Материал: part03
DICOM PS3.3 2020a - Information Object Definitions Page 261
Table A.31.3-1. RT Treatment Summary Record IOD Modules
IE |
Module |
Reference |
Usage |
Patient |
Patient |
C.7.1.1 |
M |
|
Clinical Trial Subject |
C.7.1.3 |
U |
Study |
General Study |
C.7.2.1 |
M |
|
Patient Study |
C.7.2.2 |
U |
|
Clinical Trial Study |
C.7.2.3 |
U |
Series |
RT Series |
C.8.8.1 |
M |
|
Clinical Trial Series |
C.7.3.2 |
U |
Equipment |
General Equipment |
C.7.5.1 |
M |
Treatment |
RT General Treatment Record |
C.8.8.17 |
M |
Record |
RT Treatment Summary Record |
C.8.8.23 |
M |
|
|||
|
General Reference |
C.12.4 |
U |
|
SOP Common |
C.12.1 |
M |
|
Common Instance Reference |
C.12.2 |
U |
Note
The Curve Module (Retired) was previously included in this IOD but has been retired. See PS3.3-2004.
A.32 Visible Light Image Information Object Definitions
The Visible Light (VL) Image Information Object Definition (IOD) specifies images that are acquired by means of a camera or other sensors that are sensitive to visible or near-visible light.
Examples of types of equipment that create Visible Light Images include:
a.Rigid and flexible endoscopy equipment
b.Operation microscopes / colposcopes
c.Ophthalmology equipment
d.Digital or Video Cameras
e.Analysis microscopes
Separate IODs have been defined for specialized applications. Some support only single-frame images, some support both single- frame and multi-frame video images.
A.32.1 VL Endoscopic Image IOD
A.32.1.1 VL Endoscopic Image IOD Description
The VL Endoscopic Image IOD specifies the Attributes of Single-frame VL Endoscopic Images.
A.32.1.2 VL Endoscopic Image IOD Entity-Relationship Model
This IOD uses the E-R Model in Section A.1.2, with only the Image IE below the Series IE. The Frame of Reference IE is not a com- ponent of this IOD.
- Standard -
Page 262 |
DICOM PS3.3 2020a - Information Object Definitions |
Note
1.An endoscopic procedure might include multiple Series of single-frame endoscopic images as well as one or more ad- ditional Series of related diagnostic images. The procedure might involve multiple Performed Procedure Steps, multiple endoscopes, and multiple anatomic regions and might be supervised, performed, and/or interpreted by one or more in- dividuals.
2.Several distinct diagnostic or therapeutic processes might occur during an endoscopic procedure. For example: Endo- scopic examination of duodenal mucosa, biopsy, lavage, or biliary stone removal.
3.The Curve entity was previously include in the list of entities that are not used, but has been retired from DICOM. It is still not used in this IOD. See PS3.3-2004.
Table A.32.1-1. VL Endoscopic Image IOD Modules
IE |
Module |
Reference |
Usage |
Patient |
Patient |
C.7.1.1 |
M |
|
Clinical Trial Subject |
C.7.1.3 |
U |
Study |
General Study |
C.7.2.1 |
M |
|
Patient Study |
C.7.2.2 |
U |
|
Clinical Trial Study |
C.7.2.3 |
U |
Series |
General Series |
C.7.3.1 |
M |
|
Clinical Trial Series |
C.7.3.2 |
U |
Equipment |
General Equipment |
C.7.5.1 |
M |
Image |
General Image |
C.7.6.1 |
M |
|
General Reference |
C.12.4 |
U |
|
Image Pixel |
C.7.6.3 |
M |
|
Acquisition Context |
C.7.6.14 |
M |
|
Device |
C.7.6.12 |
U |
|
Specimen |
C.7.6.22 |
U |
|
VL Image |
C.8.12.1 |
M |
|
Overlay Plane |
C.9.2 |
U |
|
ICC Profile |
C.11.15 |
U |
|
SOP Common |
C.12.1 |
M |
|
Common Instance Reference |
C.12.2 |
U |
A.32.1.3 VL Endoscopic Image IOD Content Constraints
A.32.1.3.1 Modality
The value of Modality (0008,0060) shall be ES.
A.32.2 VL Microscopic Image IOD
A.32.2.1 VL Microscopic Image IOD Description
The VL Microscopic Image IOD specifies the Attributes of Single-frame VL Microscopic Images, including both imaging of specimens and direct microscopic imaging of the Patient (e.g., perioperative microscopy). Microscopic Images with Slide Coordinates shall not be encoded with this IOD.
- Standard -
DICOM PS3.3 2020a - Information Object Definitions |
Page 263 |
A.32.2.2 VL Microscopic Image IOD Entity-Relationship Model
This IOD uses the E-R Model in Section A.1.2, with only the Image IE below the Series IE. The Frame of Reference IE is not a com- ponent of this IOD.
Note
1.The Curve entity was previously include in the list of entities that are not used, but has been retired from DICOM. It is still not used in this IOD. See PS3.3-2004.
2.TheSpecimenIdentificationModulewaspreviouslyincludedinthisIODbuthasbeenretired,anditsfunctionalityreplaced by the Specimen Module. See PS3.3-2008.
Table A.32.1-2. VL Microscopic Image IOD Modules
IE |
Module |
Reference |
Usage |
Patient |
Patient |
C.7.1.1 |
M |
|
Clinical Trial Subject |
C.7.1.3 |
U |
Study |
General Study |
C.7.2.1 |
M |
|
Patient Study |
C.7.2.2 |
U |
|
Clinical Trial Study |
C.7.2.3 |
U |
Series |
General Series |
C.7.3.1 |
M |
|
Clinical Trial Series |
C.7.3.2 |
U |
Equipment General Equipment |
C.7.5.1 |
M |
|
Image |
General Image |
C.7.6.1 |
M |
|
General Reference |
C.12.4 |
U |
|
Image Pixel |
C.7.6.3 |
M |
|
Acquisition Context |
C.7.6.14 |
M |
|
Device |
C.7.6.12 |
U |
|
Specimen |
C.7.6.22 |
C - Required if Imaging |
|
|
|
Subject is a specimen |
|
VL Image |
C.8.12.1 |
M |
|
Optical Path |
C.8.12.5 |
U |
|
Overlay Plane |
C.9.2 |
U |
|
ICC Profile |
C.11.15 |
U |
|
SOP Common |
C.12.1 |
M |
|
Common Instance Reference |
C.12.2 |
U |
A.32.2.3 VL Microscopic Image IOD Content Constraints
A.32.2.3.1 Modality
The value of Modality (0008,0060) shall be GM.
A.32.3 VL Slide-Coordinates Microscopic Image IOD
A.32.3.1 VL Slide-Coordinates Microscopic Image IOD Description
The VL Slide-Coordinates Microscopic Image IOD specifies the Attributes of VL single frame Slide-Coordinates Microscopic Images.
- Standard -
Page 264 |
DICOM PS3.3 2020a - Information Object Definitions |
A.32.3.2 VL Slide-Coordinates Microscopic Image IOD Entity-Relationship Model
This IOD uses the E-R Model in Section A.1.2, with only the Image IE below the Series IE.
Note
1.The Curve entity was previously include in the list of entities that are not used, but has been retired from DICOM. It is still not used in this IOD. See PS3.3-2004.
2.TheSpecimenIdentificationModulewaspreviouslyincludedinthisIODbuthasbeenretired,anditsfunctionalityreplaced by the Specimen Module. See PS3.3-2008.
3.TheFrameofReferenceIEwaspreviously(incorrectly)identifiedasnotusedinthisIOD,althoughtheFrameofReference Module was specified as Mandatory. See PS3.3-2009.
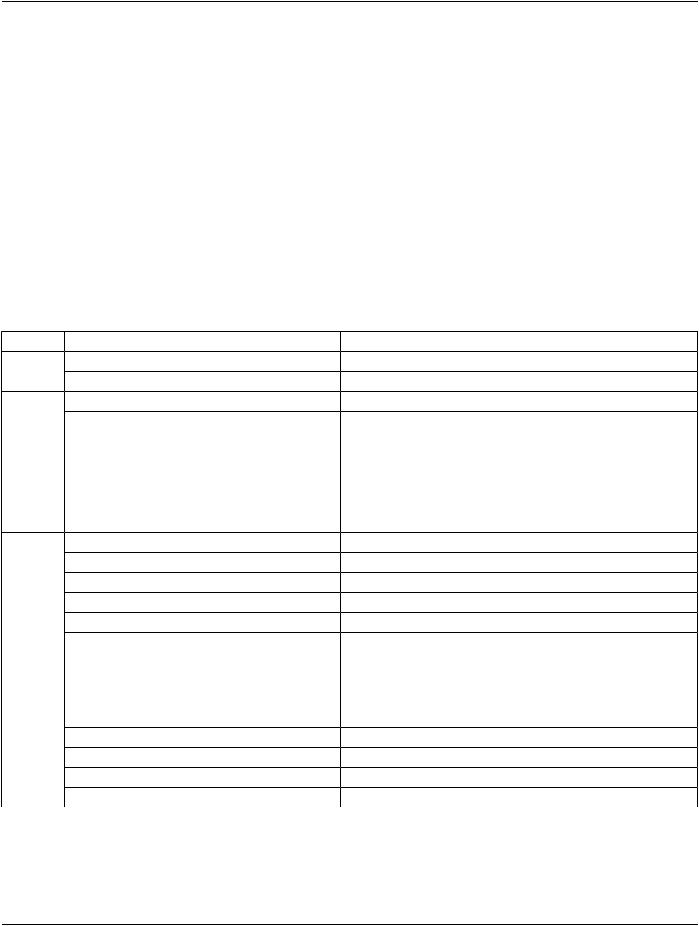
Table A.32.1-3. VL Slide-Coordinates Microscopic Image IOD Modules
IE |
Module |
Reference |
Usage |
Patient |
Patient |
C.7.1.1 |
M |
|
Clinical Trial Subject |
C.7.1.3 |
U |
Study |
General Study |
C.7.2.1 |
M |
|
Patient Study |
C.7.2.2 |
U |
|
Clinical Trial Study |
C.7.2.3 |
U |
Series |
General Series |
C.7.3.1 |
M |
|
Clinical Trial Series |
C.7.3.2 |
U |
Frame of |
Frame of Reference |
C.7.4.1 |
M |
Reference |
|
|
|
Equipment |
General Equipment |
C.7.5.1 |
M |
Image |
General Image |
C.7.6.1 |
M |
|
General Reference |
C.12.4 |
U |
|
Image Pixel |
C.7.6.3 |
M |
|
Acquisition Context |
C.7.6.14 |
M |
|
Device |
C.7.6.12 |
U |
|
Specimen |
C.7.6.22 |
M |
|
VL Image |
C.8.12.1 |
M |
|
Slide Coordinates |
C.8.12.2 |
M |
|
Optical Path |
C.8.12.5 |
U |
|
Overlay Plane |
C.9.2 |
U |
|
ICC Profile |
C.11.15 |
U |
|
SOP Common |
C.12.1 |
M |
|
Common Instance Reference |
C.12.2 |
U |
A.32.3.3 VL Slide-coordinates Microscopic Image IOD Content Constraints
A.32.3.3.1 Modality
The value of Modality (0008,0060) shall be SM.
- Standard -
DICOM PS3.3 2020a - Information Object Definitions |
Page 265 |
A.32.4 VL Photographic Image IOD
A.32.4.1 VL Photographic Image IOD Description
The VL Photographic Image IOD specifies the Attributes of VL Single-frame photographic Images.
A.32.4.2 VL Photographic Image IOD Entity-Relationship Model
This IOD uses the E-R Model in Section A.1.2, with only the Image IE below the Series IE. The Frame of Reference IE is not a com- ponent of this IOD.
Note
1.The Curve entity was previously include in the list of entities that are not used, but has been retired from DICOM. It is still not used in this IOD. See PS3.3-2004.
2.TheSpecimenIdentificationModulewaspreviouslyincludedinthisIODbuthasbeenretired,anditsfunctionalityreplaced by the Specimen Module. See PS3.3-2008.
Table A.32.4-1. VL Photographic Image IOD Modules
IE |
Module |
Reference |
Usage |
Patient |
Patient |
C.7.1.1 |
M |
|
Clinical Trial Subject |
C.7.1.3 |
U |
Study |
General Study |
C.7.2.1 |
M |
|
Patient Study |
C.7.2.2 |
U |
|
Clinical Trial Study |
C.7.2.3 |
U |
Series |
General Series |
C.7.3.1 |
M |
|
Clinical Trial Series |
C.7.3.2 |
U |
EquipmentGeneral Equipment |
C.7.5.1 |
M |
|
|
VL Photographic Equipment |
C.8.12.10 |
U |
Image |
General Image |
C.7.6.1 |
M |
|
General Reference |
C.12.4 |
U |
|
Image Pixel |
C.7.6.3 |
M |
|
Acquisition Context |
C.7.6.14 |
M |
|
Device |
C.7.6.12 |
U |
|
Specimen |
C.7.6.22 |
C - Required if Imaging |
|
|
|
Subject is a specimen |
|
VL Image |
C.8.12.1 |
M |
|
VL Photographic Acquisition |
C.8.12.11 |
U |
|
VL Photographic Geolocation |
C.8.12.12 |
U |
|
Overlay Plane |
C.9.2 |
U |
|
ICC Profile |
C.11.15 |
U |
|
SOP Common |
C.12.1 |
M |
|
Common Instance Reference |
C.12.2 |
U |
- Standard -