Материал: 21_chin2011
Published on 03 March 2011. Downloaded by Lomonosov Moscow State University on 17/07/2013 21:31:02.
View Article Online
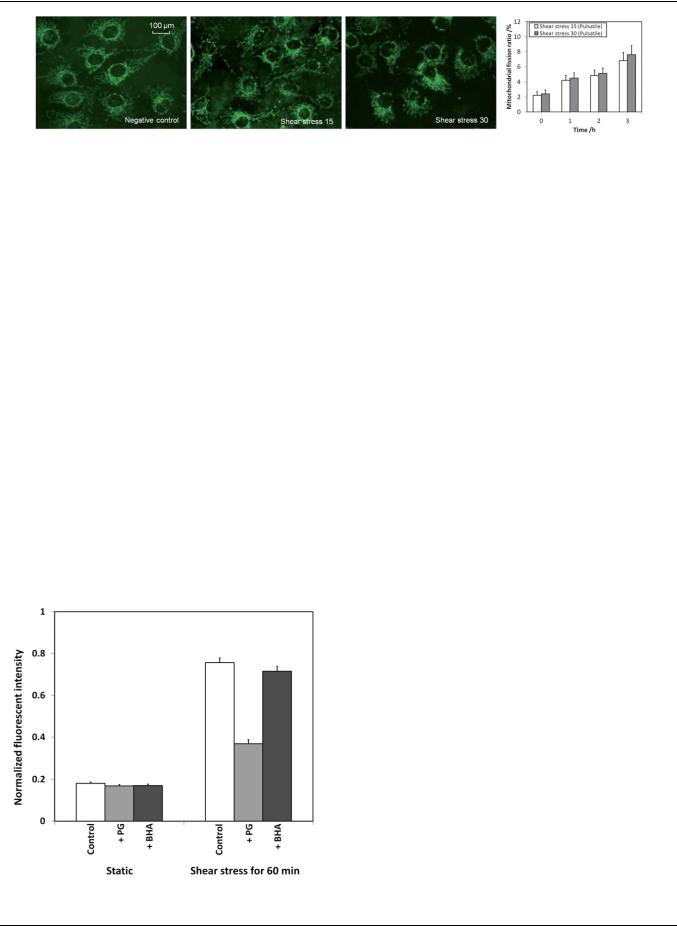
Fig. 8 Mitochondrial morphology of endothelial cells under different shear treatment conditions for 3 hours.
presented.25,26 Therefore, it is more realistic to expose the endothelial cells using pulsatile shear stress under different chemical conditions in the experiments.
The mitochondrial morphology of endothelial cells being exposed under different shear treatment conditions was investigated using MitoTracker staining and the fluorescent images are shown in Fig. 8. The endothelial cells under static condition (negative control) were mostly tubular with the appearance of filamentous reticular networks. Only 2% of the cells had shorter and smaller mitochondria, indicating mitochondrial fragmentation. When the endothelial cells were exposed to a physiological pulsatile shear stress, some of the cells showed mitochondrial morphological changes. The mitochondrial fission ratios of the endothelial cells under these conditions for the first 3 hours are shown in Fig. 8. The levels of endothelial cells with mitochondrial fragmentation after being exposed to pulsatile shear stress of 15 and 30 dyne cm 2 for 3 hours were measured as 6.8% and 7.6%, respectively. This is reasonable as endothelial cells should remain viable and functioning under normal physiological pulsatile shear stress or during exhaustive exercise.
To evaluate the shear-induced ROS production, two antioxidants were examined, i.e. propyl gallate (PG) and butylated hydroxyanisole (BHA). PG is an antioxidant which inhibits the oxidation by hydrogen peroxide and oxidative free radicals. On the other hand, BHA is an antioxidant which stabilizes free radicals and acts as a free radical scavenger. In the experiments,
Fig. 9 Effects of propyl gallate (PG) and butylated hydroxyanisole (BHA) on shear-induced increase of ROS production in endothelial cells.
the endothelial cells were pretreated with either one of the antioxidants at 20 mM for 30 min. Subsequently, the endothelial cells were being exposed to the pulsatile shear stress of 30 dyne cm 2 for one hour, and the ROS levels were measured as shown in Fig. 9. The ROS level was increased by 4-fold without the addition of antioxidants. The ROS level was significantly reduced by PG, but not BHA suggesting that the fast pulsatile shear stress may cause the production of hydrogen peroxide in endothelial cells.
Effects of glucose concentrations on ROS levels
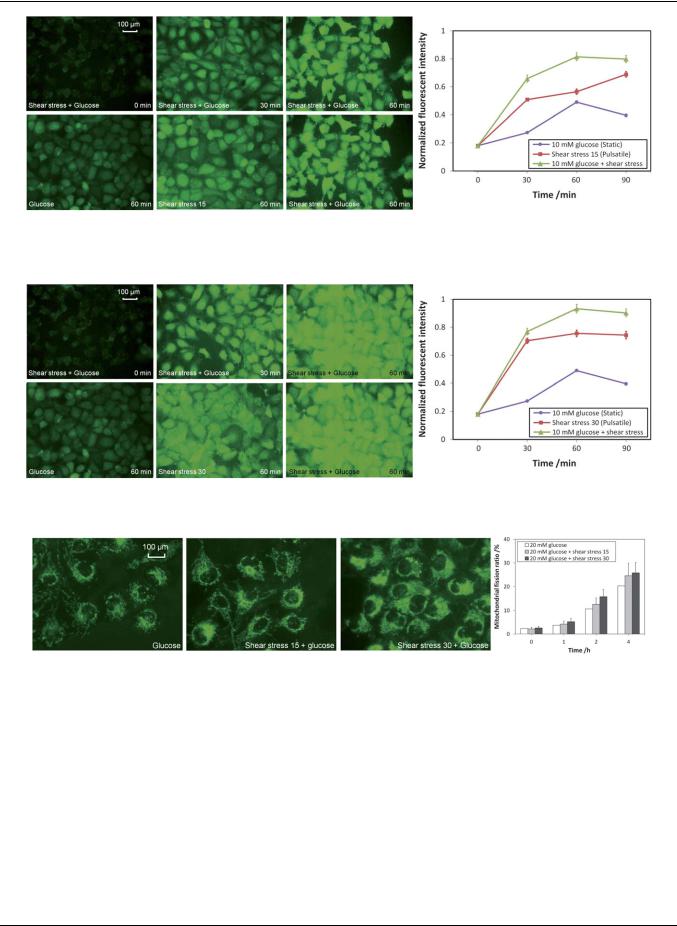
For normal people, the concentration of glucose in the blood is ranging from 4 to 6 mM. For diabetes patients, however, the concentration of glucose in the blood is above 6 mM and may be up to 20 mM. In these experiments, 10 mM of glucose was added to mimic the plasma condition of a diabetes patient and the ROS level of the endothelial cells was monitored under normal physiological pulsatile shear stress condition or under exhaustive exercise. The experimental results for the normal physiological pulsatile shear stress condition are shown in Fig. 10. When the endothelial cells were being exposed to 10 mM glucose under the normal pulsatile shear stress of 15 dyne cm 2, the ROS level was quickly increased for over 4-fold in the first 60 min and stabilized afterward. By comparing the ROS level at the 60 min time point between the cells treated with 10 mM glucose in a static state and that under a normal shear stress of 15 dyne cm 2, the ROS level was increased 1.7 fold (0.81/0.49) (graph in Fig. 10). It can be concluded that although high concentration of glucose can elevate the level of intracellular ROS, a much higher level of ROS can be detected if the glucose solution is delivered under a normal pulsatile shear stress condition.
Fig. 11 shows the results of endothelial cells being exposed to the fast pulsatile shear stress of 30 dyne cm 2 (exhaustive exercise). Similar to the ROS elevation profile observed under the normal pulsatile shear stress of 15 dyne cm 2, the ROS level was significantly increased for nearly 5-fold in the first 60 min and stabilized afterward. By comparing the ROS level at the 60 min time point between the cells treated with 10 mM glucose in a static state and that under the high level of shear stress of 30 dyne cm 2, the ROS level was increased 1.9-fold (0.93/0.49) (graph in Fig. 11). By comparing the ROS level in the 60 min time point between the one with normal pulsatile shear stress of 15 dyne cm 2 and the one with a fast pulsatile shear stress of 30 dyne cm 2, the ROS level was elevated by 15% (0.93/0.81) (graphs in Fig. 10 and 11). It can be concluded that the high glucose level under exhaustive exercise condition can substantially increase the intracellular level of ROS.
This journal is ª The Royal Society of Chemistry 2011 |
Lab Chip, 2011, 11, 1856–1863 | 1861 |
Published on 03 March 2011. Downloaded by Lomonosov Moscow State University on 17/07/2013 21:31:02.
View Article Online
Fig. 10 The ROS level in endothelial cells being exposed to 10 mM glucose with normal pulsatile shear stress of 15 dyne cm 2.
Fig. 11 The ROS level in endothelial cells being exposed to 10 mM glucose with fast pulsatile shear stress of 30 dyne cm 2.
Fig. 12 Mitochondrial morphology of endothelial cells with 20 mM glucose under different shear treatment conditions.
Effects of glucose concentrations on mitochondrial morphology
To investigate the mitochondrial morphological changes of endothelial cells, 20 mM of glucose was added and the endothelial cells were being exposed under different shear treatment conditions for 4 hours. The fluorescent images of the endothelial cells after being exposed for 4 hours and the mitochondrial fission ratios monitored for the first 4 hours are shown in Fig. 12. For endothelial cells treated with 20 mM glucose for 4 hours, mitochondrial fragmentation was prevalent with a mitochondrial fission ratio of 20%. When the endothelial cells were being exposed with 20 mM glucose plus the pulsatile shear stress of 15 or 30 dyne cm 2, the mitochondrial fission ratio was further
elevated from 20% to 25% and 26%, respectively. The results show that the chemical treatment (i.e. high concentration of glucose) produced a more dominant effect in promoting mitochondrial fission than the physical treatment (i.e. pulsatile shear stress), especially in such an extreme case of 20 mM of glucose.
Conclusions
In this paper, the intracellular ROS level and the mitochondrial morphology of endothelial cells under physiological pulsatile shear stresses and different glucose concentrations were investigated using a hemodynamic Lab-on-a-chip system. The
1862 | Lab Chip, 2011, 11, 1856–1863 |
This journal is ª The Royal Society of Chemistry 2011 |

Published on 03 March 2011. Downloaded by Lomonosov Moscow State University on 17/07/2013 21:31:02.
|
|
|
View Article Online |
||
intracellular ROS level was studied using real-time fluorescence |
3 |
L. G. Kevin, E. Novalija and D. F. Stowe, Anesth. Analg. (N.Y.), |
|
||
microscopy with the measurement of the oxidation of H2DCFDA |
|
2005, 101, 1275. |
|
||
4 |
M. Giorgio, M. Trinei, E. Migliaccio and P. G. Pelicci, Nat. Rev. Mol. |
||||
by hydroxyl radicals or hydrogen peroxide molecules. The |
|||||
|
Cell Biol., 2007, 8, 722. |
||||
elevated cellular ROS level led to morphological changes of |
|
||||
5 |
D. R. Green and G. Kroemer, Science, 2004, 305, 626. |
||||
mitochondria from filamentous reticular networks to diffused and |
6 |
T. Yu, S.-S. Sheu, J. L. Robotham and Y. Yoon, Cardiovasc. Res., |
|||
short fragments. The mitochondrial morphology was studied via |
|
2008, 79, 341. |
|
|
|
7 |
J. B. Brocard, G. L. Rintoul and I. J. Reynolds, Biol. Cell, 2003, 95, |
||||
fluorescent MitoTracker staining. The shear-induced cellular |
|||||
|
239. |
|
|
||
|
|
|
|
||
responses of endothelial cells under glucose concentration of 10 |
8 |
L. L. Ji, Proc. Soc. Exp. Biol. Med., 1999, 222, 283. |
|||
mM or 20 mM were realized and studied by mimicking the |
9 |
T. Mori and A. W. Cowley,Jr, Hypertension, 2004, 43, 341. |
|||
physiological pulsatile flow profiles in the blood vessel, i.e. during |
10 |
T. Inoguchi, P. Li, F. Umeda, H. Y. Yu, M. Kakimoto, M. Imamura, |
|||
|
T. Aoki, T. Etoh, T. Hashimoto, M. Naruse, H. Sano, H. Utsumi and |
||||
resting and exhaustive exercising. The results show that ROS level |
|
||||
|
H. Nawata, Diabetes, 2000, 49, 1939. |
||||
was elevated during exhaustive exercise (shear stress of 30 dyne |
11 |
F. Cosentino, M. Eto, P. D. Paolis, B. van der Loo, M. Bachschmid, |
|||
cm 2) and with high glucose concentration (diabetes patient). The |
|
V. Ullrich, A. Kouroedov, C. D. Gatti, H. Joch, M. Volpe and |
|||
results also show that pulsatile shear stress is an essential element |
|
T. F. Luscher,€ |
Circulation, 2003, 107, 1017. |
||
12 |
O. I. Aruoma, Free Radical Biol. Med., 1996, 20, 675. |
||||
|
|||||
to mimic the physiological conditions in the blood vessel, which |
13 |
T. Sano, F. Umeda, T. Hashimoto, H. Nawata and H. Utsumi, |
|||
also highlights the potential of using the developed Lab-on-a-chip |
|
Diabetologia, 1998, 41, 1355. |
|||
system in future hemodynamic studies as compared to the |
14 |
Z. Zhang, N. I. Dmitrieva, J.-H. Park, R. L. Levine and M. B. Burg, |
|||
|
Proc. Natl. Acad. Sci. U. S. A., 2004, 101, 9491. |
||||
conventional experimental setup. |
|
||||
15 |
H.-J. Hsieh, C.-C. Cheng, S.-T. Wu, J.-J. Chiu, B.-S. Wung and |
||||
|
|
D. L. Wang, J. Cell. Physiol., 1998, 175, 156. |
|||
Acknowledgements |
16 |
J. El-Ali, P. K. Sorger and K. F. Jensen, Nature, 2006, 442, 403. |
|||
17 |
L. K. Chin, A. Q. Liu, Y. C. Soh, C. S. Lim and C. L. Lin, Lab Chip, |
||||
This work was supported by the Academic Research Fund |
|
2010, 10, 1072. |
|
|
|
18 |
Y. Manevich, A. Al-Mehdi, V. Muzykantov and A. B. Fisher, Am. J. |
||||
(AcRF) Tier 1 (RG45/09, M52120104) by the Minister of |
|
Physiol. Heart Circ. Physiol., 2001, 280, H2126–H2135. |
|||
Education in Singapore and the research project (Grant No. |
19 |
E. Leclerc, Y. Sakai and T. Fujii, Biomed. Microdevices, 2003, 5, 109–114. |
|||
20 |
D. N. Ku, Annu. Rev. Fluid Mech., 1997, 29, 399–434. |
||||
MEWR C651/06/171) by Environmental and Water Industry |
|||||
21 |
X. He, D. N. Ku and J. E. Moore,Jr, Ann. Biomed. Eng., 1993, 21, 45–49. |
||||
|
|||||
Development Council of Singapore. |
22 |
D. J. Duncker and R. J. Bache, Physiol. Rev., 2008, 88, 1009–1086. |
|||
|
23 |
G. Hellstrom€ and N. G. Wahgren, Neurosurg. Rev., 1993, 16, 151– |
|||
|
|
156. |
|
|
|
References |
24 |
H. Lu, L. Y. Koo, W. M. Wang, D. A. Lauffenburger, L. G. Griffith |
|||
|
|
and K. F. Jensen, Anal. Chem., 2004, 76, 5257–5264. |
|||
1 J. S. Armstrong and M. Whiteman, Methods Cell Biol., 2007, 80, 355. |
25 |
P. F. Davies, Physiol. Rev., 1995, 75, 519. |
|||
2 T. Finkel and N. J. Holbrook, Nature, 2000, 408, 239. |
26 |
P. F. Davies and S. C. Tripathi, Circulation, 1993, 72, 239. |
|||
This journal is ª The Royal Society of Chemistry 2011 |
Lab Chip, 2011, 11, 1856–1863 | 1863 |