Материал: part20

Page 36 |
DICOM PS3.20 2020a - Imaging Reports using HL7 Clinical Document Architecture |
- Standard -

DICOM PS3.20 2020a - Imaging Reports using HL7 Clinical Document Architecture |
Page 37 |
6 Conformance
The CDA specification section 1.3 provides conformance requirements for Document Originators and Document Recipients.
Note
1.Consolidated CDA Implementation Guide Section 2.8 includes recommended best practices for Document Recipients displaying CDA documents.
2.There may be other CDA-related standards to which an application may claim conformance. For example, IHE Patient Care Coordination Technical Framework specifies a Document Consumer actor with four options for conformance.
A CDA document instance in accordance with this Standard asserts its conformance to a template by inclusion of the specified tem- plateID elements in the document, sections, and entries.
- Standard -

Page 38 |
DICOM PS3.20 2020a - Imaging Reports using HL7 Clinical Document Architecture |
- Standard -

DICOM PS3.20 2020a - Imaging Reports using HL7 Clinical Document Architecture |
Page 39 |
7 Document-level Templates
Document-leveltemplatesdescribethepurposeandrulesforconstructingaconformingCDAdocument.Documenttemplatesinclude constraints on the CDA header and sections by referring to templates, and constraints on the vocabulary used in those templates.
7.1 Imaging Report
Template ID |
1.2.840.10008.9.1 |
Name |
Imaging Report |
Effective Date |
2015/03/24 |
Version Label |
DICOM-20150324 |
Status |
Active |
Description |
This CDA Imaging Report document template defines the report content and technical |
|
constraintsfortoplevelelements,attributes,sections,andentriestobeusedinimagingreport |
|
instances. This template may apply to screening, diagnostic, or therapeutic radiology, |
|
cardiology, or other imaging reports. |
|
The body of an Imaging Report may contain five main imaging report sections: |
|
•Clinical information (optionally); |
|
•Current imaging procedure description; |
|
•Comparison studies (optionally); |
|
•Findings (optionally); |
|
•Impression; |
|
•plus potentially an Addendum(s) |
|
The report templates sponsored by the RSNA Radiology Reporting Initiative (http:// |
|
www.radreport.org) adhere to this general section outline. |
|
The section and subsection structure of this template is also identified by LOINC panel code |
|
87416-4. |
Classification |
CDA Document Level |
Relationships |
|
Context |
parent node |
Open/Closed |
Open |
Revision History |
DICOM-20150324: Initial version |
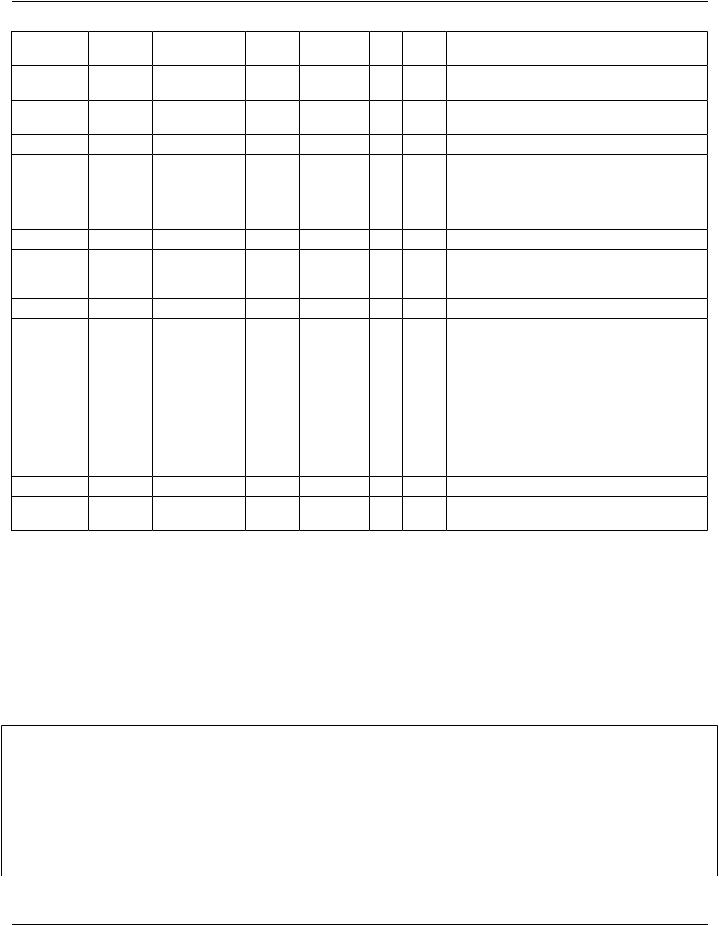
Business |
NestLevelElement/ |
Card |
Elem/Attr DataValueValue |
Subsidiary Template |
||
Name |
|
Attribute |
|
Conf |
TypeConf |
|
Imaging |
|
Clinical |
|
|
|
|
Report |
|
Document |
|
|
|
|
|
> |
templateId |
1..1 |
SHALL |
II |
|
|
>@ |
@root |
1..1 |
SHALL |
UIDSHALL1.2.840.10008.9.1 |
|
DocType |
> |
code |
1..1 |
SHALL |
CD SHALLValueSetLOINCImaging |
|
|
|
|
|
|
CWE Document Codes |
|
|
|
|
|
|
noNull1.3.6.1.4.1.12009.10.2.5 |
|
|
> |
|
1..1 |
SHALL |
|
8.1 General Header |
|
|
|
|
|
|
1.2.840.10008.9.20 |
- Standard -
Page 40 |
|
DICOM PS3.20 2020a - Imaging Reports using HL7 Clinical Document Architecture |
||||
Business |
NestLevelElement/ |
Card |
Elem/Attr DataValueValue |
Subsidiary Template |
||
Name |
|
Attribute |
|
Conf |
TypeConf |
|
|
> |
|
1..1 |
SHALL |
|
8.2 Imaging Header |
|
|
|
|
|
|
1.2.840.10008.9.21 |
|
> |
|
0..1 |
MAY |
|
8.3 Parent Document |
|
|
|
|
|
|
1.2.840.10008.9.22 |
|
> |
component |
1..1 |
SHALL |
|
|
|
>> |
structuredBody1..1 |
SHALL |
|
|
|
|
>>> |
component |
0..1 |
MAY |
|
|
Clinical |
>>>> |
section |
|
|
|
9.2ClinicalInformation |
Information |
|
|
|
|
|
1.2.840.10008.9.2 |
|
>>> |
component |
1..1 |
SHALL |
|
|
Procedure |
>>>> |
section |
|
|
|
9.3ImagingProcedure |
Description |
|
|
|
|
|
Description |
|
|
|
|
|
|
1.2.840.10008.9.3 |
|
>>> |
component |
0..1 |
MAY |
|
|
Comparison>>>> |
section |
|
|
|
9.4 Comparison Study |
|
Study |
|
|
|
|
|
1.2.840.10008.9.4 |
|
>>> |
component |
0..1 |
MAY |
|
|
Findings |
>>>> |
section |
|
|
|
9.5 Findings |
|
|
|
|
|
|
2.16.840.1.113883. |
|
|
|
|
|
|
10.20.6.1.2 |
|
>>> |
component |
1..1 |
SHALL |
|
|
Impression >>>> |
section |
|
|
|
9.6 Impression |
|
|
|
|
|
|
|
1.2.840.10008.9.5 |
|
>>> |
component |
0..* |
COND |
|
|
Addendum[*]>>>> |
section |
|
|
|
9.7 Addendum |
|
|
|
|
|
|
|
1.2.840.10008.9.6 |
7.1.1 clinicalDocument/code
Most of the codes in Value Set LOINC Imaging Document Codes are pre-coordinated with the imaging modality, body part examined, and/or specific imaging method. When pre-coordinated codes are used, any coded values elsewhere in the document describing the modality, body part, etc., must be consistent with the document type code. Local codes used for report types may be included as a translation element in the code.
Note
Use of Value Set LOINC Imaging Document Codes is harmonized with HL7 Consolidated CDA Templates for Clinical Notes, Release 2. DICOM CID 7001 “Diagnostic Imaging Report Headings”, used in TID 2000 “Basic Diagnostic Imaging Report”, is a subset of the LOINC Imaging Document Codes.
Example 7.1.1-1. clinicalDocument/code example with translation element for local code
<code code="18748-4"
displayName="Diagnostic Imaging Report" codeSystem="2.16.840.1.113883.6.1" codeSystemName="LOINC" >
<translation code="XRPEDS" displayName="Pediatric Radiography Report"
- Standard -