Материал: MasterPass _ Pharmacology in 7 Days for Medical Students

PHARMACOLOGY IN 7 DAYS FOR MEDICAL STUDENTS
Proton Pump Inhibitors (PPIs): These drugs are given orally in the form of capsules in which enteric-coated granules are enclosed to protect them from HCl destruction in the stomach. In intestines, shell is dissolved and granules are liberated. They are highly lipid-soluble and absorbed completely and rapidly from the intestines. Through blood, they reach their site of action, i.e. gastric canaliculi lined by parietal cells. Since the fluid in the gastric canaliculi is acidic and PPIs are basic in nature, the latter get ionised/protonated and trapped in the canaliculi, leading to a 1000-fold rise in their concentration. They are pro-drugs. They are activated into sulfenamide and sulfenic acid, which are interconvertable. These inhibitors combine with H+/ K+-ATPase proton pump (via the SH-group of cysteine amino acids in the extracellular domain of the proton pump) thus inhibiting it. This leads to total inhibition of HCl synthesis (called anacidity).
Antacids: Antacids are weak bases that react with gastric HCl to form salt and water. Although their principal mechanism of action is reduction of intragastric acidity, they may also promote mucosal defence mechanisms through stimulation of mucosal prostaglandin production. After a meal approximately 45 mEq/hr of HCl is secreted. A single dose of 156 mEq of antacid given 01 hr after a meal effectively neutralises gastric acid for up to 01 hr. However, acid neutralisation capacity among different proprietary formulations of antacids is highly variable depending on their:
1Rate of dissolution (tablet vs liquid).
2Water solubility.
3Rate of reaction with acid.
4Rate of gastric emptying.
Antileukotriene drugs
Drugs included: zileuton; zafirlukast; montelukast.
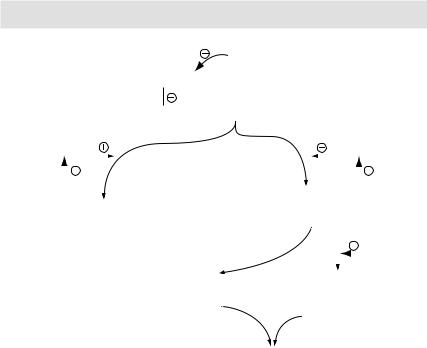
Leukotrienes are one of the many pro-inflammatory mediators released by the mast cells (and many other cells including basophils, eosinophils and neutrophils). The 5-lipoxygenase pathway of arachidonic acid metabolism leads to the formation of leukotrienes B4 (LTB4) and cysteinyl leukotrienes (which include LTC4, LTD4 and LTE4). Physiologically, LTB4 is a powerful chemoattractant for neutrophils and eosinophils. Cysteinyl leukotrienes, on the other hand, cause bronchoconstriction and increased endothelial permeability (→ ↑ mucus secretion). The net result is narrowing of the airways. Antileukotriene drugs block the leukotriene-induced narrowing of the airways.
Zileuton directly inhibits the 5-lipoxygenase enzyme, thus blocking the synthesis of all leukotrienes. Zafirlukast and montelukast, on the other hand, reversibly inhibit the cysteinyl leukotriene-1 receptor, thus blocking the cysteinyl leukotrienes from exerting their physiological actions.
Antileukotriene drugs are only approved for asthma prophylaxis. In cases of acute, severe, attack of asthma in which immediate bronchodilatation is required, these drugs are not very effective (β2-agonists like salbutamol, terbutaline, and anticholinergic drugs like ipratropium bromide are effective in emergency situations). When used as prophylactic agents, these drugs improve the respiratory functions and cause modest reductions in the doses of other antiasthmatics agents like β2-agonists and corticosteroids.
54
MECHANISMS OF ACTION
Glucocorticoids
Phospholipase A2
Plasmalogens 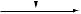 Arachidonic acid
Arachidonic acid
Cyclooxygenase |
|
|
|
|
Lipoxygenase |
|||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||||||
NSAIDs |
|
|
|
|
|
Zileuton |
||||||||||||
|
Cyclooxygenase |
|
Lipoxygenase products |
|||||||||||||||
|
(leukotrienes – LTB4; LTC4; LTD4; LTE4) |
|||||||||||||||||
|
|
|
products |
|||||||||||||||
(PGs; thromboxane A2) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||
|
|
|
|
|
|
|
|
|
Montelukast |
|||||||||
|
|
|
|
|
|
|
|
|
||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Zafirlukast |
||
|
|
|
|
|
Neutrophils |
|
Bronchoconstriction |
|||||||||||
|
|
|
|
|
& eosinophils chemotaxis |
& mucus secretion |
||||||||||||
|
|
|
|
|
|
|
causing airway narrowing |
|||||||||||
Worsening of asthma symptoms
Figure 3.2 Arachidonic acid metabolism
Cromolyn and nedocromil
These agents are mast-cell stabilisers. They decrease the release of pro-inflammatory mediators (like histamine and leukotrienes) from the mast cells. They do not in any way block the effects of those mediators that have already been released. Thus the only role of these agents is in asthma prophylaxis. Unlike β2-agonists, they cannot cause bronchodilatation; they can just prevent bronchoconstriction. They are particularly useful in allergic asthma in children, in whom they prevent allergeninduced bronchoconstriction and thus precipitation of an acute attack of asthma.
These agents are extremely insoluble and thus poorly absorbed so that even large doses of these agents given orally yield sub-therapeutic blood levels. Thus the only way to give these drugs is by aerosols in asthma patients.
Thiazide diuretics
As diuretic: After having reached their site of action at distal-convoluted tubules (DCT), thiazide diuretics inhibit reabsorption of NaCl by inhibiting NaCl symporter or co-transporter. NaCl is excreted by an equal amount of water to cause diuresis. Thiazides are moderate efficacy diuretics – only 5–10% of total Na+ load in tubular filtrate is inhibited by these drugs.
As antihypertensive:
AInitially there is a transient fall in blood pressure because of the diuretic effect as
explained below:
↑ Excretion of NaCl → ↓ ECF volume → ↓ venous return and cardiac output → ↓ BP.
55
56
Site of action of |
|
|
|
|
Na+ |
|
thiazide diuretics |
|
Na+ |
+ |
/K |
+ |
|
|
|
Na |
|
|||
Thiazide |
Na+/Cl– |
|
pump |
|||
diuretics |
pump |
K+ |
|
|
K+ |
|
|
|
Cl– |
|
|
Cl– |
|
Site of action of |
Luminal |
|
K+/Cl– |
|||
|
pump |
|||||
ADH, aldosterone |
|
|||||
surface |
|
|
|
|
||
& K+-sparing diuretics |
|
|
|
|
||
MoA of thiazide diuretics |
||||||
Site of action |
||||||
|
|
|
|
|
||
of loop diuretics |
|
|
|
|
|
|
|
|
|
|
|
ADH |
|
|
|
|
|
|
|
|
|
Aldosterone H2O |
H2O |
H2O |
||
|
|
|
|
K+-sparing diuretics |
|
|
|
|
|
|
|
|
|
|
Na+ |
|
Na+ |
Na+ |
|
|
|
|
|
|
Na+ |
|
|
Na+/K+ |
|
|
|
Na |
+ |
Na+/K+ |
|
|
pump |
|
|
|
|
|
|
K |
|
||||
|
Na+/K+/2 |
|
K |
+ |
+ |
K+ |
|||
Loop diuretics |
|
|
pump |
|
|
||||
Cl– pump |
K+ |
Luminal |
|
|
|
||||
|
|
|
|
|
|||||
|
|
K+ |
K+ |
surface |
MoA of K+-sparing |
|
|||
|
|
|
|
K+/Cl– |
|
|
diuretics |
|
|
|
|
|
|
pump |
|
|
|
|
|
Luminal |
|
|
Cl– |
|
|
Cl– |
|
|
|
|
|||||
surface MoA |
|
|
|
|
|
||
of loop diuretics |
|
||||||
STUDENTS MEDICAL FOR DAYS 7 IN PHARMACOLOGY
Figure 3.3 Sites and mechanism of action of diuretics.

MECHANISMS OF ACTION
Once ECF volume is made up by compensatory mechanism in the body (i.e. more NaCl and water reabsorption from DCT), blood pressure returns to normal.
BNatriuretic action: ↓ concentration of Na+ in vascular beds → loss of response of vascular smooth muscles to circulating catecholamines → vasodilatation → fall in BP.
Good points of thiazide diuretics as an antihypertensive include the following:
ACost-effectiveness.
BGood compliance.
CConvenient route of administration – oral.
DAntihypertensive/diuretic effect is independent of metabolic acidosis or alkalosis.
ETo have synergistic effect, they are combined with other antihypertensive drugs (like ACEIs, beta-blockers).
FSince the therapeutic index is high, these drugs are reasonably safe.
Loop diuretics
Main mechanism: On oral administration, having being absorbed readily, they are secreted in the proximal convoluted tubules (PCT) by an active secretory process, which is specific for organic acids. They reach their site of action, i.e. thick ascending limb (TAL) of loop of Henle. Here they inhibit the Na+/K+/2Cl– transporter, resulting in NaCl reabsorption inhibition from TAL of loop of Henle. NaCl in the urinary filtrate produces osmotic pressure and inhibits reabsorption of water from the remaining parts of nephron. As compared to thiazide diuretics, loop diuretics cause a greater volume of water and solutes to be excreted from the kidneys. Between 15% and 25% of total Na+ load in tubular filtrate is inhibited by these drugs, leading to massive diuresis.
Contributory mechanisms:
ADecreased filtration fraction (the ratio of GFR/renal blood flow).
BInterference with counter-current multiplier exchange system.
CChanges in renal hemodynamic state.
DDecrease degradation of PGI2 and PGE2.
Potassium-sparing diuretics
Potassium-sparing diuretics prevent K+ secretion by antagonising the effects of aldosterone at the late distal and cortical collecting tubules. Inhibition may occur either by direct pharmacologic antagonism of mineralocorticoid receptors (spironolactone and eplerenone) or by inhibition of Na+ influx through ion channels in the luminal membrane (amiloride and triamterene).
Sulfonylureas
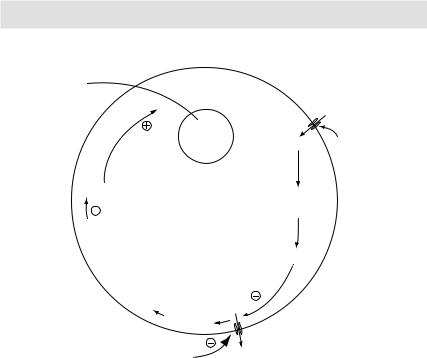
Effects on pancreas
1These agents are insulin secretagogues, i.e. they stimulate insulin release from pancreatic β-cells by blocking the ATP-sensitive K+ channels. They have specific
binding receptors on the ATP-sensitive K+ channels. The ligand-receptor binding leads to decreased K+ efflux, leading to depolarisation of the β-cells. Also, it causes
opening up of the voltage-dependent Ca++ channels. Ca++ influx in turn causes increased insulin release from the β-cells, leading to lowering of the glucose levels.
2Decrease in serum glucagon level.
57
PHARMACOLOGY IN 7 DAYS FOR MEDICAL STUDENTS
Plasma
Insulin 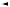
|
crea |
an |
|
P |
|
tic
β-cell
Insulin vesicle
Ca++  Ca++
Ca++
M e m
b
r
a
n e d
e |
|
|
|
|
p |
|
|
|
|
o |
|
|
|
|
l |
|
|
|
|
a |
|
|
|
|
r |
|
|
|
|
i |
|
|
|
|
z |
|
|
|
|
a |
|
|
|
|
t |
|
|
|
|
i |
|
|
|
+ |
o |
|
|
K |
|
n |
↓ |
+ |
|
|
|
|
K |
efflux |
|
|
|
|
|
|
Glucose |
|
Glucose |
GLUT2 |
|
transporter |
||
|
Glucose metabolism
ATP
 K+/ATP channel
K+/ATP channel
K+
Sulfonylurea /meglitinide
Figure 3.4 Physiology of insulin release from pancreatic β-cells, and mechanism of action of sulfonylurea and meglitinide
Effect on peripheral insulin receptors
3Unregulation of insulin receptors on target organs thus enhancing peripheral utilisation of glucose (especially in muscles and adipose tissue).
Effects on liver
4Decrease in hepatic insulin degradation.
5Decrease in hepatic gluconeogenesis.
Note: It is important to understand here that the main pharmacologic effect of sulfonylureas (i.e. ↑ insulin secretion from the pancreas) is dependent on functioning β-cells. In type-1 diabetes mellitus in which β-cells are destroyed (probably by an autoimmune mechanism), these agents are ineffective.
Meglitinides
Just like sulfonylureas, these agents are insulin secretagogues, i.e. they stimulate insulin release from pancreatic β-cells. Their mechanism of action is also similar to that of sulfonylureas, i.e. they block the ATP-sensitive K+ channels (→ ↑ insulin secretion). Meglitinides have two binding sites in common with sulfonylureas and one unique binding site.
58