Материал: part03

Page 116 |
DICOM PS3.3 2020a - Information Object Definitions |
Note
TheinformationcontentofthisentityrelatestothegeneralidentificationofaProcedureTyperatherthantoitsdecomposition into the protocol(s) required to perform a specific instance of a Requested Procedure for a particular Patient.
7.3.1.5 Requested Procedure
A Requested Procedure is an instance of a Procedure of a given Procedure Type. An instance of a Requested Procedure includes all of the items of information that are specified by an instance of a Procedure Plan that is selected for the Requested Procedure by the imaging service provider. This Procedure Plan is defined by the imaging service provider on the basis of the Procedure Plan templates associated with the considered Procedure Type. An Imaging Service Request may include requests for several different Requested Procedures. The purpose of this entity is to establish the association between Imaging Service Requests and Procedure Types, to convey the information that belongs to this association and to establish the relationships between Requested Procedures and the other entities that are needed to describe them. A single Requested Procedure of one Procedure Type is the smallest unit of service that can be requested, reported, coded and billed. Performance of one instance of a Requested Procedure is specified by exactlyoneProcedurePlan.ARequestedProcedureleadstooneormoreScheduledProcedureStepsinvolvingProtocolsasspecified by a Procedure Plan. A Requested Procedure may be associated with one or more Visits. A Requested Procedure may involve one or more pieces of equipment.
7.3.1.6 Scheduled Procedure Step
A Modality Scheduled Procedure Step is an arbitrarily defined scheduled unit of service, that is specified by the Procedure Plan for aRequestedProcedure.AModalityScheduledProcedureStepprescribesaProtocol,whichmaybeidentifiedbyoneormoreprotocol codes. A Modality Scheduled Procedure Step involves equipment (e.g., imaging Modality equipment, anesthesia equipment, surgical equipment,transportationequipment),humanresources,consumablesupplies,location,andtime(e.g.,starttime,stoptime,duration). While in the context of imaging services the scheduling of a Modality Scheduled Procedure Step might include only a general desig- nation of imaging Modality that could be satisfied by multiple pieces of the same equipment type, the performance of one instance of a Modality Scheduled Procedure Step involves one and only one piece of imaging Modality equipment.
The performance of a Modality Scheduled Procedure Step may result in the creation of zero or more Modality Performed Procedure Step instances.
Note
1.The Procedure Step entity is provided to support management of the logistical aspects of procedures (e.g., materials management, human resources, scheduling). The full definition of the contents of Procedure Steps and protocols ac- cording to which they are performed is implementation dependent and is beyond the scope of this Standard.
2.A Modality Scheduled Procedure Step may contribute to more than one Requested Procedure (e.g., a Modality ScheduledProcedureSteprequiringanintravenousiodinecontrastinjectionmightbesharedbyanintravenouspyelogram and a CT examination). However, for billing purposes an instance of a Modality Scheduled Procedure Step is typically considered to be a part of only one Requested Procedure.
7.3.1.7 Procedure Plan
A Procedure Plan is a specification that defines the set of Protocols that must be done in order to perform the Scheduled Procedure StepsofaRequestedProcedure.EachScheduledProcedureStepisperformedaccordingtoasingleProtocol,whichmaybeidentified by one or more Protocol Codes and may be described in a Defined Procedure Protocol. The Protocols actually performed during a Procedure Step may be recorded in a Performed Procedure Protocol and may differ from those prescribed in the related Procedure Plan. Audit of actually performed Protocols versus the prescribed Procedure Plan is an important element of quality control.
7.3.1.8 Protocol
A Protocol is a specification of actions prescribed by a Procedure Plan to perform a specific Procedure Step. A Scheduled Procedure Step contains only one Protocol, which may be conveyed by one or more Protocol Codes.
A Protocol may be specified by a Defined Procedure Protocol to be used on any appropriate Patient.
A Protocol can be documented, once a Procedure Step has been performed, in a Performed Procedure Protocol.
- Standard -

DICOM PS3.3 2020a - Information Object Definitions |
Page 117 |
7.3.1.8.1 Defined Procedure Protocol
A Defined Procedure Protocol describes a set of parameters and associated details for the prescribed action. The Defined Procedure Protocolmayprovidespecificvaluesforrelevantparameters,ormayprovideconstraintsonthoseparameters(suchasanacceptable range) to guide the choice of specific values.
Defined Procedure Protocol is not associated with any particular Patient or Scheduled Procedure Step. A Defined Procedure Protocol may contain parameters specific to a particular model or version of device, or it may be generic in that it only describes parameters common to multiple device models.
A Defined Procedure Protocol may include information such as the clinical purpose, indications, and appropriate device models, in- tended for selection and management.
7.3.1.8.2 Performed Procedure Protocol
A Performed Procedure Protocol encodes the parameter values used. A Performed Procedure Protocol is always associated with a specific Patient and Performed Procedure Step. The Performed Procedure Protocol may reference the Defined Procedure Protocol on which it was based, but does not otherwise record the orginal constraints and whether or not they were satisfied by the final values as recorded in the Performed Procedure Protocol.
7.3.1.9 Modality Performed Procedure Step
A Performed Procedure Step is an arbitrarily defined unit of service that has actually been performed (not just scheduled). Logically itcorrespondstoaScheduledProcedureStep,butreal-worldconditionsmaydictatethatwhatisactuallyperformeddoesnotcorrespond exactly with what was requested or scheduled.
Note
Forexample,twoormoreScheduledProcedureSteps,RequestedProceduresorImagingServiceRequestsmayhavebeen generated by different Referring Physicians but may be satisfied be a single Performed Procedure Step at the discretion of a Performing Physician or Operator. Alternatively, a single Scheduled Procedure Step may need to be satisfied by multiple Performed Procedure Steps on different types or instances of equipment, due to clinical need or failure conditions, or over extended periods of time.
Itcontainsinformationdescribingthetypeofprocedureactuallyperformed.ThisinformationisrepresentedbythePerformedProtocol that may be defined by one or more Protocol Codes.
A Requested Procedure results in the creation of zero or more Performed Procedure Steps.
A Scheduled Procedure Step results in the creation of zero or more Performed Procedure Steps.
The Performed Procedure Step contains information about its state (e.g., in progress, discontinued or completed).
A Modality Performed Procedure Step is a Performed Procedure Step that results from activity (such as the acquisition of images from a Patient or other Imaging Subject) on a Modality.
It contains information describing the performance of a step of an imaging procedure, including data about the performance of the procedure itself, and data for billing and material management.
The Modality Performed Procedure Step contains references to zero or more Series of Images and other Composite SOP Instances that may be created as part of the procedure step. A particular Series is part of only one Modality Performed Procedure Step.
The purpose of the Modality Performed Procedure Step is to report what was performed; it does not imply any storage semantics. While the MPPS represents a unit of service within a workflow, the specification of the workflow itself is beyond the scope of the Standard, and the MPPS does not identify or control any subsequent activities to be performed.
Note
1.For example, a modality may create both "for processing" images for automated analysis and "for presentation" images for human review from the same acquisition. The Standard does not specify whether the production of these is a single unit of service, or two. A single Modality Performed Procedure Step instance could list both the "for processing" images and the "for presentation" images, regardless of whether or not both sets of images were stored to the same or different
- Standard -

Page 118 |
DICOM PS3.3 2020a - Information Object Definitions |
AEs, or indeed were stored at all, since the MPPS is independent of the storage semantics. Alternatively, the modality may treat these two sets of images as two separate units of service, and send two separate MPPS Instances.
A Radiation Dose SR from the irradiation events of an acquisition could be referenced in the same MPPS Instance as that of the acquired images, again irrespective of where such a Radiation Dose Structured Report might be transmitted, if at all. Alternatively, the modality may treat the production of the Radiation Dose SR as a separate unit of service, and report it in a distinct MPPS.
Another example is the case of thin and thick slice CT images acquired from the same acquisition (raw) data. When the reconstruction of both sets of images is prospectively defined and automatically initiated by the protocol selection, then both sets might be referenced from a single MPPS Instance. However, if the reconstruction of one or the other set is performed retrospectively by manual intervention some time after the acquisition MPPS had been completed, the sub- sequent instances will necessarily be referenced in a new MPPS Instance, since the acquisition MPPS cannot be modified once completed.
2.ThecompletionofanMPPSmaybeasignificanteventthattriggersorenablesdownstreamactivity,butitisnottheintent to require the modality to be configured to "manage" such activity. The "units of service" that the modality describes in an MPPS, and how the modality relates those Performed Procedure Steps to Scheduled Procedure Steps, are imple- mentation decisions beyond the scope of the Standard. The IHE Radiology Scheduled Workflow Profile provides addi- tional guidance for implementation.
3.An MPPS may describe instances that were acquired but that have not been, nor may ever be, stored. For example, a modality may be capable of storing a CT acquisition as multiple single-frame CT Image Storage SOP Instances, as a singlemulti-frameEnhancedCTImageStorageSOPInstance,orasseveralEnhancedCTImageStorageSOPInstances that together comprise a Concatenation. An MPPS may describe all three possibilities, even though only one choice may ultimately be stored, perhaps depending on the negotiated capabilities of the storage recipient. Alternatively, sep- arate MPPS instances could be used for different storage SOP Classes.
4.The MPPS contains only the instances that the modality created, not instances converted and created subsequently in response to a query (e.g., during legacy conversion).
5.The MPPS is not a substitute for, nor is equivalent to, a Storage Commitment request, nor an Instance Availability Noti- fication.
7.3.1.10 General Purpose Scheduled Procedure Step (Retired)
Retired. See PS3.3-2011.
7.3.1.11 General Purpose Performed Procedure Step (Retired)
Retired. See PS3.3-2011.
7.3.1.12 Workitem (Retired)
Retired. See PS3.3-2011.
7.3.1.13 Clinical Document
A Clinical Document is a part of the medical record of a Patient. A Clinical Document is a documentation of clinical observations and services and has the following characteristics:
•Persistence - A clinical document continues to exist in an unaltered state, for a time period defined by local and regulatory require- ments.
•Stewardship - A clinical document is maintained by an organization entrusted with its care.
•Potential for authentication - A clinical document is an assemblage of information that is intended to be legally authenticated.
•Context - A clinical document establishes the default context for its contents.
- Standard -

DICOM PS3.3 2020a - Information Object Definitions |
Page 119 |
•Wholeness - Authentication of a clinical document applies to the whole and does not apply to portions of the document without the full context of the document.
•Human readability - A clinical document is human readable.
Note
This definition is from ANSI/HL7 CDA R1.0-2000, and HL7 v3 CDA R2-2005.
ClinicalDocumentsmayprovidesignificantcontextfortheperformanceofimagingandrelatedprocedures,e.g.,patientclinicalhistory, pre-imaging-procedure lab test results, or patient advance medical directives.
Clinical Documents may be associated with Service Episodes, Service Requests, Requested Procedures, or other entities subsidiary to the Patient in the Real-World Model. Such associations are not explicitly modeled for the purposes of the Modality-IS context.
Clinical Documents are one sub-class of the class of healthcare Structured Documents; Structured Documents, in general, are not necessarily related to a Patient. Structured Documents may be used for imaging procedure operational instructions, e.g., in product labeling, Procedure Plans, or patient care plans.
Note
1.The format and semantics of Structured Documents, including Clinical Documents, are defined outside the scope of the DICOM Standard (e.g., by HL7). DICOM provides the means to reference Structured Documents within the Modality- IS context.
2.The general class of Structured Documents is not modeled in the Real-World Model; only specific sub-classes, e.g., Clinical Documents, are modeled.
7.4 Extension of the DICOM Model of the Real World for the General Purpose Worklist (Retired)
Retired. See PS3.3-2011.
7.5 Organizing Large Sets of Information
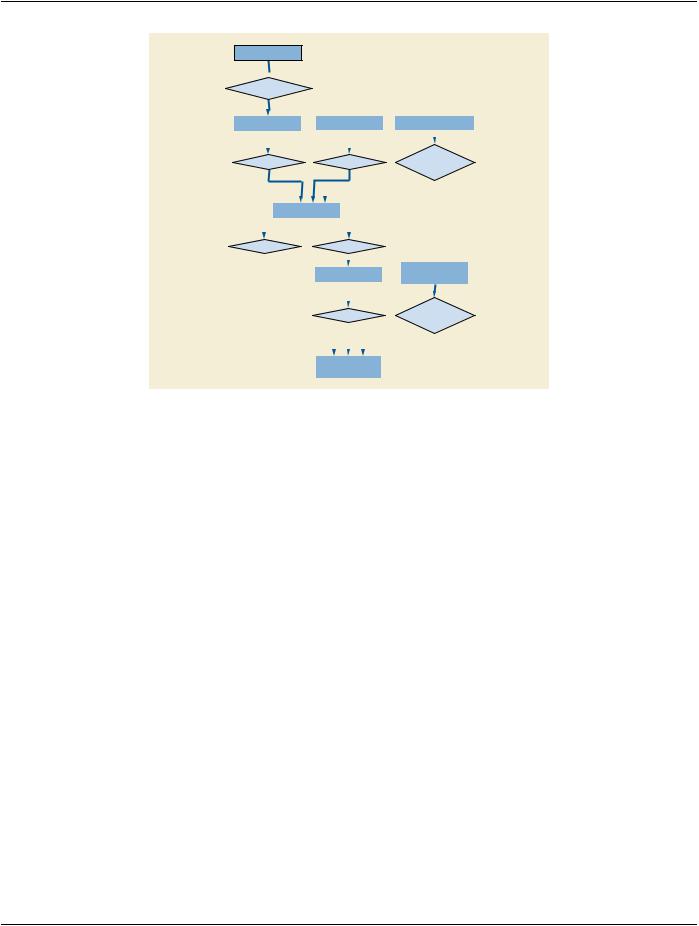
For the purpose of accommodating large sets of frames in Multi-frame Image SOP Instances the Real-World Entity Relationship Diagram has been extended to describe the relationships of these instances: Concatenation (see Section 7.5.1) and Dimension Or- ganization (see Section 7.5.2). Figure 7.5-1 depicts the additions to Figure 7-1a.
- Standard -
Page 120 |
DICOM PS3.3 2020a - Information Object Definitions |
Patient
 1 is the subject of
1 is the subject of
|
|
|
1-n |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||
|
|
|
|
|
|
|
|
|
|
||||||||||||
Study |
|
|
|
|
|
Equipment |
|
Frame of Reference |
|||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
1 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||
1 |
|
|
|
1 |
|
|
spatially |
||||||||||||||
|
|
|
|
|
|
|
|||||||||||||||
contains |
|
|
|
creates |
|
|
or temporally |
|
|
||||||||||||
|
|
|
1-n |
|
|
|
|
|
|
|
|
1-n |
|
|
defines |
0-n |
|||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Series |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||
|
|
1 |
|
|
|
|
|
|
1 |
|
|
|
|
|
|
||||||
|
|
|
|
|
|
|
|
|
|
||||||||||||
contains |
|
|
|
contains |
|
|
|
|
|
|
|||||||||||
|
|
|
0-n |
|
|
|
|
|
|
|
|
0-n |
|
|
|
|
|
|
|||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||||||||
|
|
|
|
|
|
|
Concatenation |
|
|
Dimensional |
|
|
|||||||||
|
|
|
|
|
|
|
|
|
Organization |
|
|
||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
1 |
|
|
|
|
|
|
|
|
1 |
|
|
Specifies |
|||||||||||
|
|
|
|
|
|
|
|
|
|
|
|||||||||||
|
|
|
|
|
|
|
|
|
|
contains |
|
|
|
|
|||||||
|
|
|
|
|
|
|
|
|
|
|
|
organization of |
|
|
|||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
1-n |
|
|
|
|
||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
1-n |
||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||
|
|
|
|
|
|
|
|
|
|
Image |
|
|
|
|
|
|
|||||
|
|
|
|
|
|
|
|
|
(Multi-frame) |
|
|
|
|
|
|
||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Figure 7.5-1. Extension of the Real World Model with Concatenations and Dimensions
7.5.1 Concatenation
For implementation specific reasons (such as practical limits on the maximum size of an individual SOP Instance) the content of a multi-frame image may need to be split into more than one SOP Instance. These SOP Instances together form a Concatenation, which is a group of SOP Instances within a Series that is uniquely identified by Concatenation UID (0020,9161).
7.5.2 Dimension Organization
The Dimension Organization contains a set of dimensions. A dimension is a set of Attributes that change on a per-frame basis in a manner that is known before the image is acquired, are defined by the generating application and are especially intended for presentation. Other Attributes may also change on a per-frame basis but if they are not present in the Dimension Organization, they are not considered significant as a dimension for organizational purposes.
Receiving applications shall use the order of dimensions for guidance when presenting images if the Multi-frame Dimension Module is present. The first Item of the Dimension Index Sequence shall be the slowest varying index.
Note
See Section C.7.6.17 for an example.
7.6 Extension of the DICOM Model of the Real World for Clinical Trials and Re- search
TheDICOMModeloftheRealWorldisextendedforClinicalTrialsandresearchwiththeadditionofseveralobjectswhoserelationships to each other and existing DICOM Real World objects are shown in Figure 7.6-1.
Attributes of the Clinical Trial Sponsor, Clinical Trial Protocol, Clinical Trial Subject, and Clinical Trial Site objects are represented in the Clinical Trial Subject Module within the Patient IOD. Attributes of the Clinical Trial Time Point object are represented in the Clinical Trial Study Module within the Study IOD. The Clinical Trial Coordinating Center Attribute is represented in the Clinical Trial Series Module within Image IODs.
- Standard -