Page 428 |
DICOM PS3.3 2020a - Information Object Definitions |
A.85.3.4.3 Modality
Enumerated Values:
M3D
A.86 RT Second Generation
A.86.1 RT Second Generation Objects
This section provides a brief description of the IODs of RT Second Generation. Specifically, this description includes:
•The Real-World Object which is represented by the IOD.
•Information as to the scope of the represented object, if appropriate.
A.86.1.1 RT Second Generation Common Information
This section provides a description of the Module structure which is shared by the RT Second Generation IODs.
A.86.1.1.1 RT Second Generation Entity-Relationship Model
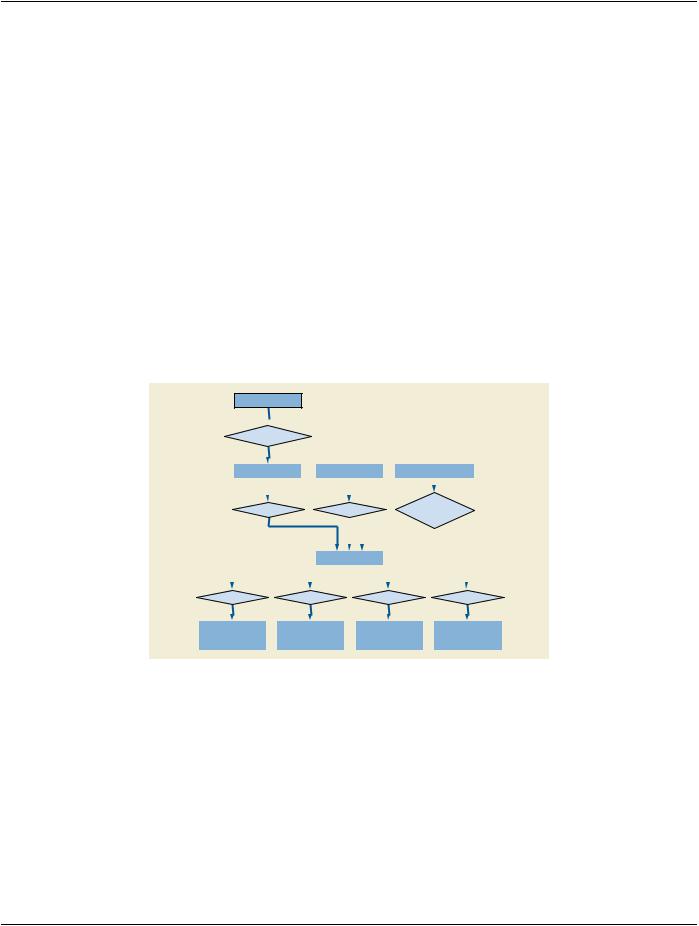
The E-R Model in Figure A.86.1.1.1-1 depicts those components of the DICOM Information Model that are relevant to RT Second Generation IODs.
Patient
 1 is the subject of
1 is the subject of
|
|
|
|
1-n |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Study |
|
|
|
Equipment |
|
|
Frame of Reference |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
1 |
|
|
|
|
|
|
1 |
|
|
|
|
|
0-1 |
|
|
|
|
|
|
|
|
|
|
|
|
|
spatially |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
contains |
|
|
|
creates |
|
|
|
or temporally |
|
|
|
|
1-n |
|
|
1-n |
|
|
|
defines |
0-n |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Series |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
1 |
|
|
|
1 |
|
|
|
|
|
1 |
|
|
|
1 |
contains |
contains |
contains |
|
|
contains |
|
0-n |
|
|
|
0-n |
|
|
|
0-n |
|
|
|
|
0-n |
|
|
|
|
|
|
|
|
|
|
RT Physician |
|
|
RT Segment |
|
|
|
RT Radiation |
|
RT Radiation |
Intent |
|
|
Annotation |
|
|
|
Set |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Figure A.86.1.1.1-1. RT Second Generation IOD Information Model
A.86.1.1.1.1 Use of Study and Series in RT Second Generation Radiotherapy
For first generation IODs, no specific semantics are attached to a Study or a Series in RT. Similarly, for RT Second Generation IODs, internalreferencesshallbeusedtorelateandlocateSOPInstancesratherthanmakingassumptionsabouthowrelatedSOPInstances are grouped into Studies or Series. For practical reasons it may be indicated to create a new Study separate from imaging Studies that are used for radiotherapeutic planning because of billing or reimbursement for Series that contain RT instances.
ImplementersshouldalsonotethattheDICOMStandard,ingeneral,doesplacesomerestrictionsonhowsuchSOPInstancesshould be grouped, as defined in Section A.1.2.3.
For non-image modalities like radiotherapy, the Series may not be the most efficient way to search for objects. Instead, an application might find it easier to use references in the RT Course object, Key Object Selection objects or Unified Worklist Procedure Steps to directly retrieve required instances rather than search for them.