI: 2001, Establishment of persistent infection with non-cytopathic bovine viral diarrhea virus in cattle is associated with a failure to induce type I interferon. J Gen Virol 82:1893–1897.
Collen T, Douglas AJ, Paton DJ, et al.: 2000, Single amino acid differences are sufficient for CD4(+) T-cell recognition of a heterologous virus by cattle persistently infected with bovine viral diarrhea virus. Virology 276:70–82.
Collen T, Morrison WI: 2000, CD4(+) T-cell responses to bovine viral diarrhea virus in cattle. Virus Res 67:67–80.
Collen T, Carr V, Parsons K, et al.: 2002, Analysis of the repertoire of cattle CD4(+) T cells reactive with bovine viral diarrhea virus. Vet Immunol Immunopathol 87:235–238.
Corapi WV, Elliott RD, French TW, et al.: 1990, Thrombocytopenia and hemorrhages in veal calves infected with bovine viral diarrhea virus. J Am Vet Med Assoc 196:590–596.
Coria MF, McClurkin AW: 1978, Specific immune tolerance in an apparently healthy bull persistently infected with bovine viral diarrhea virus. J Am Vet Med Assoc 172:449–451.
David GP, Crawshaw TR, Gunning RF, et al.: 1994, Severe disease in adult dairy cattle in three UK dairy herds associated with BVD virus infection. Vet Rec 134:468–472.
Done JT, Terlecki S, Richardson C, et al.: 1980, Bovine virus diarrhea-mucosal disease virus: Pathogenicity for the fetal calf following maternal infection. Vet Rec 106:473–479.
Drew TW, Sandvik T, Wakeley P, et al.: 2002, BVD virus genotype 2 detected in British cattle. Vet Rec 151:551.
Dubovi EJ: 1994, Impact of bovine viral diarrhea virus on reproductive performance in cattle. Vet Clin North Am Food Anim Pract 10:503–514
Duffell SJ, Harkness JW: 1985, Bovine virus diar- rhea-mucosal disease infection in cattle. Vet Rec 117:240–245.
Duffell SJ, Sharp MW, Winkler CE, et al.: 1984, Bovine virus diarrhea-mucosal disease virusinduced fetopathy in cattle: Efficacy of prophylactic maternal pre-exposure. Vet Rec 114:558–561.
Ellis JA, West KH, Cortese VS, et al.: 1998, Lesions and distribution of viral antigen following an experimetal infection of young seronegative calves with virulent bovine virus diarrhea virus-type II. Can J Vet Res 62:161–169.
Elvander M, Baule C, Persson M, et al.: 1998, An experimental study of a concurrent primary infection with bovine respiratory syncytial virus (BRSV) and bovine viral diarrhea virus (BVDV) in calves. Acta Vet Scand 39:251–264.
Fray MD, Mann GE, Bleach EC, et al.: 2002, Modulation of sex hormone secretion in cows by acute infection with bovine viral diarrhea virus.
Reproduction 123:281–289.
Fray MD, Mann GE, Clarke MC, Charleston B: 2000a, Bovine viral diarrhea virus: Its effects on ovarian function in the cow. Vet Microbiol 77:185–194.
Fray MD, Prentice H, Clarke MC, Charleston B: 1998, Immunohistochemical evidence for the localization of bovine viral diarrhea virus, a singlestranded RNA virus, in ovarian oocytes in the cow. Vet Pathol 35:253–259.
Fray MD, Supple EA, Morrison WI, Charleston B: 2000b, Germinal centre localization of bovine viral diarrhea virus in persistently infected animals. J Gen Virol 81:1669–1673.
Fredriksen B, Press CM, Loken T, Odegaard SA: 1999b, Distribution of viral antigen in uterus, placenta and foetus of cattle persistently infected with bovine virus diarrhea virus. Vet Microbiol 64:109–22.
Fredriksen B, Press CM, Sandvik T, et al.: 1999a, Detection of viral antigen in placenta and fetus of cattle acutely infected with bovine viral diarrhea virus. Vet Pathol 36:267–275.
Fricke J, Gunn M, Meyers G: 2001, A family of closely related bovine viral diarrhea virus recombinants identified in an animal suffering from mucosal disease: New insights into the development of a lethal disease in cattle. Virology 291:77–90.
Frink S, Grummer B, Pohlenz JF, Liebler-Tenorio EM: 2002, Changes in distribution and numbers of CD4+ and CD8+ T-lymphocytes in lymphoid tissues and intestinal mucosa in the early phase of ex-