Материал: part03
|
DICOM PS3.3 2020a - Information Object Definitions |
|
|
|
Page 221 |
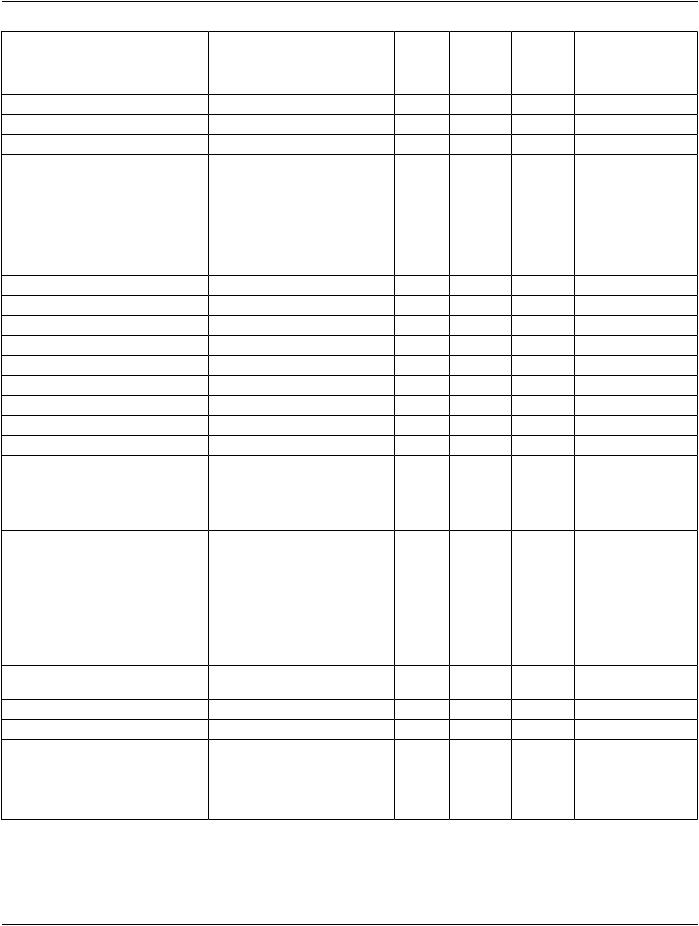
||||
IODs |
Gray PresCol Pres Pseudo |
Blend |
XA RF |
Planar Vol Rend |
Adv |
||||
Modules |
State |
State |
Col Pres Pres |
PresStateMPR Vol Vol Pres |
Blend |
||||
|
|
State |
State |
|
PresState State |
|
Pres |
||
|
|
|
|
|
|
|
|
|
State |
Overlay Plane |
C |
C |
C |
|
C |
|
|
|
|
Disp. Area |
M |
M |
M |
M |
M |
|
|
U |
|
Overlay Actvn. |
C |
C |
C |
|
C |
|
|
|
|
Graphic Annot. |
C |
C |
C |
C |
C |
U |
U |
U |
|
Spatial Trans. |
C |
C |
C |
C |
C |
|
|
C |
|
Graphic Layer |
C |
C |
C |
C |
C |
C |
C |
C |
|
Graphic Group |
U |
U |
U |
U |
|
U |
U |
U |
|
Modality LUT |
C |
|
C |
|
|
|
|
|
|
Softcopy VOI LUT |
C |
|
C |
|
C |
|
|
|
|
Softcopy Presn. LUT |
M |
|
|
|
M |
|
|
|
|
Presn. State Ident. |
M |
M |
M |
M |
M |
|
|
M |
|
Presn. State Reln. |
M |
M |
M |
|
M |
|
|
|
|
Presn. State Shutter |
M |
M |
M |
|
M |
|
|
|
|
Presn. State Mask |
M |
|
M |
|
|
|
|
|
|
Presn. State Blending |
|
|
|
M |
|
|
|
|
|
Adv. Presn. State Blending |
|
|
|
|
|
|
|
M |
|
Adv. Presn. State Display |
|
|
|
|
|
|
|
M |
|
XA/XRF Presn. State Mask |
|
|
|
|
C |
|
|
|
|
XA/XRF Presn. State Shutter |
|
|
|
|
C |
|
|
|
|
XA/XRF Presn. State Presn. |
|
|
|
|
C |
|
|
|
|
Volumetric Presentation State |
|
|
|
|
|
M |
M |
|
|
Identification |
|
|
|
|
|
|
|
|
|
Volumetric Presentation State |
|
|
|
|
|
M |
M |
|
|
Relationship |
|
|
|
|
|
|
|
|
|
Volume Cropping |
|
|
|
|
|
C |
C |
|
|
Presentation View Description |
|
|
|
|
|
M |
M |
|
|
Multi-PlanarReconstructionGeometry |
|
|
|
|
M |
|
|
|
|
Volume Render Geometry |
|
|
|
|
|
|
M |
|
|
Render Shading |
|
|
|
|
|
|
U |
|
|
MPR Volumetric Presentation State |
|
|
|
|
|
M |
|
|
|
Display |
|
|
|
|
|
|
|
|
|
Render Display |
|
|
|
|
|
|
M |
|
|
Volumetric Graphic Annotation |
|
|
|
|
|
U |
U |
|
|
Presentation Animation |
|
|
|
|
|
U |
U |
|
|
ICC Profile |
|
M |
M |
M |
|
|
|
M |
|
Common Instance Reference |
|
|
|
|
|
M |
M |
M |
|
SOP Common |
M |
M |
M |
M |
M |
M |
M |
M |
|
- Standard -
Page 222 DICOM PS3.3 2020a - Information Object Definitions
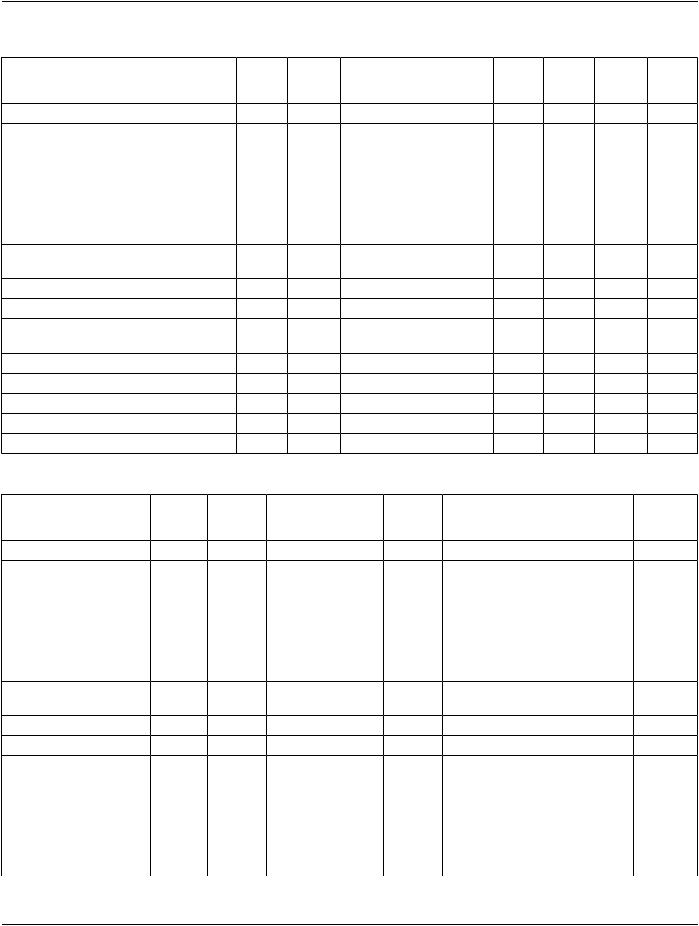
Table A.1-7. Composite Information Object Modules Overview - Structured Reports
IODs |
Basic Enhd Comp Comp ExtnsKey ObjMamChCAD Col |
Impl Prc Log |
|||||||||
Modules |
Text SR SR |
SR |
3D SR SR |
Sel |
CAD |
|
CAD Plan SR |
||||
|
|
|
|
|
|
|
|
|
|
|
|
Patient |
M |
M |
M |
M |
M |
M |
M |
M |
M |
M |
M |
Clinical Trial Subject |
U |
U |
U |
U |
U |
U |
U |
U |
U |
U |
U |
General Study |
M |
M |
M |
M |
M |
M |
M |
M |
M |
M |
M |
Patient Study |
U |
U |
U |
U |
U |
U |
U |
U |
U |
U |
U |
Clinical Trial Study |
U |
U |
U |
U |
U |
U |
U |
U |
U |
U |
U |
Clinical Trial Series |
U |
U |
U |
U |
U |
U |
U |
U |
U |
U |
U |
SR Document Series M |
M |
M |
M |
M |
|
M |
M |
M |
M |
M |
|
Key Object Document |
|
|
|
|
|
M |
|
|
|
|
|
Series |
|
|
|
|
|
|
|
|
|
|
|
Sync. |
|
U |
U |
U |
U |
|
|
U |
|
|
M |
General Equip. |
M |
M |
M |
M |
M |
M |
M |
M |
M |
M |
M |
Enhanced General |
|
|
|
|
M |
|
|
|
M |
M |
|
Equip. |
|
|
|
|
|
|
|
|
|
|
|
SR Document GeneralM |
M |
M |
M |
M |
|
M |
M |
M |
M |
M |
|
SR Document ContentM |
M |
M |
M |
M |
M |
M |
M |
M |
M |
M |
|
Key Object Document |
|
|
|
|
|
M |
|
|
|
|
|
Timezone |
|
|
|
|
|
|
|
|
|
|
|
SOP Common |
M |
M |
M |
M |
M |
M |
M |
M |
M |
M |
M |
Table A.1-7b. Composite Information Object Modules Overview - More Structured Reports
IODs |
RD SRR-RD SRPatRDSRSpRxRpMac GrdAcq CtxSimp Card Plan IA |
Perf IA |
|||||||
Modules |
|
|
|
|
Rp |
SR |
Echo SRAdmin SRAdmin SR |
||
|
|
|
|
|
|
|
|
|
|
Patient |
M |
M |
M |
M |
M |
M |
M |
M |
M |
Clinical Trial Subject |
U |
U |
U |
U |
U |
U |
U |
U |
U |
General Study |
M |
M |
M |
M |
M |
M |
M |
M |
M |
Patient Study |
U |
U |
U |
U |
U |
U |
U |
U |
U |
Clinical Trial Study |
U |
U |
U |
U |
U |
U |
U |
U |
U |
Clinical Trial Series |
U |
U |
U |
U |
U |
U |
U |
U |
U |
SR Document Series |
M |
M |
M |
M |
M |
M |
M |
M |
M |
Key Object Document |
|
|
|
|
|
|
|
|
|
Series |
|
|
|
|
|
|
|
|
|
Sync. |
C |
C |
C |
|
|
C |
C |
|
M |
General Equip. |
M |
M |
M |
M |
M |
M |
M |
M |
M |
Enhanced General Equip.M |
M |
M |
M |
M |
M |
M |
M |
M |
|
SR Document General |
M |
M |
M |
M |
M |
M |
M |
M |
M |
SR Document Content |
M |
M |
M |
M |
M |
M |
M |
M |
M |
Key Object Document |
|
|
|
|
|
|
|
|
|
Timezone |
|
|
|
|
|
|
M |
|
|
SOP Common |
M |
M |
M |
M |
M |
M |
M |
M |
M |
- Standard -
DICOM PS3.3 2020a - Information Object Definitions Page 223
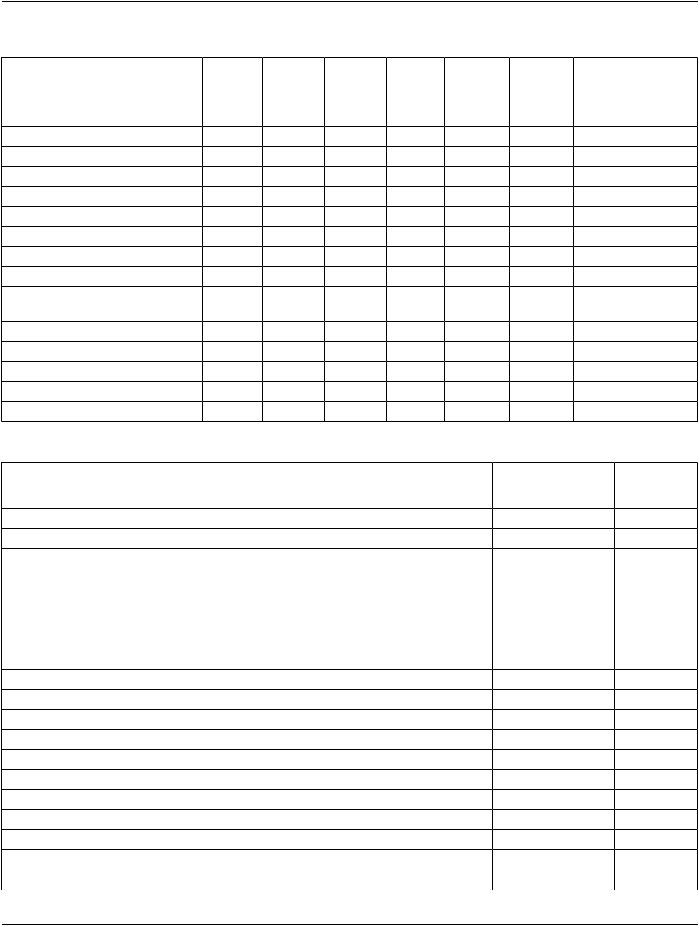
Table A.1-8. Composite Information Object Modules Overview - Waveforms
IODs |
Basic |
12 Lead General Ambul HemoWF Basic |
Arterial Resp WFGeneral |
||||||
Modules |
Voice |
ECG |
ECG WFECG WF |
Cardiac Pulse WF |
Audio WF |
||||
Audio |
|
|
|
|
EP WF |
|
|
|
|
Patient |
M |
M |
M |
M |
M |
M |
M |
M |
M |
Clinical Trial Subject |
U |
U |
U |
U |
U |
U |
U |
U |
U |
General Study |
M |
M |
M |
M |
M |
M |
M |
M |
M |
Patient Study |
U |
U |
U |
U |
U |
U |
U |
U |
U |
Clinical Trial Study |
U |
U |
U |
U |
U |
U |
U |
U |
U |
General Series |
M |
M |
M |
M |
M |
M |
U |
U |
U |
Clinical Trial Series |
U |
U |
U |
U |
U |
U |
U |
U |
U |
Synchronization |
U |
U |
U |
U |
C |
C |
M |
M |
M |
General Equipment |
M |
M |
M |
M |
M |
M |
M |
M |
M |
Enhanced General |
|
|
|
|
|
|
M |
M |
M |
Equipment |
|
|
|
|
|
|
|
|
|
Waveform IdentificationM |
M |
M |
M |
M |
M |
M |
M |
M |
|
Waveform |
M |
M |
M |
M |
M |
M |
M |
M |
M |
Waveform Annotation |
U |
C |
C |
C |
C |
C |
C |
C |
C |
Acquisition Context |
M |
M |
M |
U |
M |
M |
M |
M |
M |
SOP Common |
M |
M |
M |
M |
M |
M |
M |
M |
M |
Table A.1-9. Composite Information Object Modules Overview - Protocols
IODs |
CTPerformedProcedure CT Defined |
Protocol |
|
Modules |
Protocol |
Procedure Protocol Approval |
|
|
|
|
|
Patient |
M |
|
|
Clinical Trial Subject |
U |
|
|
General Study |
M |
|
|
Patient Study |
U |
|
|
Clinical Trial Study |
U |
|
|
General Series |
M |
|
|
Clinical Trial Series |
U |
|
|
Enhanced Series |
M |
|
|
CT Protocol Series |
M |
|
|
Frame of Reference |
M |
|
|
General Equipment |
M |
M |
M |
Enhanced General Equipment |
M |
M |
M |
Protocol Context |
M |
M |
|
Patient Protocol Context |
U |
|
|
Clinical Trial Context |
|
U |
|
Patient Specification |
|
U |
|
Equipment Specification |
|
M |
|
Instructions |
U |
U |
|
Patient Positioning |
U |
U |
|
- Standard -
Page 224 |
DICOM PS3.3 2020a - Information Object Definitions |
|
|
||||
IODs |
|
|
CTPerformedProcedure |
CT Defined |
Protocol |
||
Modules |
|
|
Protocol |
Procedure Protocol Approval |
|||
|
|
|
|
|
|
|
|
Defined CT Acquisition |
|
|
|
|
U |
|
|
Performed CT Acquisition |
|
|
U |
|
|
|
|
Defined CT Reconstruction |
|
|
|
|
U |
|
|
Performed CT Reconstruction |
|
|
U |
|
|
|
|
Defined Storage |
|
|
|
|
U |
|
|
Performed Storage |
|
|
U |
|
|
|
|
Protocol Approval |
|
|
|
|
|
|
M |
SOP Common |
|
|
M |
|
M |
M |
|
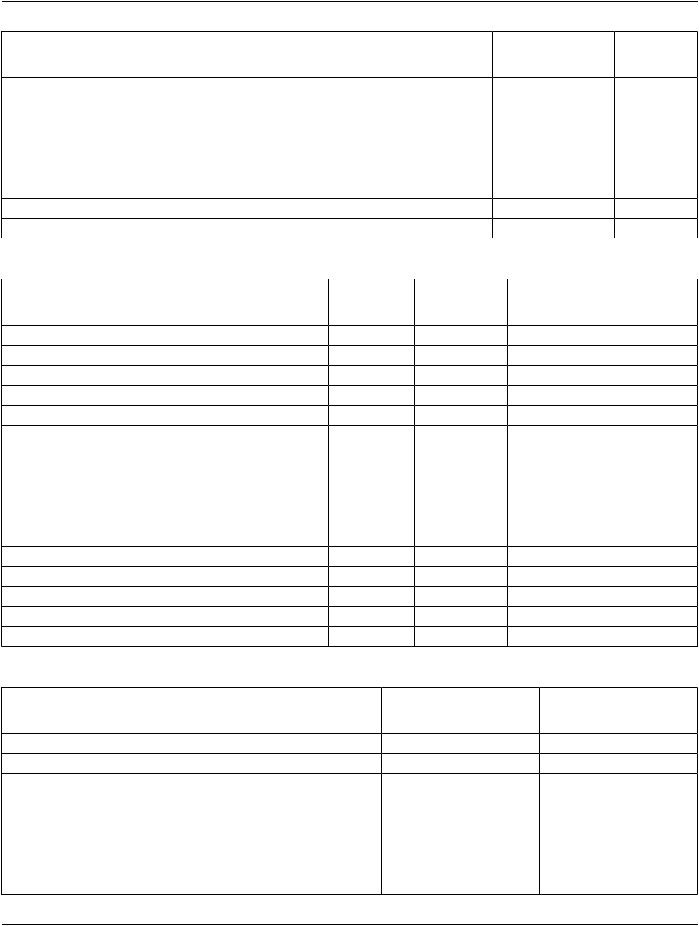
Table A.1-10. Composite Information Object Modules Overview - Encapsulated Documents |
|||||||
IODs |
|
Enc PDF |
Enc CDA |
Enc STL |
Enc OBJ |
Enc MTL |
|
Modules |
|
|
|
|
|
|
|
Patient |
|
M |
M |
M |
|
M |
M |
Clinical Trial Subject |
|
U |
U |
U |
|
U |
U |
General Study |
|
M |
M |
M |
|
M |
M |
Patient Study |
|
U |
U |
U |
|
U |
U |
Clinical Trial Study |
|
U |
U |
U |
|
U |
U |
Clinical Trial Series |
|
U |
U |
U |
|
U |
U |
Encapsulated Document Series |
M |
M |
M |
|
M |
M |
|
Frame of Reference |
|
|
|
M |
|
M |
|
General Equipment |
|
M |
M |
M |
|
M |
M |
Enhanced General Equipment |
|
|
|
M |
|
M |
M |
SC Equipment |
|
M |
M |
|
|
|
|
Encapsulated Document |
|
M |
M |
M |
|
M |
M |
Manufacturing 3D Model |
|
|
|
M |
|
M |
M |
ICC Profile |
|
|
|
U |
|
U |
U |
SOP Common |
|
M |
M |
|
|
M |
M |
Common Instance Reference |
|
|
|
C |
|
C |
C |
Table A.1-11. Real-Time Object Modules Overview - Images |
|
||||||
|
IODs |
|
|
RTV EN |
|
RTV PH |
|
Modules |
|
|
|
|
|
|
|
Patient |
|
|
M |
|
|
M |
|
Clinical Trial Subject |
|
|
U |
|
|
U |
|
General Study |
|
|
M |
|
|
M |
|
Patient Study |
|
|
U |
|
|
U |
|
Clinical Trial Study |
|
|
U |
|
|
U |
|
General Series |
|
|
M |
|
|
M |
|
Clinical Trial Series |
|
|
U |
|
|
U |
|
General Equipment |
|
|
M |
|
|
M |
|
- Standard -
DICOM PS3.3 2020a - Information Object Definitions |
Page 225 |
|
IODs |
RTV EN |
RTV PH |
Modules |
|
|
Enhanced General Equipment |
M |
M |
Frame of Reference |
C |
C |
Synchronization |
M |
M |
General Image |
M |
M |
General Reference |
U |
U |
Real-Time Bulk Data Flow |
M |
M |
Acquisition Context |
M |
M |
Device |
U |
U |
Specimen |
C |
C |
VL Image |
M |
M |
ICC Profile |
M |
U |
SOP Common |
M |
M |
Common Instance Reference |
M |
M |
Real-Time Acquisition |
M |
M |
Current Frame Functional Groups |
M |
M |
Table A.1-12. Real-Time Object Modules Overview - Waveforms |
||
IODs |
|
RTV Basic Voice Audio |
Modules |
|
|
Patient |
M |
|
Clinical Trial Subject |
U |
|
General Study |
M |
|
Patient Study |
U |
|
Clinical Trial Study |
U |
|
General Series |
M |
|
Clinical Trial Series |
U |
|
General Equipment |
M |
|
Enhanced General Equipment |
M |
|
Synchronization |
M |
|
Waveform Identification |
M |
|
Real-Time Bulk Data Flow |
M |
|
Acquisition Context |
M |
|
SOP Common |
M |
|
Current Frame Functional Groups |
M |
|
A.2 Computed Radiography Image IOD
A.2.1 CR Image IOD Description
The Computed Radiography (CR) Image Information Object Definition specifies an image that has been created by a computed ra- diography imaging device.
- Standard -