DICOM PS3.3 2020a - Information Object Definitions |
Page 371 |
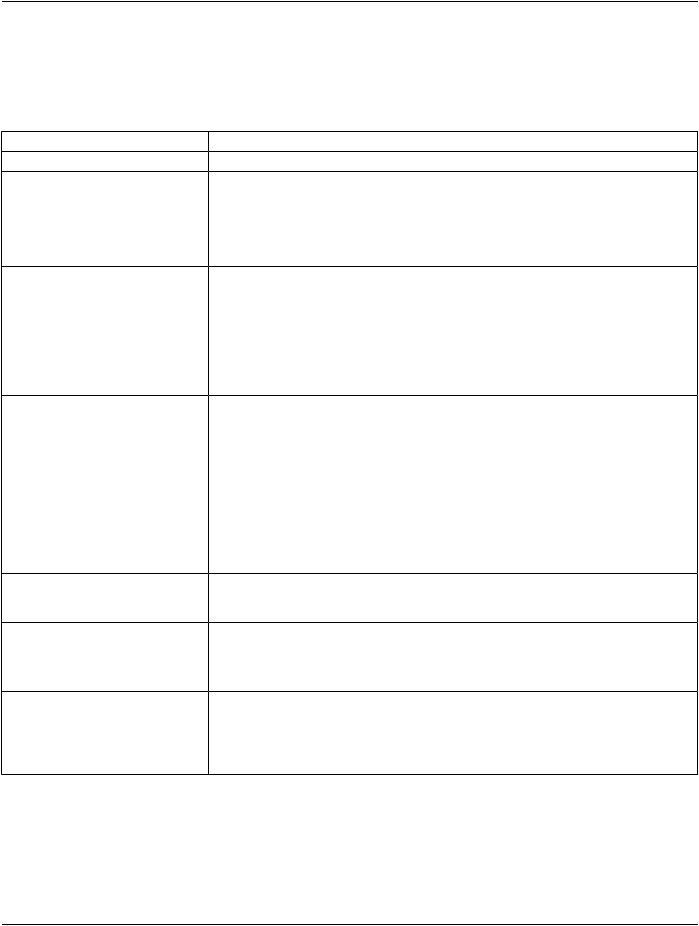
Functional Group Macro |
Section |
Usage |
|
Derivation Image |
C.7.6.16.2.6 |
C - Required if Image Type (0008,0008) |
|
|
Value 1 equals DERIVED. |
|
Frame Anatomy |
C.7.6.16.2.8 |
M |
|
Pixel Value Transformation |
C.7.6.16.2.9 |
U |
|
Frame VOI LUT |
C.7.6.16.2.10 |
M |
|
Real World Value Mapping |
C.7.6.16.2.11 |
U |
|
Contrast/Bolus Usage |
C.7.6.16.2.12 |
C - Required if the Enhanced |
|
|
|
Contrast/Bolus Module is present. |
X-Ray 3D Frame Type |
C.8.21.5.1 |
M |
|
A.54.4.1 X-Ray 3D Craniofacial Image Functional Group Macros Content Constraints
A.54.4.1.1 Frame Anatomy Functional Group Macro
The Defined CID for the Anatomic Region Sequence (0008,2218) shall be CID 4028 “Craniofacial Anatomic Regions” or CID 4016 “Anatomic Region for Intra-oral Radiography”.
In the case of CID 4016 “Anatomic Region for Intra-oral Radiography”, the Defined CID for the Anatomic Region Modifier Sequence (0008,2220) shall be CID 4017 “Anatomic Region Modifier for Intra-oral Radiography”, if present, and the Defined CID for the Primary AnatomicStructureSequence(0008,2228)shallbeCID4026“PrimaryAnatomicStructureforIntra-oralandCraniofacialRadiography - Teeth”, if present.
A.55 Breast Tomosynthesis Image IOD
A.55.1 Breast Tomosynthesis Image IOD Description
This Section defines the Information Object for multi-dimensional reconstructed breast tomosynthesis X-Ray Images that includes those Attributes and Information Objects necessary for the interchange of multi-dimensional breast tomosynthesis X-Ray volumes.
Note
This IOD is not intended for interchange of source projection X-Ray images.
A.55.2 Breast Tomosynthesis Image IOD Entity-Relationship Model
This IOD uses the E-R Model in Section A.1.2, with only the Image IE below the Series IE.
A.55.3 Breast Tomosynthesis Image IOD Module Table
Table A.55-1 specifies the Modules of the Breast Tomosynthesis Image IOD.
Table A.55-1. Breast Tomosynthesis Image IOD Modules
IE |
Module |
Reference |
Usage |
Patient |
Patient |
C.7.1.1 |
M |
|
Clinical Trial Subject |
C.7.1.3 |
U |
Study |
General Study |
C.7.2.1 |
M |
|
Patient Study |
C.7.2.2 |
U |
|
Clinical Trial Study |
C.7.2.3 |
U |
Series |
General Series |
C.7.3.1 |
M |
|
Clinical Trial Series |
C.7.3.2 |
U |
|
Enhanced Mammography Series |
C.8.11.10 |
M |