Материал: Bovine Viral Diarrhea Virus Diagnosis, Management, and Control
Clinical Features |
107 |
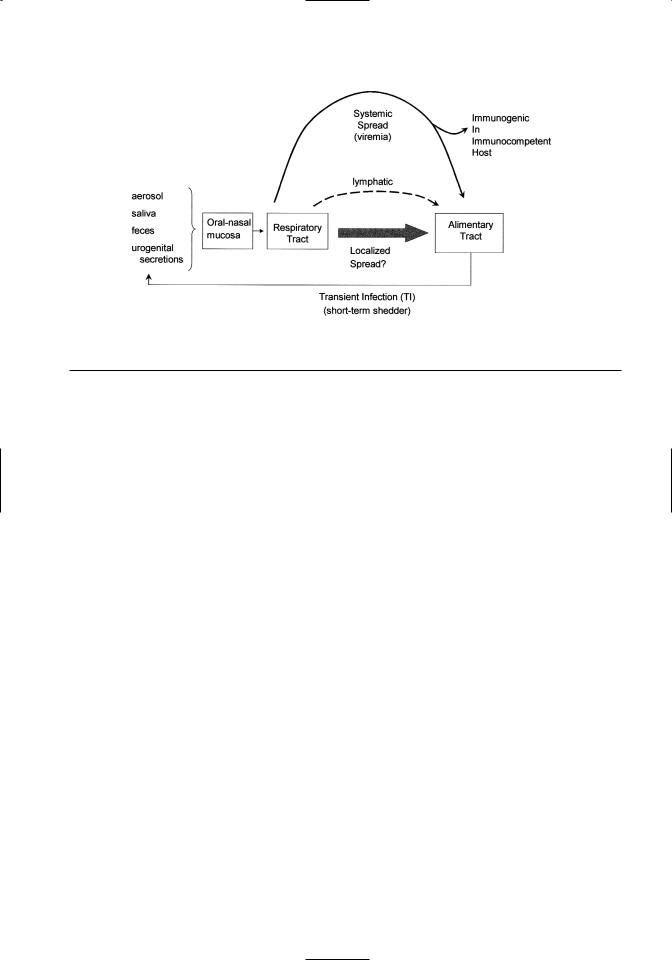
Figure 6.2. Schematic depicting the spread of BVDV in secretions and excretions during a transient infection (TI). Three modes of virus dispersal within the animal’s body are noted. These include lymphatics, viremia, and localized spread via the alimentary tract.
discharge at 12 dpi (Wilhelmsen et al., 1990). Although clinical signs observed with the BVDV strain used were minimal, there were significant, immunosuppressive effects of BVDV infection. A 25% or greater decrease in leukocyte count was observed that lasted up to 12 days. In 6-month-old calves there was more evidence of lymphoid involvement of the gastrointestinal tract than in the 2-month-old colostrum-deprived calves. There was gross swelling and edema of mesenteric lymph nodes and lymphocytolysis and edema in the gut-associated lymphoid tissue. This makes it clearer that the following comments are a continuation of the proceeding discussion. Differences noted between BVDV infection of 2-month-old and 6-month-old calves may have resulted from age differences, BVDV strain variation, or a combination of both (Pellerin et al., 1994). Nonetheless, it is apparent that BVDV is capable of multiple systemic effects resulting in different clinical manifestations. Grooms et al. (2002), appropriately wrote about the “diseases” caused by BVDV and indicated that there were at least six initial forms of BVDV infection in nonpregnant cattle and at least two in pregnant cattle (Table 6.1).
BVDV IN PREGNANT CATTLE
The effects of BVDV on pregnant cattle are listed in Table 6.1. Early embryonic death and abortion may be seen following BVDV infection of naive heifers and cows (Bolin, 1990b). Usually up to 85% of cattle become infected, undergo a transient viremia, and seroconvert prior to breeding age. However, this
leaves approximately 15% of the population at risk at the time of initial breeding. It is estimated that BVDV causes about 6–10% of infectious abortions in cattle (Figure 6.3; Dubovi, 1994). Although this infection may result in serious reproductive losses in a few herds, the predominant form of infection with BVDV is the congenital infection (Muñoz-Zanzi et al., 2003). The fetus is highly susceptible between 45 and 125 days of gestation (Dubovi, 1994). Calves infected during this time are at high risk of developing fetal abnormalities that may affect the brain (cerebellar hypoplasia), eyes (retinal atrophy, cataracts), growth retardation with arrested bone development, and pulmonary hypoplasia. Another form of congenital defect is immune tolerance, in which the developing fetus becomes infected with a noncytopathic strain (see Chapter 3) of BVDV between 90 and 120 days (Figure 6.4). The immune system develops tolerance to the infecting strain of BVDV. The result is that no immune response to the virus is mounted by the fetal immune system and the fetus becomes persistently viremic or persistently infected (PI). Figure 6.1 depicts the clinical manifestations of BVDV, with particular attention on the propagation of BVDV PI offspring (see Chapter 7). Following birth, the PI calf is vulnerable to developing mucosal disease (MD) by one of several mechanisms (Figure 6.5).
The impact of different BVDV strains on reproductive performance, in particular age predilection of the fetus for infection with different BVDV genotypes and subgenotypes, was recently reported

Table 6.1. Subclinical and clinical manifestations of bovine viral diarrhea virus (BVDV)*
Category |
Manifestations |
|
|
Acute—Nonpregnant |
|
|
|
Acute/asymptomatic |
Mild fever, leukopenia, seroconversion to BVDV, short-term viremia, |
|
short-term shedding. |
Acute/symptomatic |
Fever, leukopenia, depression, anorexia ocular-nasal discharge, oral le- |
|
sions, diarrhea, decreased milk production in lactating cattle, short- |
|
term viremia (15 days), short-term shedding. |
Severe acute |
Peracute course, fever, pneumonia, sudden death (10–25% mortality). |
|
Associated with BVDV type 2. |
Hemorrhagic |
Marked thrombocytopenia, bloody diarrhea, epistaxis, hyphema, bleeding |
|
from injection and branding sites, pyrexia, leukopenia, and death. As- |
|
sociated with BVDV type 2. |
Acute/bovine respiratory distress |
Fever, pneumonia, anorexia, prolonged treatment, and leading cause of |
|
death in feedlot cattle. Secondary infections with Pasteurella multo- |
|
cida, Mannheimia hemolytica, and Mycoplasma bovis complicate the |
|
pathogenesis. |
Acute/immunosuppressive |
Leukocyte function diminished up to 25%, leukopenia, decreased CD+4 |
|
and CD+8 T lymphocytes, and decreased macrophage and neutrophil |
|
functions. Secondary or polymicrobial infections with bovine respira- |
|
tory syncytial virus, papular stomatitis virus, malignant catarrhal fever |
|
virus have been reported. |
|
|
Acute—Pregnant |
|
|
|
Abortion |
Naive cattle with no prior exposure to BVDV become infected as in acute/ |
|
asymptomatic or acute/symptomatic above. Viremia leads to placental |
|
infection. Abortion, early embryonic death may occur from placentitis. |
|
If infection of dam occurs during 90–150 days gestation, congenital in- |
|
fection (CI) of fetus occurs. One form of CI between 90–120 days may |
|
result in fetal tolerance to BVDV. |
Congenital defects |
Infection of a naive dam between 90 and 150 days gestation may result in |
|
viremia, which infects fetus resulting in congenital infection (CI). One |
|
particular CI occurs between day 90 and 125 and may result in fetal |
|
tolerance to BVDV. Calves born with this form of CI are referred to as |
|
persistently infected (PI). Calves that are PI have long-term (lifelong) |
|
viremia and are long-term shedders of virus, and are at high risk to de- |
|
velop mucosal disease (MD) |
|
|
Acute/Chronic Sequel to CI |
|
|
|
Mucosal disease |
Calves (6–18 months) that are PI are immunotolerant to noncytopatho- |
|
genic (ncp) BVDV (see Ridpath, Chapter 3). These PI calves may ap- |
|
pear clinically normal, but upon superinfection with a homologous |
|
BVDV, or mutation of the ncp virus to a cytopathic (cp) variant, or vac- |
|
cination with a MLV BVDV strain homologous to the ncp PI virus, |
|
have a high percentage of developing one of two reported forms of |
|
MD—acute or chronic. |
108

Clinical Features |
109 |
Category |
Manifestations |
Acute/Chronic Sequel to CI
Mucosal disease (continued)
Acute MD is characterized by onset of clinical symptoms within 10–14 days postinfection. Symptoms include biphasic fever, anorexia, tachycardia, polypnea, decreased milk production, and watery diarrhea. Other clinical signs may include nasal-ocular discharge, corneal opacity, excessive salivation, decreased rumination, and bloat. Cattle with acute MD become progressively dehydrated and usually die within 3–10 days. Although the mortality usually approaches 100%, a few animals may survive the acute MD, but are prone to develop chronic MD Chronic MD is a sequel of acute MD. Affected cattle are unthrifty, and may have intermittent diarrhea, chronic bloat, decreased appetite, and weight loss. Nasal-ocular discharge is commonly reported. Cattle with chronic MD rarely survive past 18 months and are usually culled due to low performance or die of severe debilitation.
*Modified from Grooms et al., (2002).
(Evermann and Ridpath, 2002). The results of this study (Table 6.2) indicated that later fetal infections leading to congenital defects and PI calves were associated more with BVDV types 1a and 1b than with BVDV type 2.
RECOGNITION OF CLINICAL SIGNS
SUBCLINICAL INFECTION
It has been estimated that 70–90% of BVDV infections in immunocompetent, seronegative cattle occur without manifestation of clinical signs (Ames, 1986). In closely monitored animals, infection typically results in mild fever, leukopenia, and the development of serum-neutralizing antibodies. In dairy cattle, a decrease in milk production has been associated with subclinical infections (Moerman et al., 1994). Viral replication appears to occur in the upper
respiratory tract and adjacent lymphoid tissues (Bolin, 1990a; Meehan et al., 1998). Subclinical infections appear to account for high levels of serumneutralizing antibody titers found in most unvaccinated cattle. However, it should be noted that subclinical disease in the pregnant dam might not reflect the severity of the effects of viral infection to the exposed fetus.
Figure 6.3. Schematic depicting the origin of persistently infected (PI) calves following viremia and placental infection.
Figure 6.4. Schematic depicting the status of samples from persistently infected calves and the occurrence of virus in plasma/white blood cells (buffy coat) and feces from affected animals.

110 |
BVDV: Diagnosis, Management, and Control |
Table 6.2. Summary of bovine viral diarrhea virus isolates and analysis of genotype compared with clinical presentation
BVDV Genotype*
and Subgenotypes
Clinical Presentation |
1a |
1b |
2 |
Figure 6.5. Schematic depicting the outcome of a persistently infected calf, which is vulnerable to mucosal disease upon reinfection with homologous BVDV, mutation of tolerizing noncytopathogenic BVDV to cytopathogenic, or vaccination with a homologous strain of BVDV.
CLINICAL BVDV INFECTION
Acute BVDV infection is the term used to describe clinical disease that occurs in non-persistently infected, immunocompetent cattle. The disease has typically been described in seronegative cattle from 6–24 months of age. Conceivably, the disease could occur in seropositive cattle that are infected with a viral strain that is heterologous to the one that has caused seroconversion. Clinical signs include varying levels of fever, anorexia, lethargy, leukopenia, ocular and nasal discharge, oral erosions and ulcers, oral papilla blunting and hemorrhage, diarrhea, and decreased milk production in lactating cows. Epithelial erosions may be present in the interdigital space, coronet, teats, or vulva. Tachypnea may be incorrectly correlated with pneumonia, but is most likely due to fever or other nonpulmonary factors.
Acute BVDV infection of neonates may result in signs of enteritis or pneumonia. Such signs are thought to be most commonly associated with calves suffering from failure of passive transfer, since passively acquired maternal antibodies are thought to be protective. Clinical disease in the face of adequate passive transfer may be related to antigenic diversity between infecting viral strains and viral strains against which passive immunity was developed. Lastly, inadequate passive immunity combined with immunosuppressive effects of BVDV infection may result in secondary diseases affecting various organ systems.
Acute BVDV infection results in damage to ep-
Early fetal infection (abortion) |
14% |
22% |
45% |
Later fetal infection (includes |
86% |
78% |
55% |
animals that are persistently infected, weak calves, chronic poor doers, and mucosal disease)
*From Evermann and Ridpath, 2002.
ithelial surfaces of the gastrointestinal, integumentary, and respiratory systems (Blowey and Weaver, 2003). Viral antigen has been identified in epithelial surfaces of the tongue, esophagus, intestine, bronchi, and skin, as well as phagocytic cells in lymph nodes, thymus, Peyer’s patches, tonsils, and spleen (Ohmann, 1983). Tonsils and respiratory tract tissues are infected first and from there dissemination occurs to other epithelial surfaces and lymphoid tissues. Virus is retained within mononuclear phagocytic cells of the lymphoid tissue (Ohmann, 1983).
Differential diagnoses for diarrheal diseases of adult cattle include Salmonellosis, winter dysentery, Johne’s disease, gastrointestinal parasites, malignant catarrhal fever, arsenic poisoning, Mycotoxicosis, and copper deficiency (Blowey and Weaver, 2003; Kahrs, 2001). Differential diagnoses for diseases causing oral lesions in cattle include malignant catarrhal fever, blue tongue, vesicular stomatitis, papular stomatitis, foot and mouth disease, and Rinderpest. Differential diagnoses for diseases causing diarrhea in neonatal calves include rotaand coronavirus infections, cryptosporidiosis, E. coli infection, Salmonellosis and coccidiosis. Differential diagnoses for respiratory diseases of calves include infection with bovine respiratory syncytial virus (BRSV), Pasteurella spp., Mannheimia spp., Hemophilus spp., Salmonella spp., and Mycoplasma spp.
SEVERE ACUTE BVDV INFECTION
In the early 1990s, an atypical and significantly more severe form of BVDV infection was recognized in the United States and Canadian cattle populations (Corapi et al., 1990; Pellerin et al., 1994;
Clinical Features |
111 |
Carman et al., 1998). A peracute disease course was described with high morbidity and mortality in all age groups of cattle. In the Quebec outbreak approximately 25% of veal calves died, and in Ontario, all age groups of cattle suffered fever, pneumonia, and sudden death (Pellerin et al., 1994; Carman et al., 1998). Severity varied between herds, though some herds experienced 10–20% mortality. Abortions were also a common occurrence. The gross lesions found in these animals were severe, resembling those of mucosal disease. The viral isolates from these outbreaks were characterized by nucleotide sequencing and compared to more classical BVDV outbreak strains (Ridpath et al., 1994). The novel group of BVDV was designated BVDV type 2, compared to the classical group of viruses, now known as BVDV type 1.
Importantly, not all BVDV type 2 outbreaks are necessarily associated with severe disease. Therefore, a severe outbreak of disease should not be assumed to have been caused by BVDV type 2. Alternatively, it is likely that some BVDV type 1 isolates may be capable of causing severe disease.
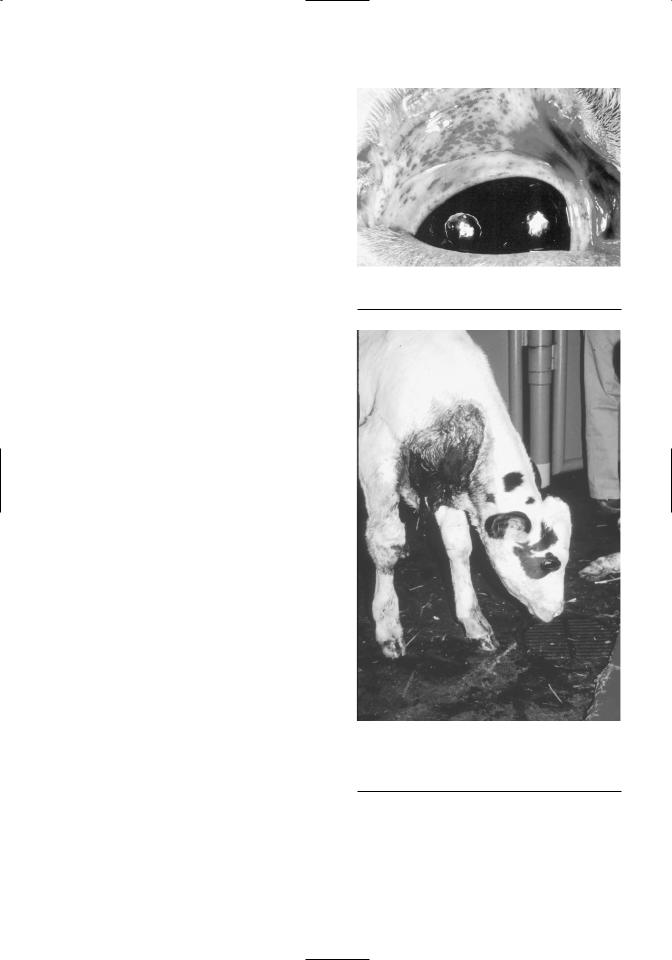
Hemorrhagic syndrome is a form of severe acute BVDV that appears to be associated with a noncytopathic, type 2 strain of BVDV. Affected cattle often suffer marked thrombocytopenia, petechiation, and ecchymoses of mucosal surfaces, epistaxis, bloody diarrhea, bleeding from injection sites or trauma, fever, leukopenia, and death (Corapi et al., 1990) (Figures 6.6 and 6.7). Importantly, during an outbreak variation exists between animals in the expression of clinical signs, and only a minority of animals develop fulminant hemorrhagic syndrome. Others may simply suffer marked lymphopenia and thrombocytopenia and succumb without overt clinical manifestation of hemorrhage. Therefore, during an outbreak of severe acute BVDV, practitioners and producers typically observe a diverse clinical picture. Whereas all cattle with hemorrhagic syndrome suffer from severe acute BVDV, not all cattle with severe acute BVDV necessarily progress to hemorrhagic syndrome. The pathophysiologic mechanism of thrombocytopenia and the hemorrhagic syndrome is not clearly understood. Viral antigen has been shown to be associated with both platelets and megakaryocytes, and in addition to thrombocytopenia, platelet function is altered (Walz et al., 1999). Common differential diagnoses for hemorrhagic syndrome include septicemia with concurrent disseminated intravascular coagulation, bracken fern toxicity, sweet clover poisoning, and anticoagulant toxicity.
Figure 6.6. Petechiation of ocular mucous membranes observed in thrombocytopenic calf.
Figure 6.7. Excessive hemorrhage in a thrombocytopenic calf several hours after being administered a subcutaneous injection (courtesy of Dr. Sheila McGuirk, University of Wisconsin).
MUCOSAL DISEASE
Typically mucosal disease is a sporadic condition wherein <5% of herds are affected. In outbreaks, it is common that multiple animals of similar age are affected because the infection is initiated in the fetus