Материал: part03
|
DICOM PS3.3 2020a - Information Object Definitions |
Page 301 |
|
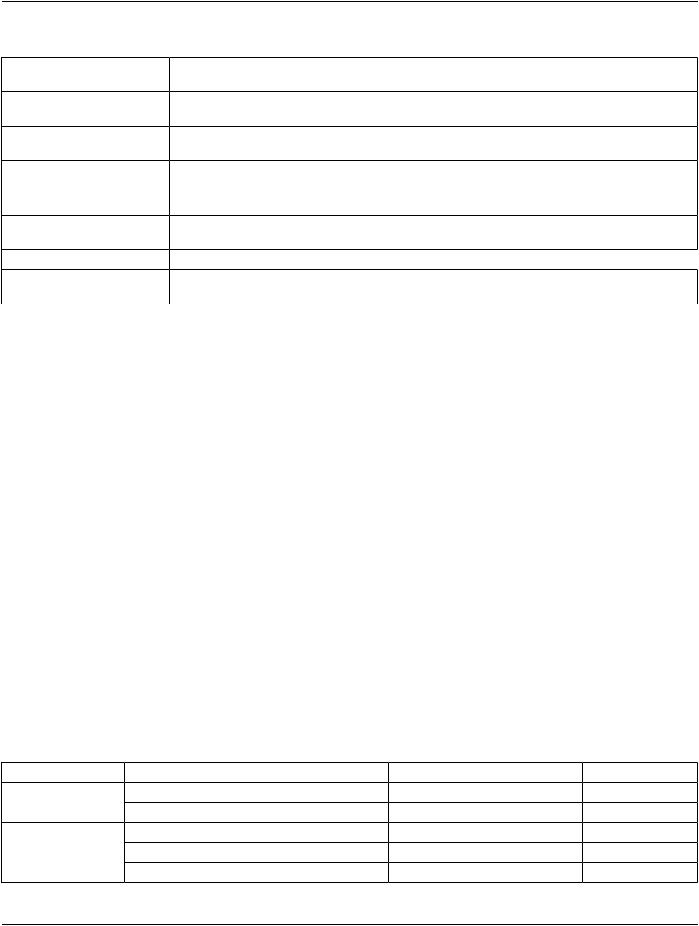
Table A.35.1-2. Relationship Content Constraints for Basic Text SR IOD |
|
||
Source Value Type |
Relationship Type (Enumerated |
Target Value Type |
|
|
Values) |
|
|
CONTAINER |
CONTAINS |
CONTAINER |
HAS OBS CONTEXT |
CONTAINER, IMAGE1, |
HAS ACQ CONTEXT |
WAVEFORM1, COMPOSITE1 |
|
any type |
HAS CONCEPT MOD |
TEXT |
HAS PROPERTIES |
PNAME |
HAS PROPERTIES |
TEXT |
INFERRED FROM |
TEXT, CODE, DATETIME, DATE, TIME, UIDREF, PNAME, COMPOSITE1, IMAGE1, WAVEFORM1, CONTAINER
TEXT, CODE, DATETIME, DATE, TIME, UIDREF, PNAME,
COMPOSITE1
TEXT, CODE, DATETIME, DATE, TIME, UIDREF, PNAME
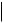 TEXT, CODE2
TEXT, CODE2
TEXT, CODE, DATETIME, DATE, TIME, UIDREF, PNAME,
IMAGE1, WAVEFORM1, COMPOSITE1
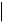 TEXT, CODE, DATETIME, DATE, TIME, UIDREF, PNAME
TEXT, CODE, DATETIME, DATE, TIME, UIDREF, PNAME 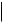
TEXT, CODE, DATETIME, DATE, TIME, UIDREF, PNAME,
IMAGE1, WAVEFORM1, COMPOSITE1
Note
1.WhichSOPClassestheIMAGE,WAVEFORMorCOMPOSITEValueTypemayreferto,isdocumentedintheConform- ance Statement for an application (see PS3.2 and PS3.4).
2.The HAS CONCEPT MOD relationship is used to modify the meaning of the Concept Name of a Source Content Item, for example to provide a more descriptive explanation, a different language translation, or to define a post-coordinated concept.
A.35.2 Enhanced SR IOD
A.35.2.1 Enhanced SR IOD Description
The Enhanced Structured Report (SR) IOD is a superset of the Basic Text SR IOD. It is also intended for the representation of reports with minimal usage of coded entries (typically Document Title and headings) and a hierarchical tree of headings under which may appear text and subheadings. In addition, it supports the use of numeric measurements with coded measurement names and units. Reference to SOP Instances (e.g., images or waveforms or SR Documents) is restricted to appear at the level of the leaves of this primarily textual tree. It enhances references to SOP Instances with spatial regions of interest (points, lines, circle, ellipse, etc.) and temporal regions of interest.
A.35.2.2 Enhanced SR IOD Entity-Relationship Model
This IOD uses the E-R Model in Section A.1.2, with only the SR Document IE below the Series IE. The Frame of Reference IE is not a component of this IOD.
A.35.2.3 Enhanced SR IOD Module Table
Table A.35.2-1 specifies the Modules of the Enhanced SR IOD.
Table A.35.2-1. Enhanced SR IOD Modules
IE |
Module |
Reference |
Usage |
Patient |
Patient |
C.7.1.1 |
M |
|
Clinical Trial Subject |
C.7.1.3 |
U |
Study |
General Study |
C.7.2.1 |
M |
|
Patient Study |
C.7.2.2 |
U |
|
Clinical Trial Study |
C.7.2.3 |
U |
- Standard -
Page 302 |
DICOM PS3.3 2020a - Information Object Definitions |
|
|
IE |
Module |
Reference |
Usage |
Series |
SR Document Series |
C.17.1 |
M |
|
Clinical Trial Series |
C.7.3.2 |
U |
Frame of Reference Synchronization |
C.7.4.2 |
U |
|
Equipment |
General Equipment |
C.7.5.1 |
M |
Document |
SR Document General |
C.17.2 |
M |
|
SR Document Content |
C.17.3 |
M |
|
SOP Common |
C.12.1 |
M |
Note
The Specimen Identification Module was previously included in this IOD but has been retired. See PS3.3-2008.
A.35.2.3.1 Enhanced SR IOD Content Constraints
A.35.2.3.1.1 Value Type
Value Type (0040,A040) in Content Sequence (0040,A730) of the SR Document Content Module is constrained to the following Enumerated Values (see Table C.17.3-7 for Value Type definitions):
Enumerated Values:
TEXT
CODE
NUM
DATETIME
DATE
TIME
UIDREF
PNAME
SCOORD
TCOORD
COMPOSITE
IMAGE
WAVEFORM
CONTAINER
A.35.2.3.1.2 Relationship Constraints
Relationships between Content Items in the content of this IOD shall be conveyed in the by-value mode. See Table C.17.3-8 for Re- lationship Type definitions.
Note
Relationships by-reference are forbidden. Therefore, Referenced Content Item Identifier (0040,DB73) is not present in any of the Content Items within the SR Document Content Module.
Table A.35.2-2 specifies the relationship constraints of this IOD.
Table A.35.2-2. Relationship Content Constraints for Enhanced SR IOD
Source Value Type |
Relationship Type (Enumerated |
Target Value Type |
|
|
Values) |
|
|
CONTAINER |
CONTAINS |
TEXT, CODE, NUM, DATETIME, DATE, TIME, UIDREF, |
|
|
|
PNAME, SCOORD, TCOORD, COMPOSITE1, IMAGE1, |
|
|
|
WAVEFORM1, CONTAINER |
|
CONTAINER |
HAS OBS CONTEXT |
TEXT, CODE, NUM, DATETIME, DATE, TIME, UIDREF, |
|
|
|
PNAME, COMPOSITE1 |
|
- Standard -

|
DICOM PS3.3 2020a - Information Object Definitions |
Page 303 |
|
Source Value Type |
Relationship Type (Enumerated |
Target Value Type |
|
|
Values) |
|
|
CONTAINER, IMAGE1, |
HAS ACQ CONTEXT |
WAVEFORM1, COMPOSITE1, |
|
NUM |
|
any type |
HAS CONCEPT MOD |
TEXT, CODE, NUM |
HAS PROPERTIES |
TEXT, CODE, NUM, DATETIME, DATE, TIME, UIDREF, PNAME
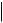 TEXT, CODE2
TEXT, CODE2
TEXT, CODE, NUM, DATETIME, DATE, TIME, UIDREF, PNAME, IMAGE1, WAVEFORM1, COMPOSITE1, SCOORD, TCOORD
PNAME |
HAS PROPERTIES |
TEXT, CODE, DATETIME, DATE, TIME, UIDREF, PNAME |
TEXT, CODE, NUM |
INFERRED FROM |
TEXT, CODE, NUM, DATETIME, DATE, TIME, UIDREF, |
|
|
PNAME, IMAGE1, WAVEFORM1, COMPOSITE1, SCOORD, |
|
|
TCOORD |
SCOORD |
SELECTED FROM |
IMAGE1 |
TCOORD |
SELECTED FROM |
SCOORD, IMAGE1, WAVEFORM1 |
Note |
|
|
1.WhichSOPClassestheIMAGE,WAVEFORMorCOMPOSITEValueTypemayreferto,isdocumentedintheConform- ance Statement for an application (see PS3.2 and PS3.4).
2.The HAS CONCEPT MOD relationship is used to modify the meaning of the Concept Name of a Source Content Item, for example to provide a more descriptive explanation, a different language translation, or to define a post-coordinated concept.
A.35.3 Comprehensive SR IOD
A.35.3.1 Comprehensive SR IOD Description
TheComprehensiveSRIODisasupersetoftheBasicTextSRIODandtheEnhancedSRIOD,whichspecifiesaclassofdocuments, the content of which may include textual and a variety of coded information, numeric measurement values, references to the SOP Instances and spatial or temporal regions of interest within such SOP Instances. Relationships by-reference are enabled between Content Items.
A.35.3.2 Comprehensive SR IOD Entity-Relationship Model
This IOD uses the E-R Model in Section A.1.2, with only the SR Document IE below the Series IE. The Frame of Reference IE is not a component of this IOD.
A.35.3.3 Comprehensive SR IOD Module Table
Table A.35.3-1 specifies the Modules of the Comprehensive SR IOD.
Table A.35.3-1. Comprehensive SR IOD Modules
IE |
Module |
Reference |
Usage |
Patient |
Patient |
C.7.1.1 |
M |
|
Clinical Trial Subject |
C.7.1.3 |
U |
Study |
General Study |
C.7.2.1 |
M |
|
Patient Study |
C.7.2.2 |
U |
|
Clinical Trial Study |
C.7.2.3 |
U |
Series |
SR Document Series |
C.17.1 |
M |
|
Clinical Trial Series |
C.7.3.2 |
U |
- Standard -

Page 304 |
DICOM PS3.3 2020a - Information Object Definitions |
|
|
IE |
Module |
Reference |
Usage |
Frame of Reference Synchronization |
C.7.4.2 |
U |
|
Equipment |
General Equipment |
C.7.5.1 |
M |
Document |
SR Document General |
C.17.2 |
M |
|
SR Document Content |
C.17.3 |
M |
|
SOP Common |
C.12.1 |
M |
Note
The Specimen Identification Module was previously included in this IOD but has been retired. See PS3.3-2008.
A.35.3.3.1 Comprehensive SR IOD Content Constraints
A.35.3.3.1.1 Value Type
Value Type (0040,A040) in Content Sequence (0040,A730) of the SR Document Content Module is constrained to the following Enumerated Values (see Table C.17.3-7 for Value Type definitions):
Enumerated Values:
TEXT
CODE
NUM
DATETIME
DATE
TIME
UIDREF
PNAME
SCOORD
TCOORD
COMPOSITE
IMAGE
WAVEFORM
CONTAINER
A.35.3.3.1.2 Relationship Constraints
Relationships between Content Items in the content of this IOD may be conveyed either by-value or by-reference. Table A.35.3-2 specifies the relationship constraints of this IOD. See Table C.17.3-8 for Relationship Type definitions.
Table A.35.3-2. Relationship Content Constraints for Comprehensive SR IOD
Source Value Type |
Relationship Type (Enumerated |
Target Value Type |
|
Values) |
|
CONTAINER |
CONTAINS |
TEXT, CODE, NUM, |
HAS OBS CONTEXT |
CONTAINER |
|
CONTAINER, IMAGE1, |
HAS ACQ CONTEXT |
WAVEFORM1, COMPOSITE1, |
|
NUM |
|
any type |
HAS CONCEPT MOD |
TEXT, CODE, NUM |
HAS PROPERTIES |
TEXT, CODE, NUM, DATETIME, DATE, TIME, UIDREF, PNAME, SCOORD, TCOORD, COMPOSITE1, IMAGE1, WAVEFORM1, CONTAINER (see below).
TEXT, CODE, NUM, DATETIME, DATE, TIME, UIDREF,
PNAME, COMPOSITE1
TEXT, CODE, NUM, DATETIME, DATE, TIME, UIDREF, PNAME, CONTAINER
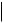 TEXT, CODE2
TEXT, CODE2
TEXT, CODE, NUM, DATETIME, DATE, TIME, UIDREF, PNAME, IMAGE1, WAVEFORM1, COMPOSITE1, SCOORD, TCOORD, CONTAINER
- Standard -
|
DICOM PS3.3 2020a - Information Object Definitions |
Page 305 |
||
Source Value Type |
Relationship Type (Enumerated |
Target Value Type |
|
|
|
Values) |
|
|
|
PNAME |
HAS PROPERTIES |
TEXT, CODE, DATETIME, DATE, TIME, UIDREF, PNAME |
||
TEXT, CODE, NUM |
INFERRED FROM |
TEXT, CODE, NUM, DATETIME, DATE, TIME, UIDREF, |
||
|
|
PNAME, IMAGE1, WAVEFORM1, COMPOSITE1, SCOORD, |
||
|
|
TCOORD, CONTAINER |
|
|
SCOORD |
SELECTED FROM |
IMAGE1 |
|
|
TCOORD |
SELECTED FROM |
SCOORD, IMAGE1, WAVEFORM1 |
|
|
Note
1.WhichSOPClassestheIMAGE,WAVEFORMorCOMPOSITEValueTypemayreferto,isdocumentedintheConform- ance Statement for an application (see PS3.2 and PS3.4).
2.The HAS CONCEPT MOD relationship is used to modify the meaning of the Concept Name of a Source Content Item, for example to provide a more descriptive explanation, a different language translation, or to define a post-coordinated concept.
The HAS CONCEPT MOD and CONTAINS relationships shall not be conveyed by-reference.
Relationships by-reference to ancestor Content Items are forbidden in this IOD to prevent loops.
A.35.4 Key Object Selection Document IOD
A.35.4.1 Key Object Selection Document IOD Description
The Key Object Selection Document IOD is intended for flagging one or more significant images, waveforms, or other composite SOP
Instances.
A.35.4.2 Key Object Selection Document IOD Entity-Relationship Model
This IOD uses the E-R Model in Section A.1.2, with only the SR Document IE below the Series IE. The Frame of Reference IE is not a component of this IOD.
A.35.4.3 Key Object Selection Document IOD Module Table
Table A.35.4-1 specifies the Modules of the Key Object Selection Document IOD.
Table A.35.4-1. Key Object Selection Document IOD Modules
IE |
Module |
Reference |
Usage |
Patient |
Patient |
C.7.1.1 |
M |
|
Clinical Trial Subject |
C.7.1.3 |
U |
Study |
General Study |
C.7.2.1 |
M |
|
Patient Study |
C.7.2.2 |
U |
|
Clinical Trial Study |
C.7.2.3 |
U |
Series |
Key Object Document Series |
C.17.6.1 |
M |
|
Clinical Trial Series |
C.7.3.2 |
U |
Equipment |
General Equipment |
C.7.5.1 |
M |
Document |
Key Object Document |
C.17.6.2 |
M |
|
SR Document Content |
C.17.3 |
M |
|
SOP Common |
C.12.1 |
M |
- Standard -