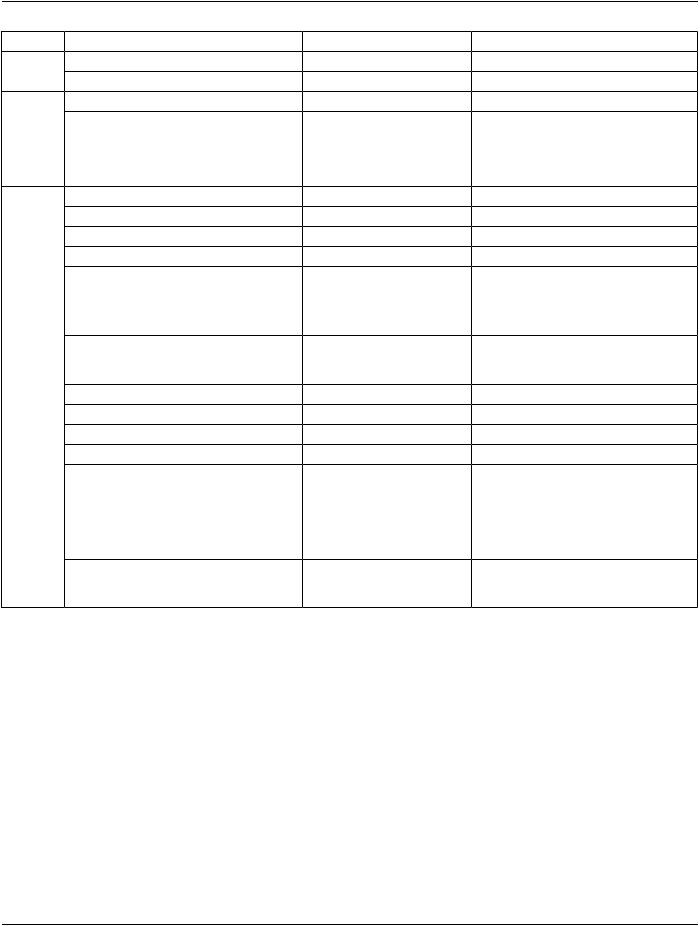
Материал: part03

|
DICOM PS3.3 2020a - Information Object Definitions |
Page 341 |
||
IE |
Module |
Reference |
|
Usage |
|
ICC Profile |
C.11.15.1 |
M |
|
|
SOP Common |
C.12.1 |
M |
|
|
Common Instance Reference |
C.12.2 |
U |
|
|
Frame Extraction |
C.12.3 |
C - Required if the SOP Instance was |
|
|
|
|
created in response to a Frame-Level |
|
|
|
|
retrieve request |
|
A.36.4.3.1 Enhanced MR Color Image IOD Content Constraints
The General Image Module, Overlay Plane Module and VOI LUT Module shall not be used in a Standard Extended SOP Class of the Enhanced MR Color Image.
Note
In order to annotate images, whether during acquisition or subsequently, SOP Instances of the Color Softcopy Presentation State Storage or the Structured Report Storage SOP Classes that reference the image SOP Instance, may be used.
NostandardmechanismisprovidedforinclusionofannotationswithintheimageSOPInstanceitself,andimplementersarediscouraged from using private extensions to circumvent this restriction.
Color Softcopy Presentation State Storage Instances that are generated during acquisition may be referenced from the Image SOP Instance by using the Referenced Presentation State Sequence in the MR Image and Spectroscopy Instance Macro invoked from the Enhanced MR Image Module. See Section C.8.13.2.
Photometric Interpretation (0028,0004) shall be RGB for uncompressed or lossless compressed Transfer Syntaxes that do not have defined color space transformations, YBR_ICT for irreversible JPEG2000 Transfer Syntaxes, YBR_RCT for reversible JPEG2000 Transfer Syntaxes, YBR_PARTIAL_420 for MPEG2, MPEG-4 AVC/H.264, HEVC/H.265 Transfer Syntaxes and YBR_FULL_422 for JPEG lossy compressed Transfer Syntaxes and YBR_FULL or RGB for RLE Transfer Syntaxes.
Note
Future lossless and lossy Transfer Syntaxes may lead to the need for new definitions and choices for Photometric Interpret- ation.
Pixel Presentation (0008,9205) shall be TRUE_COLOR.
A.36.4.4 Enhanced MR Color Image Functional Group Macros
Table A.36-2 specifies the use of the Functional Group Macros used in the Multi-frame Functional Groups Module for the Enhanced MR Color Image IOD.
A.37 Raw Data IOD
A.37.1 Raw Data IOD Description
The Raw Data Information Object Definition (IOD) specifies raw data.
A.37.2 Raw Data Entity-Relationship Model
This IOD uses the E-R Model in Section A.1.2, with only the Raw Data IE below the Series IE.
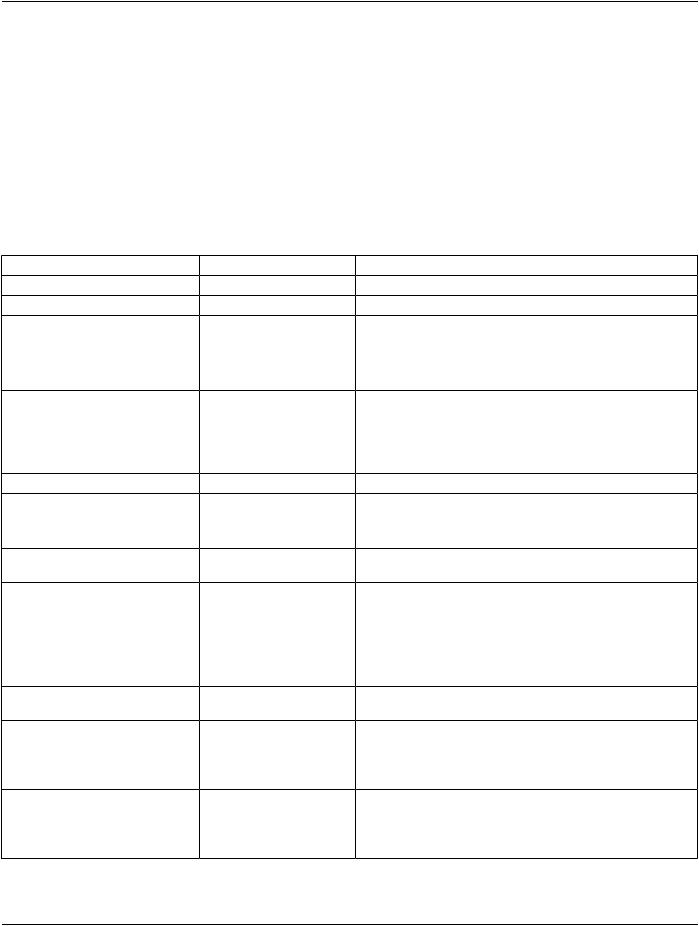
A.37.3 Raw Data IOD Module Table
Table A.37-1 specifies the Modules of the Raw Data IOD.
- Standard -
Page 342 DICOM PS3.3 2020a - Information Object Definitions
Table A.37-1. Raw Data IOD Modules
IE |
Module |
Reference |
Usage |
Patient |
Patient |
C.7.1.1 |
M |
|
Clinical Trial Subject |
C.7.1.3 |
U |
Study |
General Study |
C.7.2.1 |
M |
|
Patient Study |
C.7.2.2 |
U |
|
Clinical Trial Study |
C.7.2.3 |
U |
Series |
General Series |
C.7.3.1 |
M |
|
Clinical Trial Series |
C.7.3.2 |
U |
Frame of |
Frame of Reference |
C.7.4.1 |
U |
Reference |
Synchronization |
C.7.4.2 |
C - Required if time |
|
|||
|
|
|
synchronizationwasapplied. |
Equipment |
General Equipment |
C.7.5.1 |
M |
Raw Data |
Acquisition Context |
C.7.6.14 |
M |
|
Specimen |
C.7.6.22 |
U |
|
Raw Data |
C.19.1 |
M |
|
SOP Common |
C.12.1 |
M |
Note
The Specimen Identification Module was previously included in this IOD but has been retired, and its functionality replaced by the Specimen Module. See PS3.3-2008.
A.38 Enhanced Computed Tomography Image IOD
A.38.1 Enhanced CT Image IOD
A.38.1.1 Enhanced CT Image IOD Description
The Enhanced Computed Tomography (CT) Image Information Object Definition (IOD) specifies an image that has been created by a computed tomography imaging device.
A.38.1.2 Enhanced CT Image IOD Entity-Relationship Model
This IOD uses the E-R Model in Section A.1.2, with only the Image IE below the Series IE.
A.38.1.3 Enhanced CT Image IOD Module Table
Table A.38-1 specifies the Modules of the Enhanced CT Image IOD.
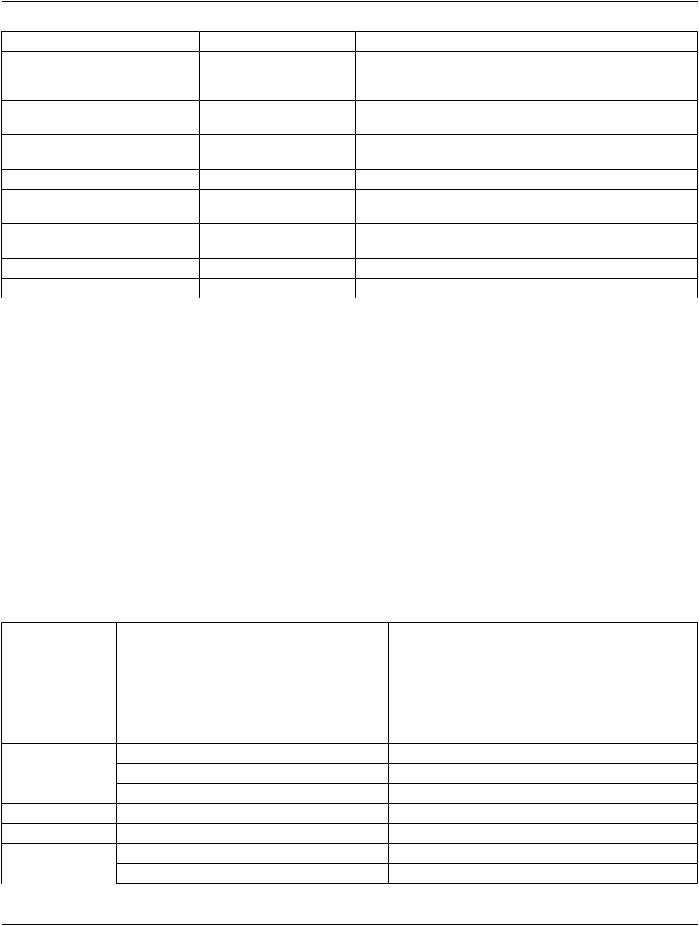
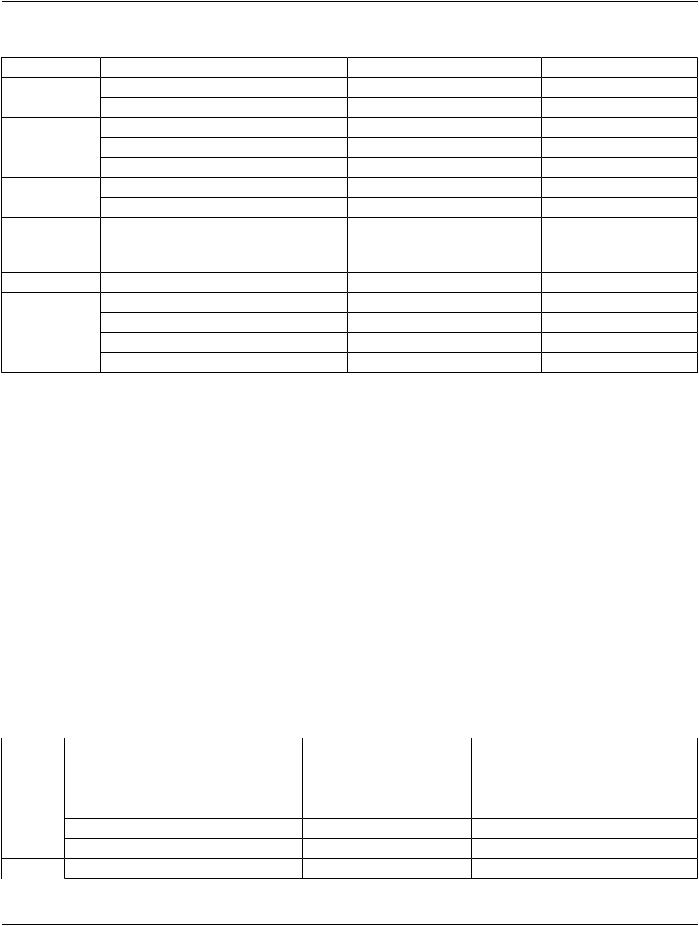
Table A.38-1. Enhanced CT Image IOD Modules
IE |
Module |
Reference |
Usage |
Patient |
Patient |
C.7.1.1 |
M |
|
Clinical Trial Subject |
C.7.1.3 |
U |
Study |
General Study |
C.7.2.1 |
M |
|
Patient Study |
C.7.2.2 |
U |
|
Clinical Trial Study |
C.7.2.3 |
U |
Series |
General Series |
C.7.3.1 |
M |