Page 376 |
DICOM PS3.3 2020a - Information Object Definitions |
The Acquisition Context information may be entered during acquisition, or obtained from the Modality Worklist using information supplied in the Protocol Context, using TID 15101 “NM/PET Protocol Context”.
A.57 Surface Segmentation IOD
A.57.1 Surface Segmentation IOD Description
The Surface Segmentation Information Object Definition (IOD) specifies a polygonal representation of a three dimensional surface. A Surface Segmentation SOP Instance may reference an externally defined coordinate system via the Frame of Reference UID (0020,0052) or establish its own coordinate system.
The Surface Segmentation IOD does not include the full set of acquisition parameters of the referenced images, e.g., cardiac phase. An application rendering or processing the segmentation may need to access the referenced images for such information.
One Segmented Surface Instance can contain one or more surfaces. Each surface within a Segmented Surface IE is represented as a single object.
A.57.2 Surface Segmentation IOD Entity-Relationship Model
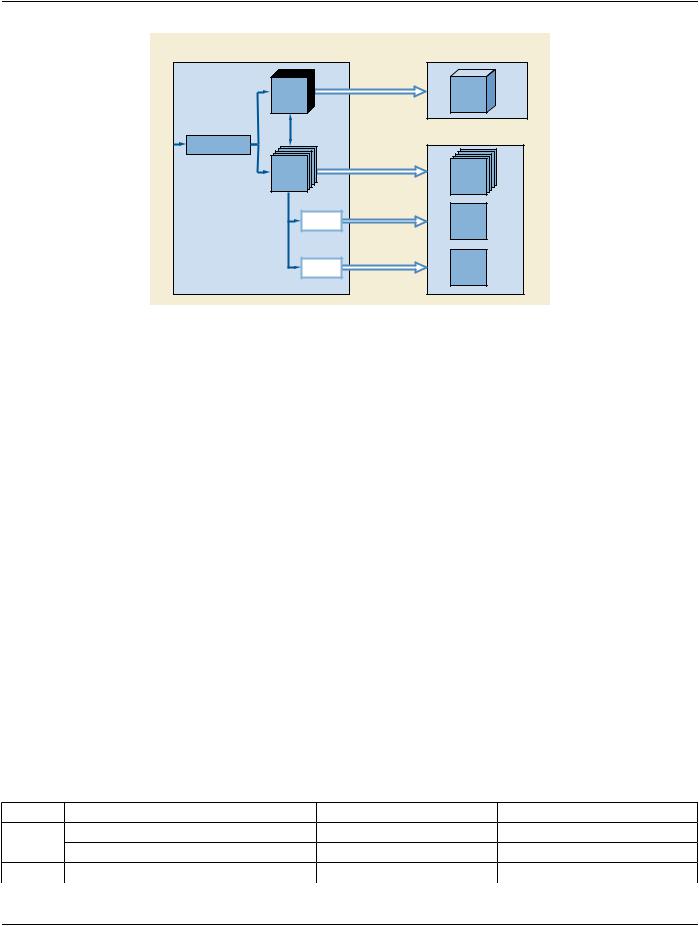
This IOD uses the E-R Model in Section A.1.2, with only the Surface IE below the Series IE.
A.57.3 Surface Segmentation IOD Module Table
Table A.57-1 specifies the Modules of the Surface Segmentation IOD.
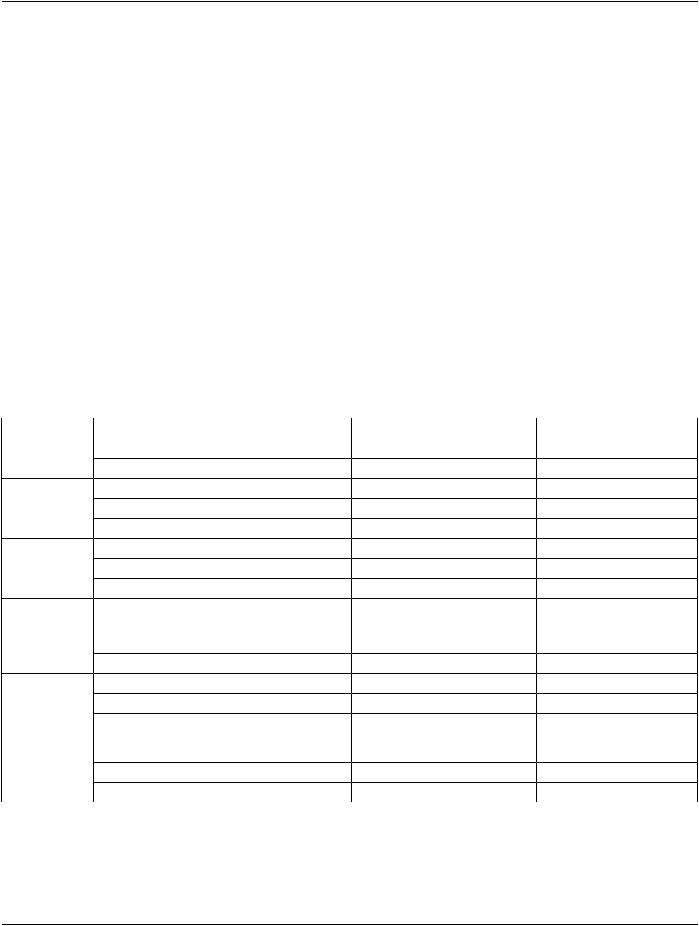
Table A.57-1. Surface Segmentation IOD Modules
IE |
Module |
Reference |
Usage |
Patient |
Patient |
C.7.1.1 |
M |
|
Clinical Trial Subject |
C.7.1.3 |
U |
Study |
General Study |
C.7.2.1 |
M |
|
Patient Study |
C.7.2.2 |
U |
|
Clinical Trial Study |
C.7.2.3 |
U |
Series |
General Series |
C.7.3.1 |
M |
|
Segmentation Series |
C.8.20.1 |
M |
|
Clinical Trial Series |
C.7.3.2 |
U |
Frame of |
Frame of Reference |
C.7.4.1 |
M |
Reference |
|
|
|
Equipment |
General Equipment |
C.7.5.1 |
M |
|
Enhanced General Equipment |
C.7.5.2 |
M |
Surface |
Surface Segmentation |
C.8.23.1 |
M |
|
Surface Mesh |
C.27.1 |
M |
|
Common Instance Reference |
C.12.2 |
C-Requiredifthesurfacehas |
|
|
|
been derived from another |
|
|
|
SOP Instance |
|
General Reference |
C.12.4 |
U |
|
SOP Common |
C.12.1 |
M |
A.57.4 Surface Segmentation IOD Content Constraints
The Defined CID for Purpose of Reference Code Sequence (0040,A170) within Source Instance Sequence (0042,0013) in the Gen- eral Reference Module shall be CID 7019 “Segmentation Non-Image Source Purposes of Reference”.