Материал: part03
DICOM PS3.3 2020a - Information Object Definitions |
Page 191 |
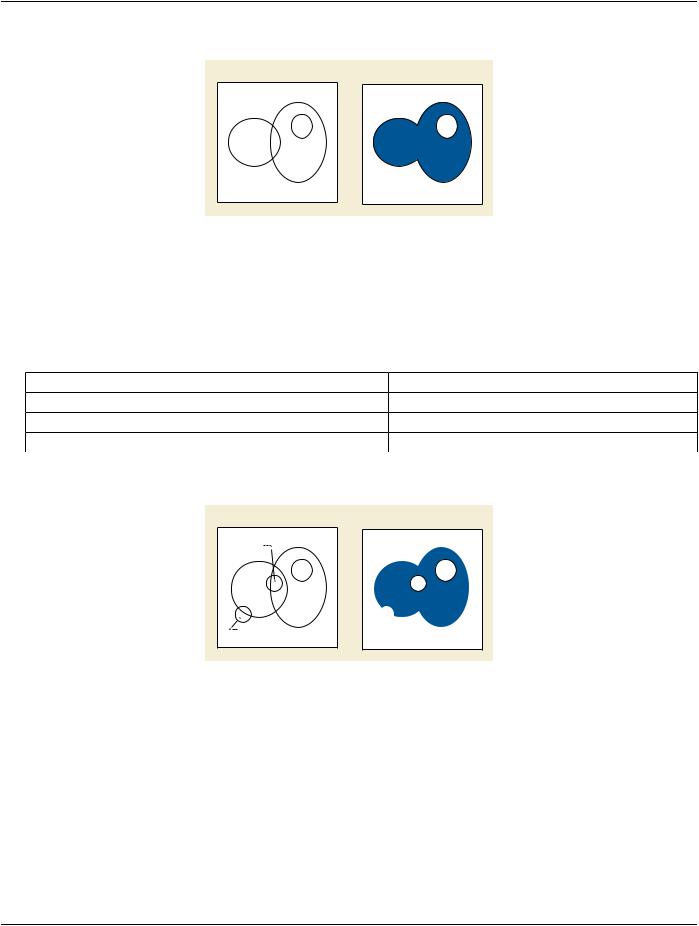
3.Union of two organs 1 and 2 with excluded volume 3 using NEGATION
Input |
Result |
CTV
(3)
Heart
(1)
Left Lung
(2)
Figure 10.34.1.1-3. Conceptual Volume Example of Intersection and Negation
Conceptual Volume Combination Expression (3010,000C): (INTERSECTION (UNION 1 2) (NEGATION 3) )
Items in Conceptual Volume Constituent Sequence (3010,0008):
Table 10.34.1.1-3. Conceptual Volume Example of Intersection and Negation
Conceptual Volume Constituent Index (3010,000D) |
Conceptual Volume |
1 |
Heart |
2 |
Left Lung |
3 |
CTV |
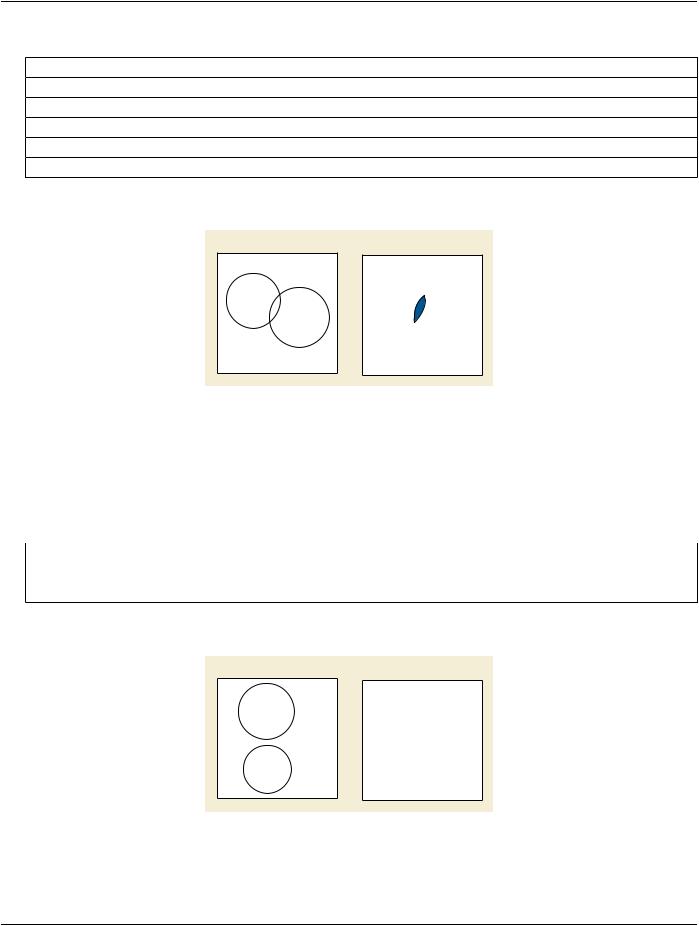
4.Union of paired organs 1 and 2, with exclusion of multiple volumes 3, 4 and 5 |
|
Input |
Result |
Node 2
(4)
CTV
Right (5)
Lung
(1)
Left Lung
(2)
Node 1
(3)
Figure 10.34.1.1-4. Conceptual Volume Example of Intersection and Union
Conceptual Volume Combination Expression (3010,000C): (INTERSECTION (UNION 1 2) (NEGATION (UNION 3 4 5) ))
Note
This combination can be expressed alternatively as: (SUBTRACTION (UNION 1 2) (UNION 3 4 5) )
Items in Conceptual Volume Constituent Sequence (3010,0008):
- Standard -
Page 192 DICOM PS3.3 2020a - Information Object Definitions
Table 10.34.1.1-4. Conceptual Volume Example of Intersection and Union
Conceptual Volume Constituent Index (3010,000D) |
Conceptual Volume |
1 |
R Lung |
2 |
L Lung |
3 |
Node 1 |
4 |
Node 2 |
5 |
CTV |
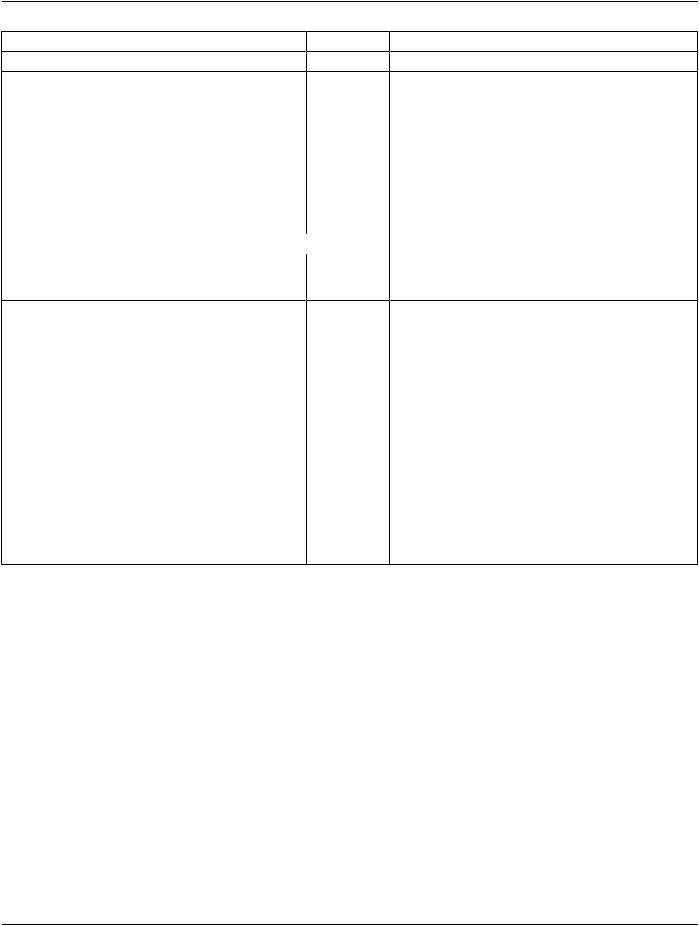
5.Intersection of overlapping volumes 1 and 2
Input Result
Prostate
PTV
(2) |
Rectum |
|
(1) |
Figure 10.34.1.1-5. Conceptual Volume Example of Intersection of Non-disjunct Volumes
Conceptual Volume Combination Expression (3010,000C): (INTERSECTION 1 2)
Items in Conceptual Volume Constituent Sequence (3010,0008):
Table 10.34.1.1-5. Conceptual Volume Example of Intersection of non-disjunct Volumes
Conceptual Volume Constituent Index (3010,000D) |
Conceptual Volume |
1 |
Rectum |
2 |
Prostate PTV |
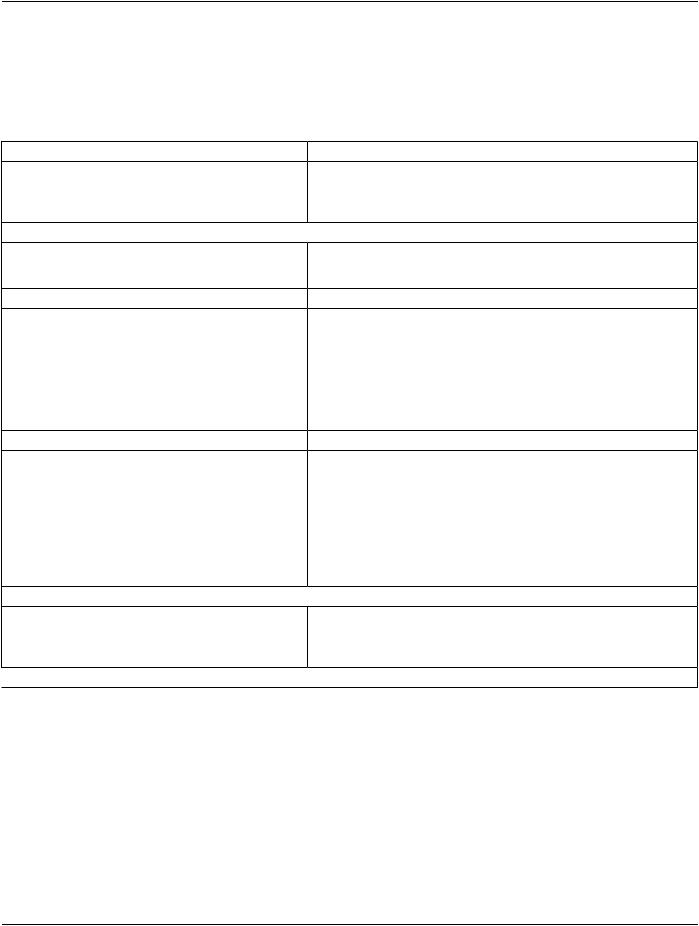
6.Intersection of disjoint volumes 1 and 2 |
|
Input |
Result |
Bladder
(1)
Prostate
(2)
Figure 10.34.1.1-6. Conceptual Volume Example of Intersection of Non-disjunct Volumes
Conceptual Volume Combination Expression (3010,000C):
- Standard -

DICOM PS3.3 2020a - Information Object Definitions |
Page 193 |
(INTERSECTION 1 2)
Items in Conceptual Volume Constituent Sequence (3010,0008):
Table 10.34.1.1-6. Conceptual Volume Example of Intersection of disjunct Volumes
Conceptual Volume Constituent Index (3010,000D) |
Conceptual Volume |
1 |
Bladder |
2 |
Prostate |
10.34.1.2 Conceptual Volume Segmentation Reference Sequence
The Conceptual Volume Constituent Segmentation Reference Sequence (3010,0012) contains a reference to a segmentation which represents the volume of this consituent geometrically. The referenced segmentations of the constituents of a combined Conceptual Volume may be in one or more Frames of References.
The Conceptual Volume constituents shall not include the combined Conceptual Volume being defined. Applications that wish to combine existing segmentations within the same Conceptual Volume must create a new Segmentation Instance.
10.34.1.3 Referenced Direct Segment Instance Sequence
A SOP Instance may only be referenced in this Sequence if it belongs to a SOP Class that includes the Section C.36.9 Segment Reference Module.
10.34.1.4 Conceptual Volume Segmentation Reference Sequence
The Conceptual Volume Segmentation Reference Sequence (3010,0011) contains a reference to a segmentation which represents this volume geometrically.
10.35 Device Model Macro
TheDeviceModelMacrocontainsgeneralAttributesneededtospecifyadevicemodelotherthanthedevicecreatingtheSOPInstance.
Table 10.35-1. Device Model Macro Attributes
Attribute Name |
Tag |
Type |
Attribute Description |
Manufacturer |
(0008,0070) |
2 |
Manufacturer of the device. |
Manufacturer's Model Name |
(0008,1090) |
2 |
Manufacturer's model name of the device. |
Manufacturer's Model Version |
(3010,001A) |
2 |
A version number of the Manufacturer's model of |
|
|
|
the device. |
10.36 Device Identification Macro
The Device Identification Macro identifies a (physical or virtual) device.
Table 10.36-1. Device Identification Macro Attributes
Attribute Name |
Tag |
Type |
Attribute Description |
Device Type Code Sequence |
(3010,002E) |
1 |
The type of the device. |
|
|
|
Only a single Item shall be included in this Sequence. |
>Include Table 8.8-1 “Code Sequence Macro Attributes” |
|
CID may be defined in the Macro invocation. |
|
Device Label |
(3010,002D) |
1 |
User-defined label for this device. |
Long Device Description |
(0050,0021) |
3 |
User-defined description for this device. |
- Standard -
Page 194 |
DICOM PS3.3 2020a - Information Object Definitions |
||
Attribute Name |
Tag |
Type |
Attribute Description |
Device Serial Number |
(0018,1000) |
2 |
Manufacturer"s serial number of the device. |
Software Versions |
(0018,1020) |
2 |
Manufacturer's designation of software version of the |
|
|
|
equipment. |
UDI Sequence |
(0018,100A) |
3 |
Unique Device Identifier (UDI) of the device. |
|
|
|
One or more Items are permitted in this Sequence. |
|
|
|
Note |
|
|
|
Multiple Items may be present if the entire |
|
|
|
equipment has UDIs issued by different Issuing |
|
|
|
Authorities. |
>Include Table 10.29-1 “UDI Macro Attributes” |
|
|
|
Manufacturer's Device Identifier |
(3010,0043) |
2 |
An identifier issued by the manufacturer. |
|
|
|
See Note. |
Device Alternate Identifier |
(3010,001B) |
2 |
An identifier intended to be read by a device such as a bar |
|
|
|
code reader. |
Device Alternate Identifier Type |
(3010,001C) |
1C |
Defines the type of Device Alternate Identifier. |
|
|
|
Required if Device Alternate Identifier (3010,001B) is |
|
|
|
present. |
|
|
|
Defined Terms: |
|
|
|
BARCODE |
|
|
|
RFID |
Device Alternate Identifier |
(3010,001D) |
1C |
Description of the format in which the Device Alternate |
Format |
|
|
Identifier (3010,001B) is issued. |
Required if Device Alternate Identifier (3010,001B) is present.
See Section 10.36.1.1.
Note
Typically, the Device Identifier is a code which can be electronically read by the machine utilizing that device, e.g. to verify the presence of that device.
10.36.1 Device Component Identification Macro Attribute Descriptions
10.36.1.1 Device Alternate Identifier Format
The Device Alternate Identifier Format (3010,001D) specifies the format of the value of the Device Alternate Identifier (3010,001B).
If the value of Device Alternate Identifier Type (3010,001C) is RFID, a big variety of RFID formats exists (some examples are DOD- 96, DOD-64 UID, GID-96, sgtin-96). Supported format values shall be defined in the Conformance Statement.
For Device Alternate Identifier Type (3010,001C) = BARCODE, see Section C.22.1.1.
10.37 Related Information Entities Macro
This Macro defines references to entities and their purpose of reference. References can be made at the Study level, Series level, Instance level or Frame Level.
- Standard -
DICOM PS3.3 2020a - Information Object Definitions |
Page 195 |
The attributes Pertinent SOP Classes in Study (3010,0052) and Pertinent SOP Classes in Series (3010,0053) allow the specification of the relevant SOP Classes for the given purpose. These attributes support filtering for certain SOP Classes, specification of corres- ponding query keys, and allowing the receiving application to assess its capabilities to handle the specified objects.
All referenced Studies, Series and Instances share the same single Purpose of Reference.
Table 10.37-1. Related Information Entities Macro Attributes
Attribute Name |
Tag |
Type |
Attribute Description |
Purpose of Reference Code |
(0040,A170) |
1 |
Describes the purpose for which the references are |
Sequence |
|
|
made. |
|
|
|
Only a single Item shall be included in this Sequence. |
>Include Table 8.8-1 “Code Sequence Macro Attributes” |
|
CID may be defined in the Macro invocation. |
|
Referenced Study Sequence |
(0008,1110) |
1 |
Studies which are relevant for the invocation context. |
|
|
|
One or more Items shall be included in this Sequence. |
>Study Instance UID |
(0020,000D) |
1 |
Uniquely identifies the referenced Study. |
>PertinentSOPClassesinStudy |
(3010,0052) |
3 |
The SOP Classes in the Study which are relevant for the |
|
|
|
invocation context. |
|
|
|
If not present, all SOP Instances included in the |
|
|
|
referenced Study are considered relevant. |
>Referenced Series Sequence |
(0008,1115) |
3 |
Series which are relevant for the invocation context. |
|
|
|
One or more Items are permitted in this Sequence. |
>>Series Instance UID |
(0020,000E) |
1 |
Uniquely identifies the referenced Series. |
>>Pertinent SOP Classes in |
(3010,0053) |
3 |
The SOP Classes in the Series which are relevant for |
Series |
|
|
the invocation context. |
|
|
|
If not present, all SOP Instances included in the |
|
|
|
referenced Series are considered relevant. |
>>Referenced Image Sequence |
(0008,1140) |
3 |
ImageSOPInstanceswhicharerelevantintheinvocation |
|
|
|
context. |
|
|
|
One or more Items are permitted for this Sequence. |
>>>Include Table 10-3 “Image SOP Instance Reference Macro Attributes” |
|||
>>Referenced Instance |
(0008,114A) |
3 |
Non-Image SOP Instances which are relevant in the |
Sequence |
|
|
invocation context. |
One or more Items are permitted for this Sequence.
 >>>Include Table 10-11 “SOP Instance Reference Macro Attributes”
>>>Include Table 10-11 “SOP Instance Reference Macro Attributes”
10.38 Outline Definition Macro
The Outline Definition Macro describes a 2D outline in a given coordinate system.
- Standard -