Материал: part03
Page 256 |
DICOM PS3.3 2020a - Information Object Definitions |
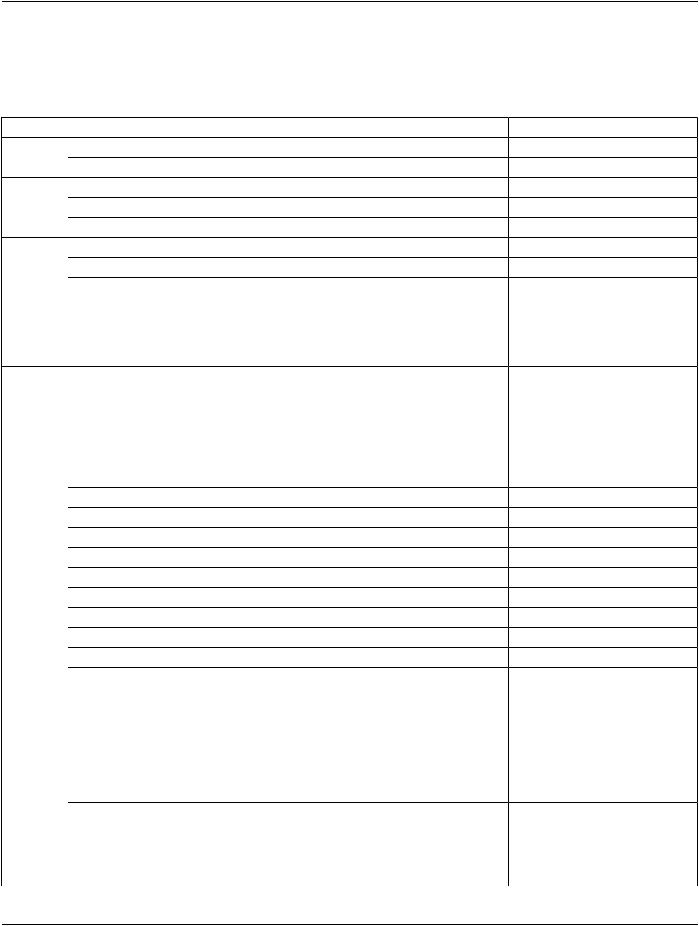
A.27.3 Digital Mammography X-Ray Image IOD Module Table
Table A.27-1 specifies the Modules of the Digital Mammography X-Ray Image IOD.
Table A.27-1. Digital Mammography X-Ray Image IOD Modules
IE |
Module |
Reference |
Usage |
Patient |
Patient |
C.7.1.1 |
M |
|
Clinical Trial Subject |
C.7.1.3 |
U |
Study |
General Study |
C.7.2.1 |
M |
|
Patient Study |
C.7.2.2 |
U |
|
Clinical Trial Study |
C.7.2.3 |
U |
Series |
General Series |
C.7.3.1 |
M |
|
Clinical Trial Series |
C.7.3.2 |
U |
|
DX Series |
C.8.11.1 |
M |
|
Mammography Series |
C.8.11.6 |
M |
Frame of |
Frame of Reference |
C.7.4.1 |
C - Required if multiple images are |
Reference |
|
obtained without releasing breast |
|
|
|
|
compression |
EquipmentGeneral Equipment |
C.7.5.1 |
M |
|
Image |
General Image |
C.7.6.1 |
M |
|
General Reference |
C.12.4 |
U |
|
Image Pixel |
C.7.6.3 |
M |
|
Contrast/Bolus |
C.7.6.4 |
U |
|
Display Shutter |
C.7.6.11 |
U |
|
Device |
C.7.6.12 |
U |
|
Intervention |
C.7.6.13 |
U |
|
Specimen |
C.7.6.22 |
U |
|
DX Anatomy Imaged |
C.8.11.2 |
M |
|
DX Image |
C.8.11.3 |
M |
|
DX Detector |
C.8.11.4 |
M |
|
X-Ray Collimator |
C.8.7.3 |
U |
|
DX Positioning |
C.8.11.5 |
U |
|
X-Ray Tomography Acquisition |
C.8.7.7 |
U |
|
X-Ray Acquisition Dose |
C.8.7.8 |
U |
|
X-Ray Generation |
C.8.7.9 |
U |
|
X-Ray Filtration |
C.8.7.10 |
U |
|
X-Ray Grid |
C.8.7.11 |
U |
|
Mammography Image |
C.8.11.7 |
M |
|
Overlay Plane |
C.9.2 |
C - Required if graphic annotation |
|
|
|
is present - See Section A.27.3 |
|
VOI LUT |
C.11.2 |
C - Required if Presentation Intent |
|
|
|
Type (0008,0068) is FOR |
|
|
|
PRESENTATION. Shall not be |
|
|
|
present otherwise. |
|
Image Histogram |
C.11.5 |
U |
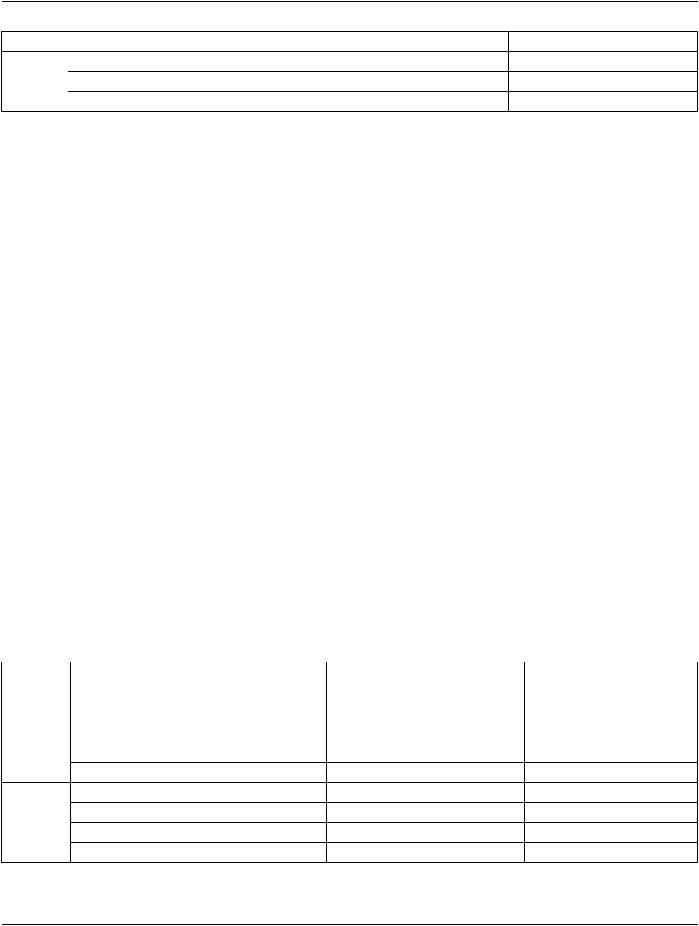
- Standard -
|
DICOM PS3.3 2020a - Information Object Definitions |
Page 257 |
|
IE |
Module |
Reference |
Usage |
|
Acquisition Context |
C.7.6.14 |
M |
|
SOP Common |
C.12.1 |
M |
|
Common Instance Reference |
C.12.2 |
U |
Note
1.The Curve Module (Retired) was previously included in this IOD but has been retired. See PS3.3-2004.
2.TheSpecimenIdentificationModulewaspreviouslyincludedinthisIODbuthasbeenretired,anditsfunctionalityreplaced by the Specimen Module. See PS3.3-2008.
A.27.4 Overlay Plane Module
If the Overlay Plane Module is present, any Overlays defined in that Module shall store the overlay data in Overlay Data (60xx,3000), and not any unused high bits in Pixel Data (7FE0,0010).
A.28 Digital Intra-Oral X-Ray Image IOD
A.28.1 Digital Intra-Oral X-Ray Image IOD Description
The Digital Intra-Oral X-Ray Image Information Object Definition specifies an image that has been created by an intra-oral projection radiography imaging device.
Note
It meets all of the requirements of the DX IOD in Section A.26 in addition to those specified in this section.
The Digital Intra-Oral X-Ray Image IOD is used in two SOP Classes as defined in PS3.4 Storage Service Class, a SOP Class for storageofimagesintendedforpresentation,andaSOPClassforstorageofimagesintendedforfurtherprocessingbeforepresentation. These are distinguished by their SOP Class UID and by the Enumerated Value of the mandatory Attribute in the DX Series Module, Presentation Intent Type (0008,0068).
A.28.2 Digital Intra-Oral X-Ray Image IOD Entity-Relationship Model
This IOD uses the E-R Model in Section A.1.2, with only the Image IE below the Series IE.
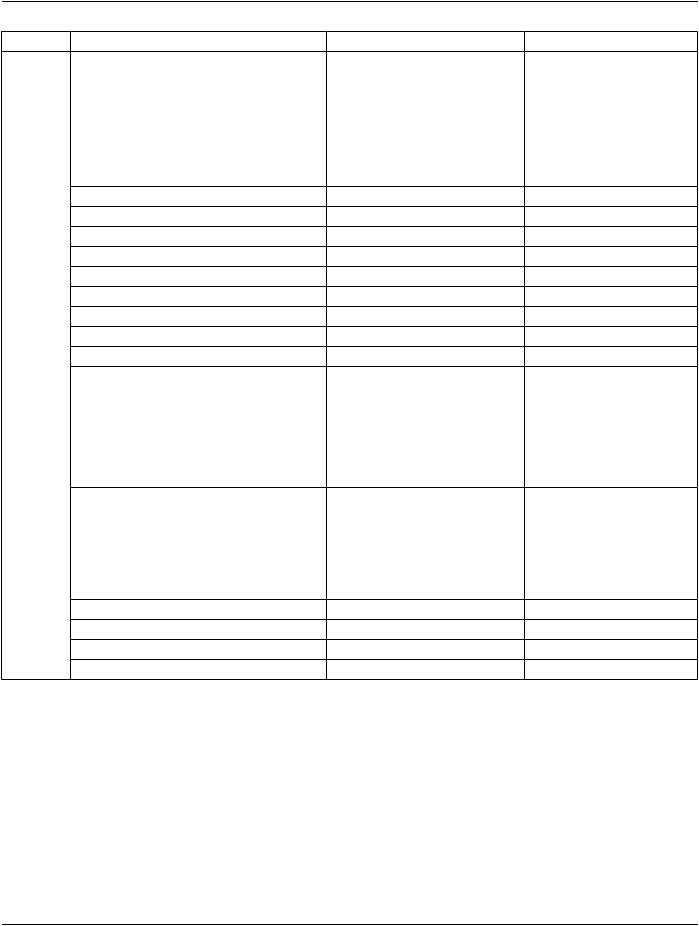
A.28.3 Digital Intra-Oral X-Ray Image IOD Module Table
Table A.28-1 specifies the Modules of the Digital Intra-Oral X-Ray Image IOD.
Table A.28-1. Digital Intra-Oral X-Ray Image IOD Modules
IE |
Module |
Reference |
Usage |
Patient |
Patient |
C.7.1.1 |
M |
|
Clinical Trial Subject |
C.7.1.3 |
U |
Study |
General Study |
C.7.2.1 |
M |
|
Patient Study |
C.7.2.2 |
U |
|
Clinical Trial Study |
C.7.2.3 |
U |
Series |
General Series |
C.7.3.1 |
M |
|
Clinical Trial Series |
C.7.3.2 |
U |
|
DX Series |
C.8.11.1 |
M |
|
Intra-Oral Series |
C.8.11.8 |
M |
- Standard -
Page 258 |
DICOM PS3.3 2020a - Information Object Definitions |
|
|
IE |
Module |
Reference |
Usage |
Frame of |
Frame of Reference |
C.7.4.1 |
U |
Reference |
|
|
|
EquipmentGeneral Equipment |
C.7.5.1 |
M |
|
Image |
General Image |
C.7.6.1 |
M |
|
General Reference |
C.12.4 |
U |
|
Image Pixel |
C.7.6.3 |
M |
|
Contrast/Bolus |
C.7.6.4 |
U |
|
Display Shutter |
C.7.6.11 |
U |
|
Device |
C.7.6.12 |
U |
|
Intervention |
C.7.6.13 |
U |
|
Specimen |
C.7.6.22 |
U |
|
DX Anatomy Imaged |
C.8.11.2 |
M |
|
DX Image |
C.8.11.3 |
M |
|
DX Detector |
C.8.11.4 |
M |
|
X-Ray Collimator |
C.8.7.3 |
U |
|
DX Positioning |
C.8.11.5 |
U |
|
X-Ray Tomography Acquisition |
C.8.7.7 |
U |
|
X-Ray Acquisition Dose |
C.8.7.8 |
U |
|
X-Ray Generation |
C.8.7.9 |
U |
|
X-Ray Filtration |
C.8.7.10 |
U |
|
X-Ray Grid |
C.8.7.11 |
U |
|
Intra-Oral Image |
C.8.11.9 |
M |
|
Overlay Plane |
C.9.2 |
C - Required if graphic |
|
|
|
annotation is present - See |
|
|
|
Section A.28.3 |
|
VOI LUT |
C.11.2 |
C - Required if Presentation |
|
|
|
Intent Type (0008,0068) is FOR |
|
|
|
PRESENTATION. Shall not be |
|
|
|
present otherwise. |
|
Image Histogram |
C.11.5 |
U |
|
Acquisition Context |
C.7.6.14 |
M |
|
SOP Common |
C.12.1 |
M |
|
Common Instance Reference |
C.12.2 |
U |
Note
1.The Curve Module (Retired) was previously included in this IOD but has been retired. See PS3.3-2004.
2.TheSpecimenIdentificationModulewaspreviouslyincludedinthisIODbuthasbeenretired,anditsfunctionalityreplaced by the Specimen Module. See PS3.3-2008.
A.28.4 Overlay Plane Module
If the Overlay Plane Module is present, any Overlays defined in that Module shall store the overlay data in Overlay Data (60xx,3000), and not any unused high bits in Pixel Data (7FE0,0010).
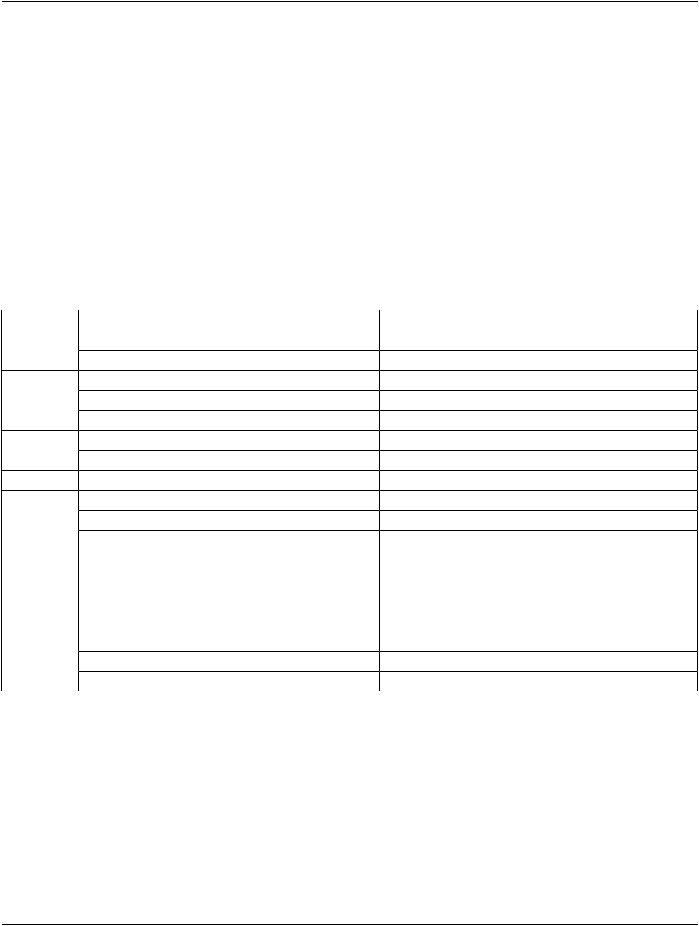
- Standard -
DICOM PS3.3 2020a - Information Object Definitions |
Page 259 |
A.29 RT Beams Treatment Record IOD
A.29.1 RT Beams Treatment Record IOD Description
The focus for this Radiotherapy Beams Treatment Record IOD (RT Beams Treatment Record IOD) is to address the requirements for transfer of treatment session reports generated by a treatment verification system during a course of external beam treatment, with optional cumulative summary information. It may also be used for transfer of treatment information during delivery.
A.29.2 RT Beams Treatment Record IOD Entity-Relationship Model
This IOD uses the E-R Model in Section A.1.2, with only the Treatment Record IE below the Series IE. The Frame of Reference IE is not a component of this IOD.
A.29.3 RT Beams Treatment Record IOD Module Table
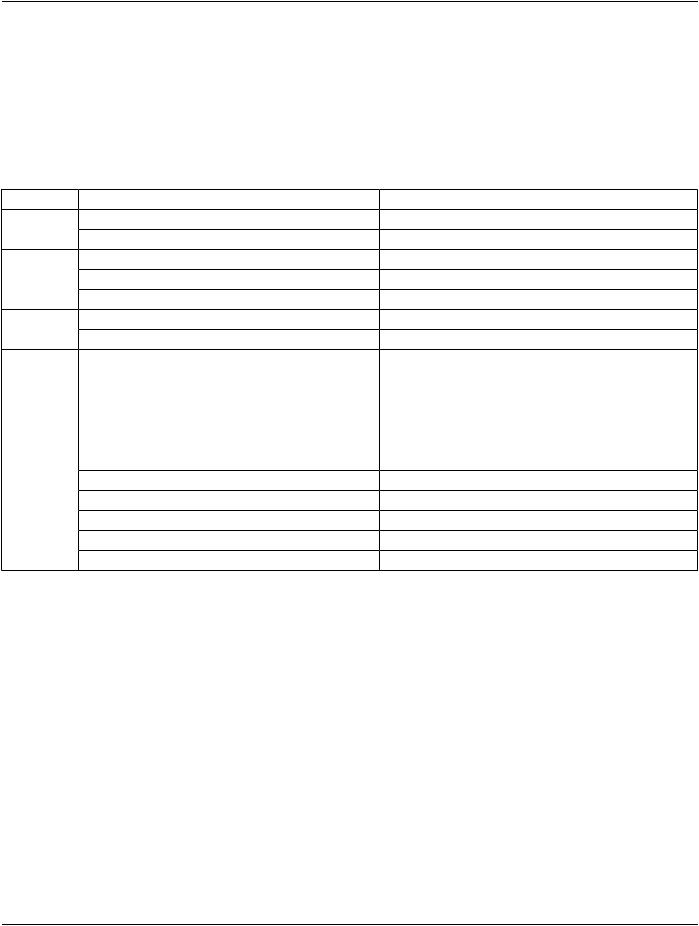
Table A.29.3-1 specifies the Modules of the RT Beams Treatment Record IOD.
Table A.29.3-1. RT Beams Treatment Record IOD Modules
IE |
Module |
Reference |
Usage |
Patient |
Patient |
C.7.1.1 |
M |
|
Clinical Trial Subject |
C.7.1.3 |
U |
Study |
General Study |
C.7.2.1 |
M |
|
Patient Study |
C.7.2.2 |
U |
|
Clinical Trial Study |
C.7.2.3 |
U |
Series |
RT Series |
C.8.8.1 |
M |
|
Clinical Trial Series |
C.7.3.2 |
U |
Equipment |
General Equipment |
C.7.5.1 |
M |
Treatment |
RT General Treatment Record |
C.8.8.17 |
M |
Record |
RT Patient Setup |
C.8.8.12 |
U |
|
|||
|
RT Treatment Machine Record |
C.8.8.18 |
M |
|
Measured Dose Reference Record |
C.8.8.19 |
U |
|
Calculated Dose Reference Record |
C.8.8.20 |
U |
|
RT Beams Session Record |
C.8.8.21 |
M |
|
RT Treatment Summary Record |
C.8.8.23 |
U |
|
General Reference |
C.12.4 |
U |
|
SOP Common |
C.12.1 |
M |
|
Common Instance Reference |
C.12.2 |
U |
Note
The Curve Module (Retired) was previously included in this IOD but has been retired. See PS3.3-2004.
A.30 RT Brachy Treatment Record IOD
A.30.1 RT Brachy Treatment Record IOD Description
The focus for this Radiotherapy Brachy Treatment Record IOD (RT Brachy Treatment Record IOD) is to address the requirements for transfer of treatment session reports generated by a treatment verification system during a course of Brachytherapy treatment, with optional cumulative summary information. It may also be used for transfer of treatment information during delivery.
- Standard -
Page 260 |
DICOM PS3.3 2020a - Information Object Definitions |
A.30.2 RT Brachy Treatment Record IOD Entity-Relationship Model
This IOD uses the E-R Model in Section A.1.2, with only the Treatment Record IE below the Series IE. The Frame of Reference IE is not a component of this IOD.
A.30.3 RT Brachy Treatment Record IOD Module Table
Table A.30.3-1 specifies the Modules of the RT Brachy Treatment Record IOD.
Table A.30.3-1. RT Brachy Treatment Record IOD Modules
IE |
Module |
Reference |
Usage |
Patient |
Patient |
C.7.1.1 |
M |
|
Clinical Trial Subject |
C.7.1.3 |
U |
Study |
General Study |
C.7.2.1 |
M |
|
Patient Study |
C.7.2.2 |
U |
|
Clinical Trial Study |
C.7.2.3 |
U |
Series |
RT Series |
C.8.8.1 |
M |
|
Clinical Trial Series |
C.7.3.2 |
U |
Equipment |
General Equipment |
C.7.5.1 |
M |
Treatment |
RT General Treatment Record |
C.8.8.17 |
M |
Record |
RT Patient Setup |
C.8.8.12 |
U |
|
|||
|
RT Treatment Machine Record |
C.8.8.18 |
M |
|
Measured Dose Reference Record |
C.8.8.19 |
U |
|
Calculated Dose Reference Record |
C.8.8.20 |
U |
|
RT Brachy Session Record |
C.8.8.22 |
M |
|
RT Treatment Summary Record |
C.8.8.23 |
U |
|
General Reference |
C.12.4 |
U |
|
SOP Common |
C.12.1 |
M |
|
Common Instance Reference |
C.12.2 |
U |
Note
The Curve Module (Retired) was previously included in this IOD but has been retired. See PS3.3-2004.
A.31 RT Treatment Summary Record IOD
A.31.1 RT Treatment Summary Record IOD Description
ThefocusforthisRadiotherapyTreatmentSummaryRecordIOD(RTTreatmentSummaryRecordIOD)istoaddresstherequirements for transfer of cumulative summary information, normally generated at the completion of a course of treatment.
A.31.2 RT Treatment Summary Record IOD Entity-Relationship Model
This IOD uses the E-R Model in Section A.1.2, with only the Treatment Record IE below the Series IE. The Frame of Reference IE is not a component of this IOD.
A.31.3 RT Treatment Summary Record IOD Module Table
Table A.31.3-1 specifies the Modules of the RT Treatment Summary Record IOD.
- Standard -