Материал: part03
DICOM PS3.3 2020a - Information Object Definitions |
Page 321 |
A.35.13.3.1 Comprehensive 3D SR IOD Content Constraints
A.35.13.3.1.1 Value Type
Value Type (0040,A040) in Content Sequence (0040,A730) of the SR Document Content Module is constrained to the following Enumerated Values (see Table C.17.3-7 for Value Type definitions):
Enumerated Values:
TEXT
CODE
NUM
DATETIME
DATE
TIME
UIDREF
PNAME
SCOORD
SCOORD3D
TCOORD
COMPOSITE
IMAGE
WAVEFORM
CONTAINER
A.35.13.3.1.2 Relationship Constraints
Relationships between Content Items in the content of this IOD may be conveyed either by-value or by-reference. Table A.35.13-2 specifies the relationship constraints of this IOD. See Table C.17.3-8 for Relationship Type definitions.
Table A.35.13-2. Relationship Content Constraints for Comprehensive 3D SR IOD
Source Value Type |
RelationshipType(Enumerated |
Target Value Type |
|
Values) |
|
CONTAINER |
CONTAINS |
TEXT, CODE, NUM, |
HAS OBS CONTEXT |
CONTAINER |
|
CONTAINER, IMAGE1, |
HAS ACQ CONTEXT |
WAVEFORM1, COMPOSITE1, |
|
NUM |
|
any type |
HAS CONCEPT MOD |
TEXT, CODE, NUM |
HAS PROPERTIES |
PNAME |
HAS PROPERTIES |
TEXT, CODE, NUM |
INFERRED FROM |
SCOORD |
SELECTED FROM |
TCOORD |
SELECTED FROM |
Note |
|
TEXT, CODE, NUM, DATETIME, DATE, TIME, UIDREF, PNAME, SCOORD, SCOORD3D, TCOORD, COMPOSITE1, IMAGE1, WAVEFORM1, CONTAINER (see below).
TEXT, CODE, NUM, DATETIME, DATE, TIME, UIDREF,
PNAME, COMPOSITE1
TEXT, CODE, NUM, DATETIME, DATE, TIME, UIDREF, PNAME, CONTAINER
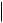 TEXT, CODE2
TEXT, CODE2
TEXT, CODE, NUM, DATETIME, DATE, TIME, UIDREF, PNAME, IMAGE1, WAVEFORM1, COMPOSITE1, SCOORD, SCOORD3D, TCOORD, CONTAINER
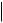 TEXT, CODE, DATETIME, DATE, TIME, UIDREF, PNAME
TEXT, CODE, DATETIME, DATE, TIME, UIDREF, PNAME 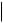
TEXT, CODE, NUM, DATETIME, DATE, TIME, UIDREF, PNAME, IMAGE1, WAVEFORM1, COMPOSITE1, SCOORD, SCOORD3D, TCOORD, CONTAINER
 IMAGE1
IMAGE1
SCOORD, SCOORD3D, IMAGE1, WAVEFORM1
1.WhichSOPClassestheIMAGE,WAVEFORMorCOMPOSITEValueTypemayreferto,isdocumentedintheConform- ance Statement for an application (see PS3.2 and PS3.4).
- Standard -

Page 322 |
DICOM PS3.3 2020a - Information Object Definitions |
2.The HAS CONCEPT MOD relationship is used to modify the meaning of the Concept Name of a Source Content Item, for example to provide a more descriptive explanation, a different language translation, or to define a post-coordinated concept.
3.SCOORD3D has no children, since its coordinates are patient rather than image-relative.
The HAS CONCEPT MOD and CONTAINS relationships shall not be conveyed by-reference.
Relationships by-reference to ancestor Content Items are forbidden in this IOD to prevent loops.
A.35.14 Radiopharmaceutical Radiation Dose SR IOD
A.35.14.1 Radiopharmaceutical Radiation Dose SR IOD Description
The Radiopharmaceutical Radiation Dose SR IOD is used to convey the exposure characteristics and dose from the administration of radiopharmaceuticals.
A.35.14.2 Radiopharmaceutical Radiation Dose SR IOD Entity-Relationship Model
This IOD uses the E-R Model in Section A.1.2, with only the SR Document IE below the Series IE.
A.35.14.3 Radiopharmaceutical Radiation Dose SR IOD Module Table
Table A.35.14-1 specifies the Modules of the Radiopharmaceutical Radiation Dose SR IOD.
Table A.35.14-1. Radiopharmaceutical Radiation Dose SR IOD Modules
IE |
Module |
Reference |
Usage |
Patient |
Patient |
C.7.1.1 |
M |
|
Clinical Trial Subject |
C.7.1.3 |
U |
Study |
General Study |
C.7.2.1 |
M |
|
Patient Study |
C.7.2.2 |
U |
|
Clinical Trial Study |
C.7.2.3 |
U |
Series |
SR Document Series |
C.17.1 |
M |
|
Clinical Trial Series |
C.7.3.2 |
U |
Frame of |
Synchronization |
C.7.4.2 |
C-shallbepresentifsystemtime |
Reference |
|
|
is synchronized to an external |
|
|
|
reference. May be present |
|
|
|
otherwise. |
Equipment |
General Equipment |
C.7.5.1 |
M |
|
Enhanced General Equipment |
C.7.5.2 |
M |
Document |
SR Document General |
C.17.2 |
M |
|
SR Document Content |
C.17.3 |
M |
|
SOP Common |
C.12.1 |
M |
A.35.14.3.1 Radiopharmaceutical Radiation Dose SR IOD Content Constraints
A.35.14.3.1.1 Template
The document may be constructed from Baseline TID 10021 “Radiopharmaceutical Radiation Dose” invoked at the root node.
A.35.14.3.1.2 Value Type
Value Type (0040,A040) in Content Sequence (0040,A730) of the SR Document Content Module is constrained to the following Enumerated Values (see Table C.17.3-7 for Value Type definitions):
- Standard -
DICOM PS3.3 2020a - Information Object Definitions |
Page 323 |
Enumerated Values:
TEXT
CODE
NUM
DATETIME
UIDREF
PNAME
CONTAINER
A.35.14.3.1.3 Relationship Constraints
RelationshipsbetweenContentItemsinthecontentofthisIODshallbeconveyedby-value.TableA.35.14-2specifiestherelationship constraints of this IOD. See Table C.17.3-8 for Relationship Type definitions.
Table A.35.14-2. Relationship Content Constraints for Radiopharmaceutical Radiation Dose SR IOD
Source Value Type |
Relationship Type (Enumerated |
Target Value Type |
|
Values) |
|
CONTAINER |
CONTAINS |
TEXT, CODE, NUM, DATETIME, UIDREF, PNAME, |
|
|
CONTAINER |
TEXT, CODE, NUM |
HAS OBS CONTEXT |
TEXT, CODE, NUM, DATETIME, UIDREF, PNAME |
CONTAINER |
HAS ACQ CONTEXT |
TEXT, CODE, NUM, DATETIME, UIDREF, PNAME, |
|
|
CONTAINER |
any type |
HAS CONCEPT MOD |
TEXT, CODE |
TEXT, CODE, NUM, PNAME |
HAS PROPERTIES |
TEXT, CODE, NUM, DATETIME, UIDREF, PNAME, |
|
|
CONTAINER |
TEXT, CODE, NUM |
INFERRED FROM |
TEXT,CODE,NUM,DATETIME,UIDREF,CONTAINER |
A.35.15 Extensible SR IOD
A.35.15.1 Extensible SR IOD Description
The Extensible SR IOD specifies support for all SR Content Item and relationship types currently defined and that may be added in the future.
It is designed specifically to support the possibility of future extensions, and the corresponding SOP Class defined in PS3.4 has specific behavior defined for a recipient in the presence of unrecognized or unsupported extensions.
A.35.15.2 Extensible SR IOD Entity-Relationship Model
This IOD uses the E-R Model in Section A.1.2, with only the SR Document IE below the Series IE.
A.35.15.3 Extensible SR IOD Module Table
Table A.35.15-1 specifies the Modules of the Extensible SR IOD.
Table A.35.15-1. Extensible SR IOD Modules
IE |
Module |
Reference |
Usage |
Patient |
Patient |
C.7.1.1 |
M |
|
Clinical Trial Subject |
C.7.1.3 |
U |
Study |
General Study |
C.7.2.1 |
M |
|
Patient Study |
C.7.2.2 |
U |
|
Clinical Trial Study |
C.7.2.3 |
U |
- Standard -

Page 324 |
DICOM PS3.3 2020a - Information Object Definitions |
|
|
IE |
Module |
Reference |
Usage |
Series |
SR Document Series |
C.17.1 |
M |
|
Clinical Trial Series |
C.7.3.2 |
U |
Frame of ReferenceSynchronization |
C.7.4.2 |
U |
|
Equipment |
General Equipment |
C.7.5.1 |
M |
|
Enhanced General Equipment |
C.7.5.2 |
M |
Document |
SR Document General |
C.17.2 |
M |
|
SR Document Content |
C.17.3 |
M |
|
SOP Common |
C.12.1 |
M |
A.35.15.3.1 Extensible SR IOD Content Constraints
A.35.15.3.1.1 Value Type
Value Type (0040,A040) in the Content Sequence (0040,A730) of the SR Document Content Module is constrained to any of the Enumerated Values defined in Section C.17.3.
A.35.15.3.1.2 Relationship Constraints
Relationships between Content Items in the content of this IOD may be conveyed either by-value or by-reference. Table A.35.15-2 specifies the relationship constraints of this IOD. See Table C.17.3-8 for Relationship Type definitions.
Table A.35.15-2. Relationship Content Constraints for Extensible SR IOD
Source Value Type |
Relationship Type (Enumerated Values) |
Target Value Type |
CONTAINER |
CONTAINS |
any type. |
any type |
any type other than CONTAINS |
any type. |
Note |
|
|
Thelackofexplicitconstraintsbetweenvaluetypesandrelationshiptypesisnecessarytosupportextensibilityinunanticipated ways (e.g., to support new coordinate value types that may be selected from new composite object value types), but that does not mean that all possible combinations will make sense or be usable (e.g., HAS OBS CONTEXT with source and target value types of IMAGE).
Relationships by-reference to ancestor Content Items are forbidden in this IOD to prevent loops.
A.35.16 Acquisition Context SR IOD
A.35.16.1 Acquisition Context SR IOD Description
The Acquisition Context SR IOD is used to represent the description of the conditions present during data acquisition of other data that is stored separately (such as images).
A.35.16.2 Acquisition Context SR IOD Entity-Relationship Model
This IOD uses the E-R Model in Section A.1.2, with only the SR Document IE below the Series IE.
A.35.16.3 Acquisition Context SR IOD Module Table
Table A.35.16-1 specifies the Modules of the Acquisition Context SR IOD.
- Standard -
DICOM PS3.3 2020a - Information Object Definitions Page 325
Table A.35.16-1. Acquisition Context SR IOD Modules
IE |
Module |
Reference |
Usage |
Patient |
Patient |
C.7.1.1 |
M |
|
Clinical Trial Subject |
C.7.1.3 |
U |
Study |
General Study |
C.7.2.1 |
M |
|
Patient Study |
C.7.2.2 |
U |
|
Clinical Trial Study |
C.7.2.3 |
U |
Series |
SR Document Series |
C.17.1 |
M |
|
Clinical Trial Series |
C.7.3.2 |
U |
Frame of |
Synchronization |
C.7.4.2 |
C - Required if time |
Reference |
|
|
synchronizationwasapplied |
Equipment |
General Equipment |
C.7.5.1 |
M |
|
Enhanced General Equipment |
C.7.5.2 |
M |
Document |
SR Document General |
C.17.2 |
M |
|
SR Document Content |
C.17.3 |
M |
|
SOP Common |
C.12.1 |
M |
A.35.16.3.1 Acquisition Context SR IOD Content Constraints
A.35.16.3.1.1 Value Type
Value Type (0040,A040) in Content Sequence (0040,A730) of the SR Document Content Module is constrained to the following Enumerated Values (see Table C.17.3-7 for Value Type definitions):
Enumerated Values:
TEXT
CODE
NUM
DATETIME
DATE
TIME
UIDREF
PNAME
SCOORD3D
CONTAINER
A.35.16.3.1.2 Relationship Constraints
Relationships between Content Items in the content of this IOD shall be conveyed in the by-value mode. See Table C.17.3-8 for Re- lationship Type definitions.
Note
Relationships by-reference are forbidden. Therefore, Referenced Content Item Identifier (0040,DB73) is not present in any of the Content Items within the SR Document Content Module.
Table A.35.16-2 specifies the relationship constraints of this IOD.
Table A.35.16-2. Relationship Content Constraints for Acquisition Context SR IOD
Source Value Type |
Relationship |
Type (Enumerated Values) |
Target Value Type |
CONTAINER |
CONTAINS |
CODE,CONTAINER,DATETIME,NUM,PNAME,TEXT, |
|
|
|
TIME, UIDREF |
|
- Standard -