Page 494 |
DICOM PS3.3 2020a - Information Object Definitions |
Attribute Name |
Tag |
Type |
Attribute Description |
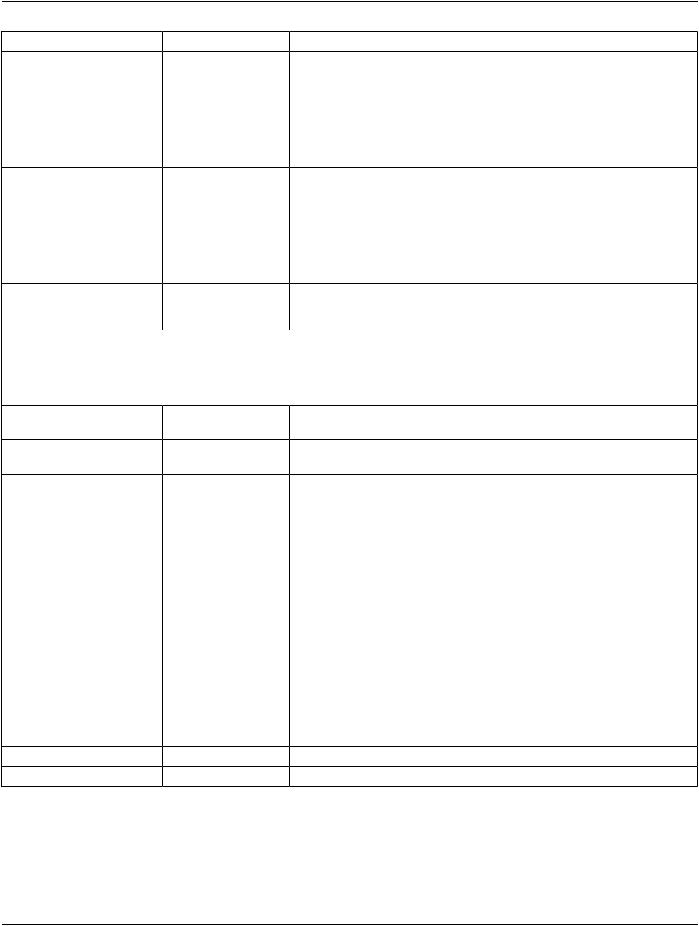
>Consent for Distribution |
(0012,0085) |
1 |
Whether or not consent to distribute has been granted for the purpose |
Flag |
|
|
described in Distribution Type (0012,0084). |
Enumerated Values:
NO
YES
WITHDRAWN
See Section C.7.2.3.1.2.
Note
Undersomecircumstances,consentmaybewithdrawn.Thepurpose ofencodingthisistowarnreceivingsystemsthatfurtherdistribution may not be appropriate, but no semantics are defined by the Standard for what action is appropriate under such circumstances, suchaswhattodowithpreviouslyreceivedimagesthathadavalue of YES.
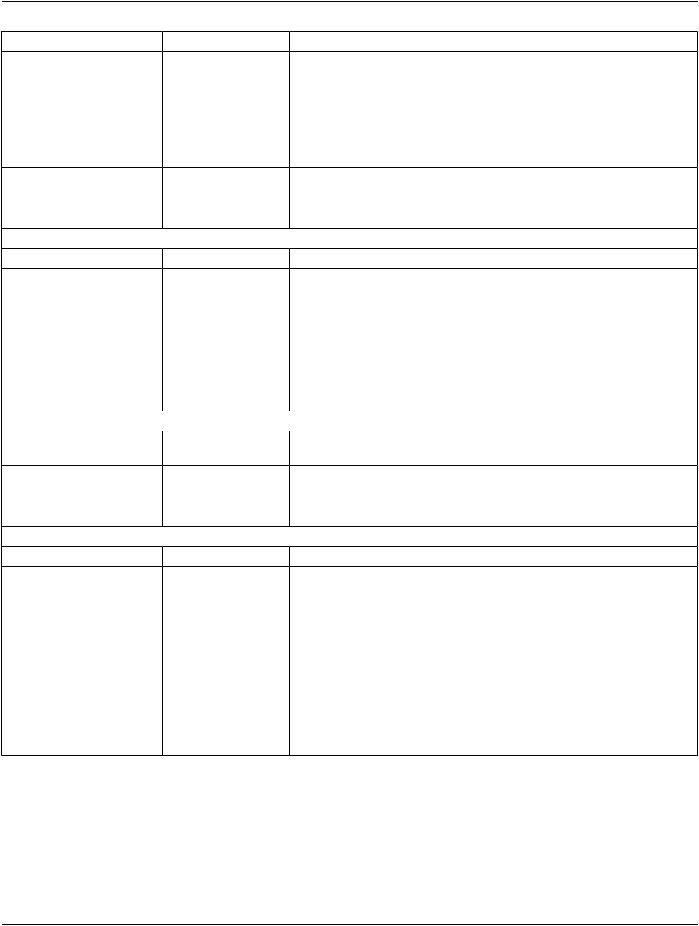
C.7.2.3.1 Clinical Trial Study Attribute Descriptions
C.7.2.3.1.1 Clinical Trial Time Point
The Clinical Trial Time Point ID (0012,0050) Attribute identifies an imaging Study within the context of an investigational protocol. This Attribute is used to define a set of Studies that are grouped together as a clinical time point or data submission in a clinical trial or research. The Clinical Trial Time Point Description (0012,0051) Attribute can be used to give a description of the Clinical Trial Time Point to which the set of Studies belongs.
C.7.2.3.1.2 Consent For Clinical Trial Use Sequence
For applications such as clinical trials or research, the distribution of Composite Instances in Studies, whether containing identifying information or partially or completely de-identified, may need to be controlled. Permission for distribution is usually granted under the control of the Patient (through informed consent), the ethics committee or institutional review board responsible for the Study, and the sponsor of the Study through contractual means. The Consent for Clinical Trial Use Sequence (0012,0083) is intended to encode the result of the consent process to allow appropriate subsequent handling of the instances.
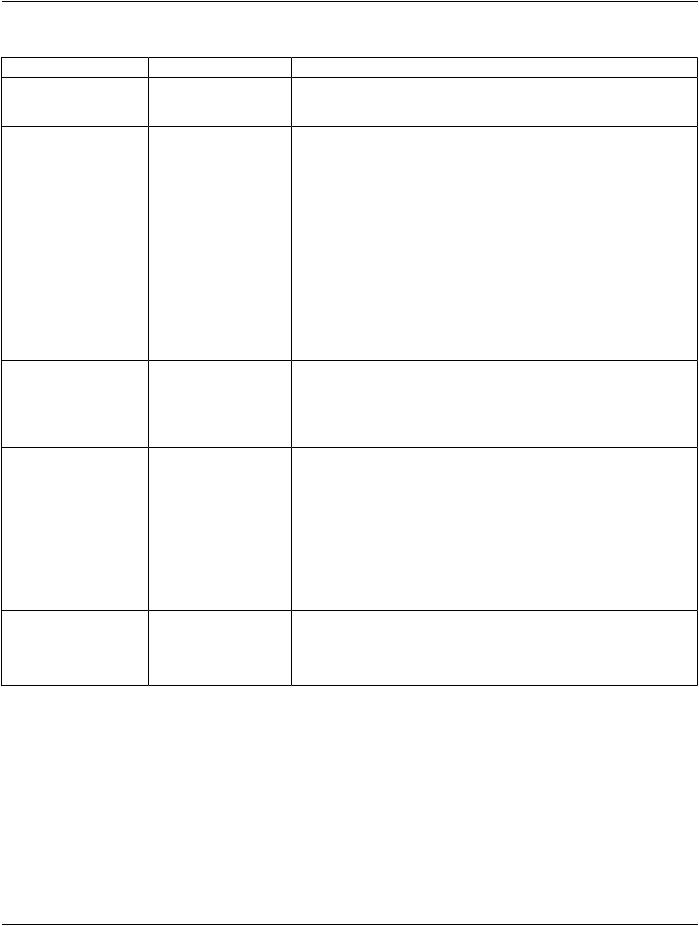
The Defined Terms for Distribution Type (0012,0084) mean that consent has been issued to distribute for the following purposes:
Defined Terms:
NAMED_PROTOCOLconducting the protocol named in Clinical Trial Protocol ID (0012,0020)
RESTRICTED_REUSEre-use for restricted purposes (not specified here) other than those for which the instances were originally created
PUBLIC_RELEASE release to the general public for re-use without restriction
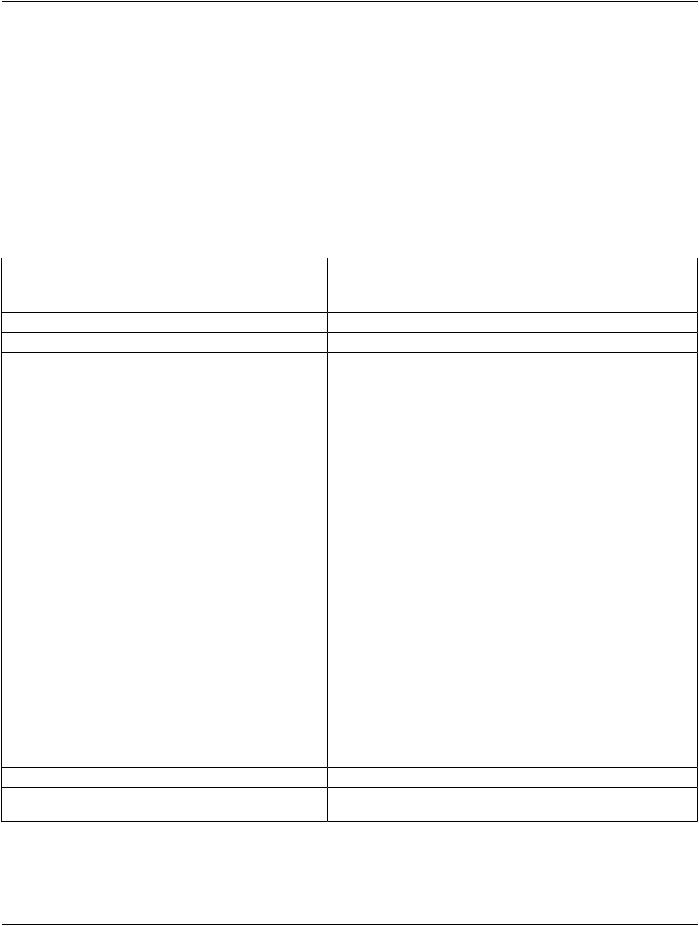
Note
1.There is no intent to convey in this Sequence further details of the often complex consent and approval process. Further information about the protocol and ethics committee may be found in the Clinical Trials Modules, if present.
2.There is no identification of an individual responsible for the approval or granting consent, since in the case of a clinical trial or research subject granting informed consent, the presence of this information would breach de-identification re- quirements.
3.Multiple Sequence Items may be present; for example a Study may be approved for distribution for conducting multiple explicitly named protocols.
4.Whether or not the instances have been adequately de-identified for any particular purpose of distribution is not defined by the Attributes in the Consent for Clinical Trial Use Sequence (0012,0083). Other Attributes address this, such as