Page 762 |
DICOM PS3.3 2020a - Information Object Definitions |
φt
Z t
Z’t
 Y’t
Y’t
Xt - Yt Plane
 X t
X t
X’t φt
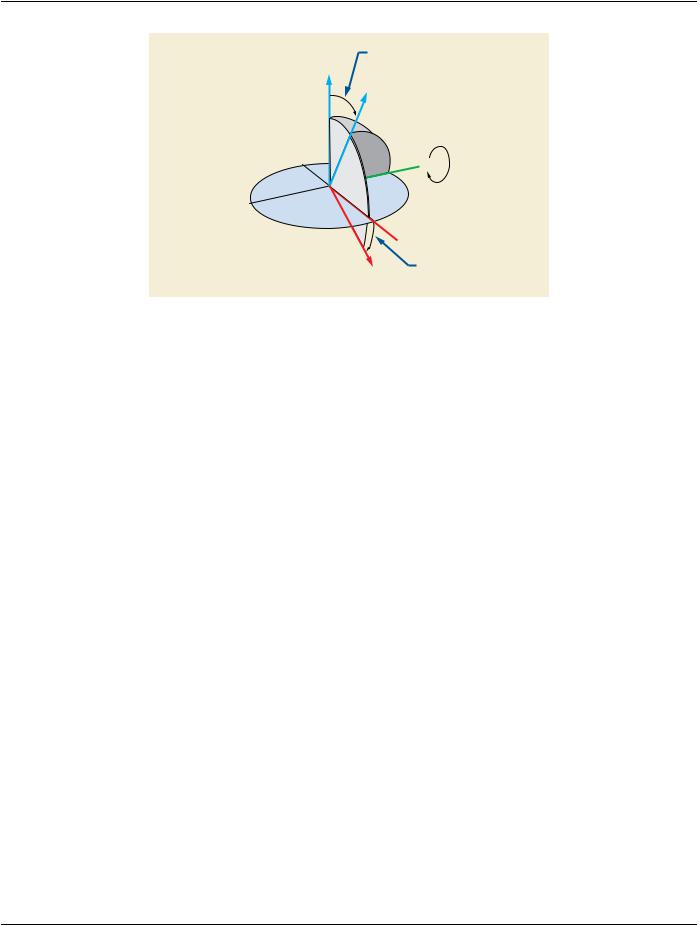
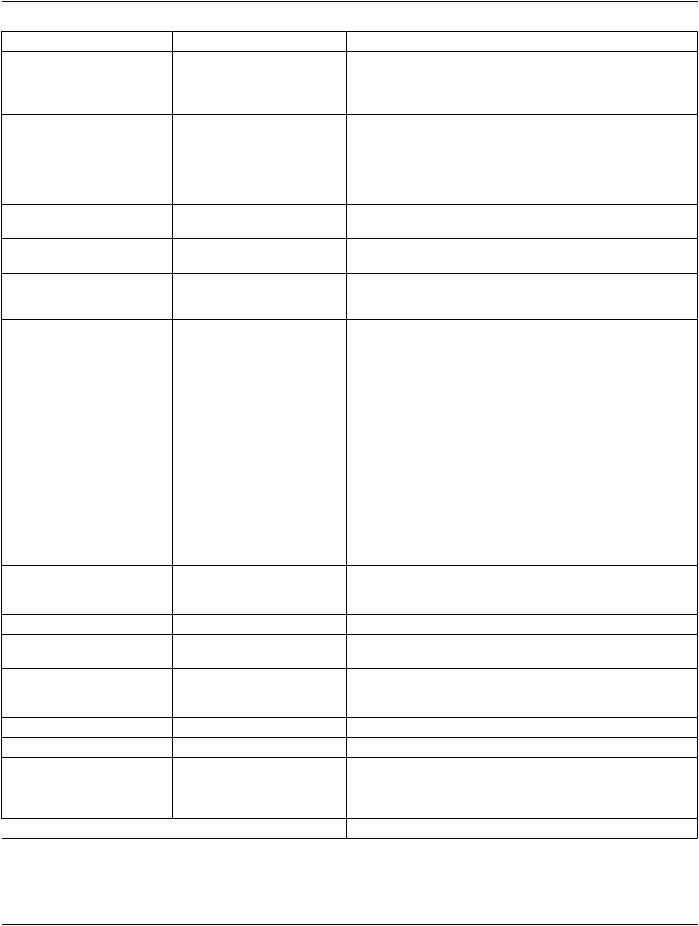
Figure C.8.8.14-2. Table Top Roll Angle
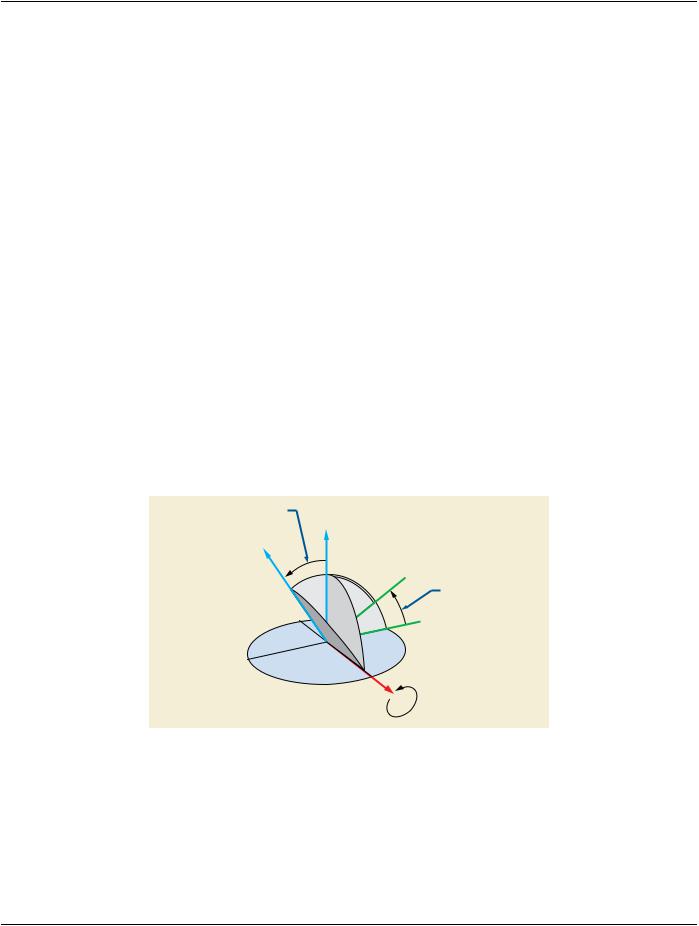
C.8.8.14.13 Angular Values in RT Beams Module
TheAttributesthatdefineanglesrefertocoordinatesystemsdefinedbyIEC61217.WhereindicatedintheDICOMAttributedefinition, the angle uses the coordinate system (orientation of the rotation axis and the origin of the rotating coordinate system) defined in IEC 61217, however DICOM makes no restrictions on the range of values stored. IEC 61217 defines restrictions that only apply to user interface presentation.
C.8.8.14.14 Effective Wedge Angle
The Effective Wedge Angle (300A,00DE) and Radiation Beam Effective Wedge Angle (300A,0654) describe the dosimetric angle of a motorized wedge accounting for the partial presence of the wedge in the beam. The presence of the wedge in the beam is either specified by the Wedge Position (300A,0118) in the Wedge Position Sequence (300A,0116) included in the Control Point Sequence (300A,0111) of the current beam or the RT Control Point Sequence of the current Radiation. When the wedge is in the beam throughout all control points, the Effective Wedge Angle (300A,00DE) and Radiation Beam Effective Wedge Angle (300A,0654) will havethesamevalueastheWedgeAngle(300A,00D5)orRadiationBeamWedgeAngle(300A,0652).OtherwisetheEffectiveWedge Angle(300A,00DE)orRadiationBeamEffectiveWedgeAngle(300A,0654)willhavealowervaluethantheWedgeAngle(300A,00D5) or Radiation Beam Wedge Angle (300A,0652).
C.8.8.14.15 Source to External Contour Distance and External Contour Entry Point
The Source to External Contour Distance (300A,0132) is the distance to the beam entry point (External Contour Entry Point), which may include Bolus, Patient Positioning Devices, Patient Immobilization Devices or other devices. This value is useful for including the attenuation effects of external devices on the dose calculation and for patient setup.
C.8.8.14.16 Referenced Control Point
The number of Items in the Beam Dose Verification Control Point Sequence (300A,008C) is not required to be the same as in the Control Point Sequence (300A,0111). A different sampling can be chosen for the Beam Dose Verification Control Point Sequence, but where the Cumulative Meterset Weight of a Control Point Sequence (300A,008C) Item is the same it shall be referenced by the Referenced Control Point Index (300C,00F0).
C.8.8.15 RT Brachy Application Setups Module
The RT Brachy Application Setups Module describes the application of a brachytherapy radiotherapy treatment. It contains one or moresources,eachassociatedwithoneormoreChannels.AChannelisadevicebywhichasourceisplacedinitsintendedtreatment position or positions. A Channel may consist of a Source Applicator plus a Transfer Tube, a Source Applicator alone, a rigid or flexible linear source, or a seed. A number of Channels (for example applicators, sources or seeds) are generally arranged in an Application Setup, which may be considered a "logical" device. It is important not to confuse Application Setup with Applicator. The model used