DICOM PS3.3 2020a - Information Object Definitions |
Page 771 |
Control Point 0: Control Point Relative Position = 30, Cumulative Time Weight = 0
Control Point 1: Control Point Relative Position = 30, Cumulative Time Weight = 25
Control Point 2: Control Point Relative Position = 20, Cumulative Time Weight = 27
Control Point 3: Control Point Relative Position = 20, Cumulative Time Weight = 52
Control Point 4: Control Point Relative Position = 10, Cumulative Time Weight = 54
Control Point 5: Control Point Relative Position = 10, Cumulative Time Weight = 79
f) Stepwise motion with consideration of source transit times between dwell positions and to first and from last dwell position; Three equally weighted dwell positions; Step size = 10; Final Cumulative Time Weight = 383:
Control Point 0: Control Point Relative Position = 1200, Cumulative Time Weight = 0
Control Point 1: Control Point Relative Position = 30, Cumulative Time Weight = 150
Control Point 2: Control Point Relative Position = 30, Cumulative Time Weight = 175
Control Point 3: Control Point Relative Position = 20, Cumulative Time Weight = 177
Control Point 4: Control Point Relative Position = 20, Cumulative Time Weight = 202
Control Point 5: Control Point Relative Position = 10, Cumulative Time Weight = 204
Control Point 6: Control Point Relative Position = 10, Cumulative Time Weight = 229
Control Point 7: Control Point Relative Position = 1200, Cumulative Time Weight = 383
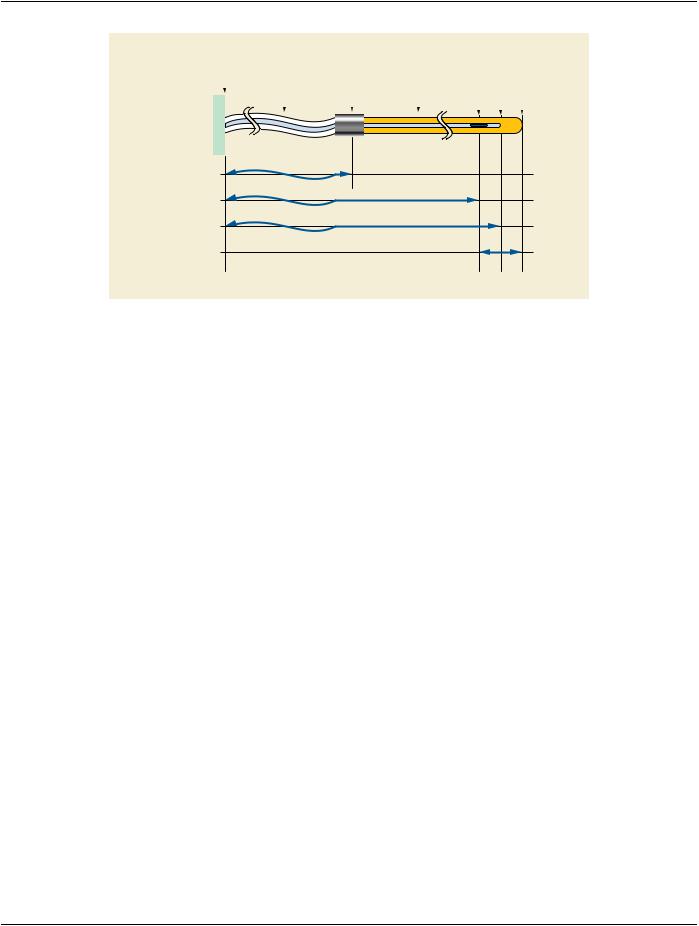
C.8.8.15.8 Source Transit Time
The Source transit times between dwell positions of a remote afterloader may be considered by specifying a non-zero increment in the Cumulative Time Weight (300A,02D6) when the Source moves between Control Points. In this case the Channel Total Time (300A,0286) shall include the overall Source transit time for the Channel.
C.8.8.15.9 Control Point Relative Position
Control Point Relative Position (300A,02D2) shall describe where the center of a given source in a channel is located with respect to the end of the channel, i.e. to the center of the distal-most possible dwell position in the channel.
C.8.8.15.10 Control Point 3D Position
Control Point 3D Position (300A,02D4) shall describe the absolute 3D coordinates of a source. This position shall correspond to the center of a source in an applicator during a remote or manually controlled afterloading treatment.
C.8.8.15.11 Cumulative Dose Reference Coefficient
The Cumulative Dose Reference Coefficient (300A,010C) is the value by which Brachy Application Setup Dose (300A,00A4) is mul- tiplied to obtain the dose to the referenced dose reference site at the current control point (and after previous control points have been successfully administered). The Cumulative Dose Reference Coefficient (300A,010C) is by definition zero for the initial control point. The Cumulative Dose Reference Coefficient (300A,010C) of the final control point multiplied by Brachy Application Setup Dose (300A,00A4)resultsinthefinaldosetothereferenceddosereferencesiteforthecurrentchannel.Dosecalculationfordosereference sites other than points is not well defined.
IfTreatmentType(300A,0202)isPDR,thentheCumulativeDoseReferenceCoefficient(3008,010C)shallspecifythedosedelivered to the dose reference during a single pulse. The total dose delivered to the dose reference shall then be expressed by Cumulative Dose Reference Coefficient (3008,010C) multiplied by Number of Pulses (300A,028A) multiplied by Brachy Application Setup Dose (300A,00A4).