Материал: part20

Page 126 |
DICOM PS3.20 2020a - Imaging Reports using HL7 Clinical Document Architecture |
||
Coding SchemeCode Value |
Code Meaning |
Map to Template Section/Subsection |
|
Designator |
|
|
|
LN |
55109-3 |
Complications |
9.3 Imaging Procedure Description / 9.8.4 |
|
|
|
Complications |
LN |
55112-7 |
Summary |
9.6 Impression |
LN |
55113-5 |
Key Images |
9.6 Impression / 9.8.6 Key Images |
LN |
73569-6 |
Radiation Exposure and Protection9.3ImagingProcedureDescription/9.8.5Radiation |
|
|
|
Information |
Exposure and Protection Information |
LN |
55752-0 |
Clinical Information |
9.2 Clinical Information |
LN |
29549-3 |
Medications Administered |
9.3ImagingProcedureDescription/10.2Procedural |
|
|
|
Medication |
LN |
73568-8 |
Communication of Critical Results9.6Impression/9.8.10CommunicationofActionable |
|
|
|
|
Findings |
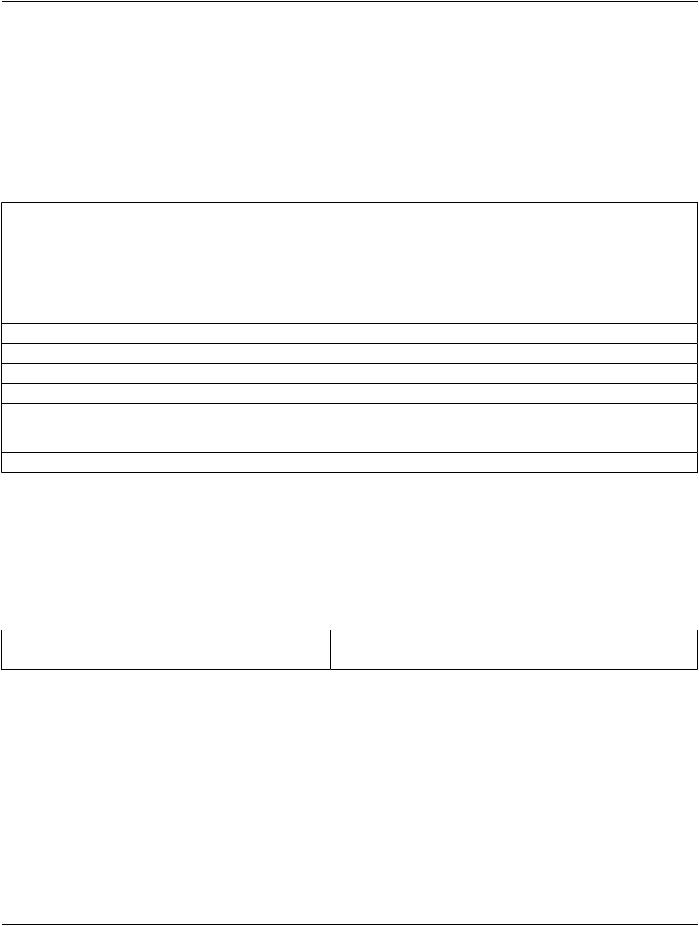
CDA Template 1.2.840.10008.9.1 requires a minimum of an Imaging Procedure Description section and an Impression section.
The section/code element shall be populated in accordance with the relevant CDA template; note that the code might not be the same as the Concept Name code of the SR section CONTAINER. The title element of each CDA section shall be populated as shown in Table C.4-2.
Table C.4-2. CDA Section mapping from SR
CDA Business Name |
DICOM SR |
<section>: Title |
Concept Name Code Sequence (0040,A043) > Code Meaning |
|
(0008,0104) [of the section CONTAINER content item] |
<section>: Text |
[See C.4.2] |
<section>: CodedObservation[*] |
[See C.4.3.1 and C.4.3.2] |
<section>: QuantityMeasurement[*] |
[See C.4.3.4] |
<section>: SOPInstance[*] |
[See C.4.3.3] |
SR allows sections to be qualified by observation context, using TID 1001 and its subsidiary templates. This capability is constrained in this mapping.
C.4.1.1 Section Observer Context
TID 1002 Observer Context allows identification of a human or device author.
Table C.4-3. CDA Section author mapping from SR
CDA Business Name |
DICOM SR |
<section>: AuthorID |
If (121005, DCM, "Observer Type") = (121007, DCM, "Device"), then (121012, |
|
DCM, "Device Observer UID") |
|
ID for human observer not represented in SR; use nullFlavor="UNK" |
<section>: AuthorName |
(121008, DCM, "Person Observer Name") |
<section>: AuthorOrganization |
(121009, DCM, "Person Observer's Organization Name") |
<section>: AuthorDeviceModel |
(121015, DCM, "Device Observer Model Name") |
<section>: AuthorSoftware |
(121013, DCM, "Device Observer Name") |
- Standard -
DICOM PS3.20 2020a - Imaging Reports using HL7 Clinical Document Architecture |
Page 127 |
C.4.1.2 Comparison Study Procedure Context
TID 1005 Procedure Context allows identification of a different procedure than the procedure identified in the SR Study IE as the context for the section observations. In the transformations of this Annex, only an identified comparison procedure is supported as ProcedureContext,theSRsectionbeingtransformedmustbeeitherPriorProcedureDescriptionsorPreviousFindings,andtheCDA section shall be in accordance with the Comparison Study section Template 1.2.840.10008.9.4.
SR Instances using TID 2006 have additional attributes of a comparison procedure specified using TID 2007, which is used in the Prior Procedure Descriptions section. The attributes of both TID 1005 and TID 2007 are source data in the Table C.4-4 mapping.
Table C.4-4. Comparison Study mapping from SR
CDA Business Name |
DICOM SR |
ComparisonStudy: ProcedureTechnique: ProcedureCode |
(121023, DCM, "Procedure Code") |
ComparisonStudy: ProcedureTechnique: EffectiveTime |
(111060, DCM, "Study Date") + (111061, DCM, "Study Time") |
ComparisonStudy: ProcedureTechnique: Modality |
(122142, DCM, "Acquisition Device Type") |
ComparisonStudy: ProcedureTechnique: MethodCode |
|
ComparisonStudy: ProcedureTechnique: TargetSite |
(123014, DCM, "Target Region") |
ComparisonStudy: ProcedureTechnique: Laterality: |
|
ComparisonStudy: ProcedureTechnique: Ref: |
|
ComparisonStudy: ProcedureTechnique: ProviderOrganization |
|
ComparisonStudy: Study[*]: StudyUID |
(121018, DCM, "Procedure Study Instance UID") |
ComparisonStudy: Study[*]: Description |
(121065, DCM, "Procedure Description") , if present, or |
|
(121023, DCM, "Procedure Code") > Code Meaning |
|
(0008,0104) |
ComparisonStudy: Study[*]: Time |
(111060, DCM, "Study Date") + (111061, DCM, "Study Time") |
C.4.1.3 Fetus Subject Context
TID1006SubjectContextallowsidentificationofadifferentsubjectthanthepatientidentifiedintheSRPatientIE.Inthetransformations ofthisAnnex,onlyanidentifiedfetussubjectissupportedasSubjectContextforaFindingssection.AnSRsectionwithafetussubject context shall be mapped to a CDA section shall be in accordance with the Fetus Findings subsection Template 1.2.840.10008.9.9. This section is subsidiary to the top level Findings section; multiple SR fetus findings sections may be mapped to separate CDA Fetus Findings subsections.
Table C.4-5. CDA Fetus subject mapping from SR
CDA Business Name |
DICOM SR |
Findings: FetusFindings[*]: FetusID |
(121030, DCM, "Subject ID") or (11951-1, LN, "Fetus ID") |
C.4.2 Section/text
DICOM TID 2002 Report Narrative specifies that sections contain imaging report elements of type CODE, TEXT, IMAGE, or NUM.
Section/text in the CDA document contains the narrative text (attested content) of the document. Section/text shall be generated from all the Content Items subsidiary to a section CONTAINER of the SR document, such that the full meaning is be conveyed in an un- ambiguous manner in the narrative block.
The narrative rendered from each Content Item shall be encapsulated in a <content> element of the narrative block, allowing the as- sociated entry to reference it.
- Standard -

Page 128 |
DICOM PS3.20 2020a - Imaging Reports using HL7 Clinical Document Architecture |
C.4.3 Content Item Mapping
Each Content Item immediately subsidiary to a section CONTAINER shall be mapped to the corresponding entry level template, and shallbeincludedsubsidiarytotheassociatedCDAsectionorsubsection.Thisisinadditiontoitsrenderinginthesection/textnarrative block.
Coded concepts that are encoded in the SR using with the Coding Scheme Designator "SRT" shall be mapped to the equivalent SNOMED CT code. Mappings for value sets invoked in both SR and CDA are provided in PS3.16.
C.4.3.1 Coded Observations
SR CODE Content Items shall be mapped to Coded Observation entries.
Table C.4-6. CDA Coded Observation mapping from SR CODE
CDA Business Name |
DICOM SR |
CodedObservation[*]: ObsName |
Concept Name Code Sequence (0040,A043) |
CodedObservation[*]: ObsValue |
Concept Code Sequence (0040,A168) |
CodedObservation[*]: Time |
Observation DateTime (0040,A032) |
CodedObservation[*]: InterpretationCode |
|
CodedObservation[*]: ActionableFindingCode |
|
CodedObservation[*]: TargetSite |
(363698007, SCT, "Finding Site") |
CodedObservation[*]:Laterality |
(363698007, SCT, "Finding Site") > (272741003, SCT, |
|
"Laterality") |
CodedObservation[*]:TopoModifier |
|
CodedObservation[*]:Method |
|
CodedObservation[*]: SOPInstance |
[See C.4.3.3] |
CodedObservation[*]: QuantityMeasurement |
[See C.4.3.4] |
CodedObservation[*]: CodedObservation |
|
TheCODEobservationsinTID2002donotspecificallyincludefindingsite,laterality,andtopographicalmodifiers,butthesemodifiers are not forbidden in the template, and may be present in a SR SOP Instance being transformed to CDA.
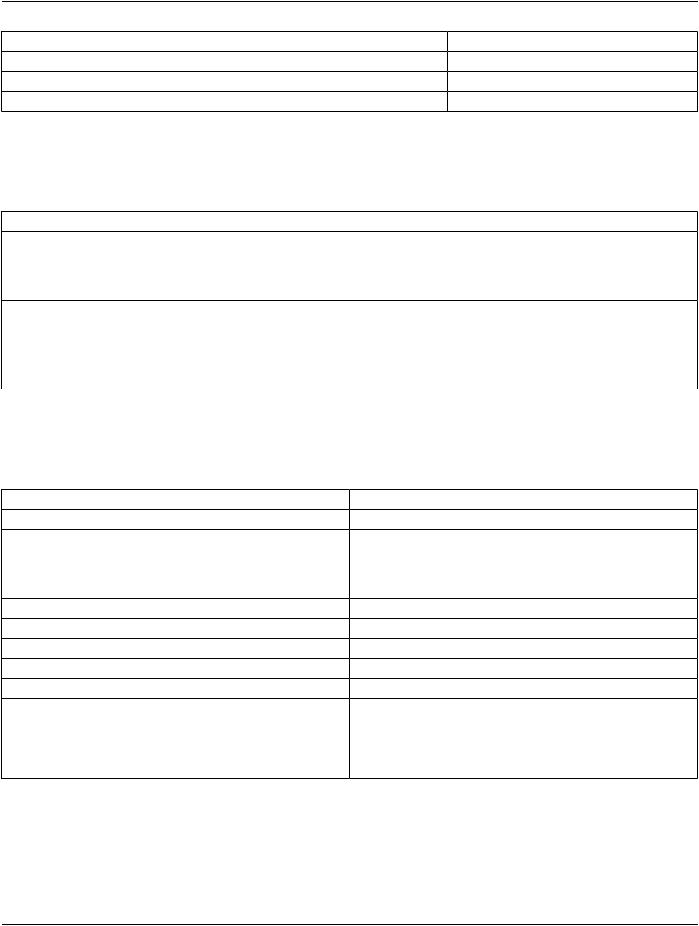
C.4.3.2 Text Observations
SR TEXT Content Items are mapped to Coded Observation entries, but the code is a nullFlavor with the text content in originalText.
Table C.4-7. CDA Coded Observation mapping from SR TEXT
CDA Business Name or XPath |
DICOM SR |
CodedObservation[*]: ObsName |
Concept Name Code Sequence (0040,A043) |
observation/value/@nullFlavor |
"NI" |
observation/value/originalText |
Text Value (0040,A160) |
CodedObservation[*]: Time |
Observation DateTime (0040,A032) |
CodedObservation[*]: InterpretationCode |
|
CodedObservation[*]: ActionableFindingCode |
|
CodedObservation[*]: TargetSite |
|
CodedObservation[*]:Laterality |
|
CodedObservation[*]:TopoModifier |
|
CodedObservation[*]:Method |
|
- Standard -
DICOM PS3.20 2020a - Imaging Reports using HL7 Clinical Document Architecture |
Page 129 |
||
CDA Business Name or XPath |
DICOM SR |
|
|
CodedObservation[*]: SOPInstance |
[See C.4.3.3] |
|
|
CodedObservation[*]: QuantityMeasurement |
[See C.4.3.4] |
|
|
CodedObservation[*]: CodedObservation |
|
|
|
C.4.3.3 Image Observations |
|
|
|
SR IMAGE Content Items shall be mapped to SOP Instance Observation entries. |
|
|
|
Table C.4-8. CDA SOP Instance Observation mapping from SR IMAGE |
|
||
CDA Business Name |
DICOM SR |
|
|
SOPInstance[*]:SOPInstanceUID |
Referenced SOP Sequence (0008,1199) > Referenced SOP Instance UID |
||
|
(0008,1155) |
|
|
SOPInstance[*]:SOPClassUID |
Referenced SOP Sequence (0008,1199) > Referenced SOP Class UID |
||
|
(0008,1150) |
|
|
SOPInstance[*]:WADOReference |
[WADOlinkconstructedfromimagereference;alsousedinlinkHtmlinnarrative |
||
|
block] |
|
|
SOPInstance[*]:PurposeOfReference |
Concept Name Code Sequence (0040,A043) |
|
|
SOPInstance[*]:ReferencedFrames |
Referenced SOP Sequence (0008,1199) > Referenced Frame Number |
||
|
(0008,1160) |
|
|
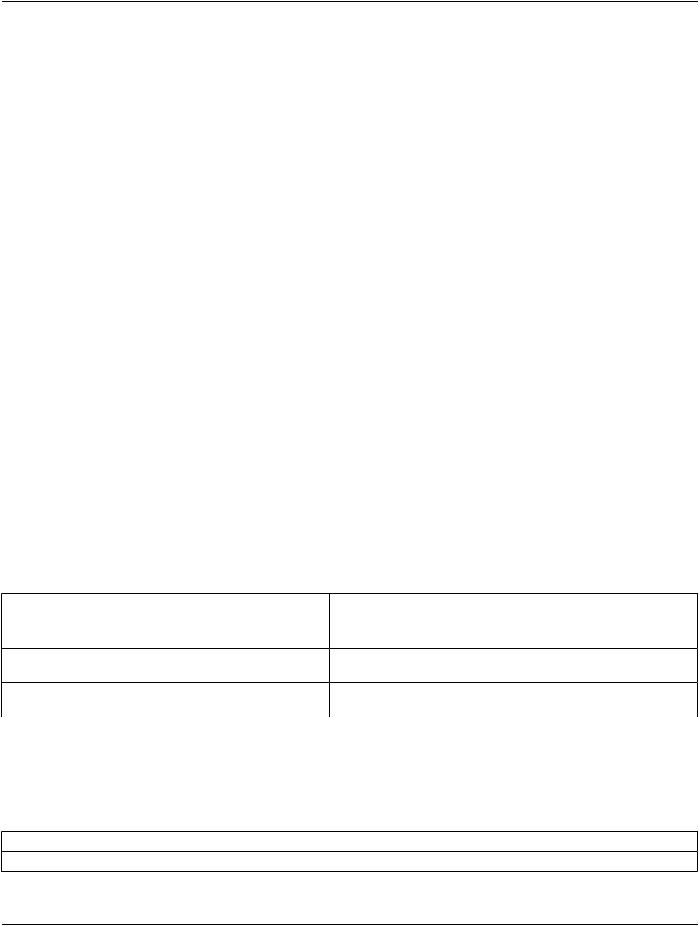
C.4.3.4 Numeric Observations
SR NUM Content Items shall be mapped to Quantity Measurement entries.
Table C.4-9. CDA Quantity Measurement mapping from SR NUM
CDA Business Name |
DICOM SR |
QuantityMeasurement[*]: MeasurementName |
Concept Name Code Sequence (0040,A043) |
QuantityMeasurement[*]: MeasurementValue |
Measured Value Sequence (0040,A300) > Numeric Value |
|
(0040,A30A) |
QuantityMeasurement[*]: MeasurementUnits |
Measured Value Sequence (0040,A300) > Measurement Units |
|
Code Sequence (0040,08EA) > Code Value (0008,0100) |
QuantityMeasurement[*]: Time |
Observation DateTime (0040,A032) |
QuantityMeasurement[*]: InterpretationCode |
|
QuantityMeasurement[*]: ActionableFindingCode |
|
QuantityMeasurement[*]: TargetSite |
(363698007, SCT, "Finding Site") |
QuantityMeasurement[*]:Laterality |
(363698007,SCT,"FindingSite")>(272741003,SCT,"Laterality") |
QuantityMeasurement[*]:Method |
(370129005, SCT, "Measurement Method") |
QuantityMeasurement[*]:TopoModifier |
(106233006, SCT, "Topographical modifier") |
QuantityMeasurement[*]: SOPInstance |
[See C.4.3.3] |
QuantityMeasurement[*]: QuantityMeasurement |
[See C.4.3.4] |
The SR templates invoked for NUM measurements from TID 2000 do not specifically include finding site, laterality, and topographical modifiers, but these modifiers are not forbidden in the template, they are used in many other NUM value templates (e.g., TID 300 Measurement), and may be present in a SR SOP Instance being transformed to CDA.
- Standard -
Page 130 |
DICOM PS3.20 2020a - Imaging Reports using HL7 Clinical Document Architecture |
C.4.3.5 Inferred From Image Observations
SR TID 2001 and TID 2002 allow Content Items to be INFERRED FROM IMAGE observations. The INFERRED FROM relationship ismappedtotheentryRelationshipwithtypeCode=SPRT,andtheIMAGEobservationismappedtoaCDASOPInstanceObservation entry subsidiary to its parent CDA Coded Observation or Quantity Measurement entry. This entryRelationship is shown in the Coded Observation and Quantity Measurement CDA Templates.
C.4.3.6 Inferred From Numeric Observations
SR TID 2001 and TID 2002 allow Content Items to be INFERRED FROM NUM observations. The INFERRED FROM relationship is mapped to the entryRelationship with typeCode=SPRT, and the NUM observation is mapped to CDA Quantity Measurement entry subsidiary to its parent CDA Coded Observation or Quantity Measurement entry. This entryRelationship is shown in the Coded Ob- servation and Quantity Measurement CDA Templates.
C.4.3.7 Inferred From Spatial Coordinates Observations
SR TID 1400, TID 1401, TID 1402, and TID 1404 allow NUM Content Items to be INFERRED FROM SCOORD observations, which areSELECTEDFROMIMAGEobservations.ThisAnnexdoesnotspecifythetransformationforSCOORDobservations;thesewould use the CDA Region Of Interest entry, which PS3.20 forbids (see Section 9.1.2.4).
C.4.4 Specific Section Content Mapping
Certain sections in a CDA Imaging Report have specific mappings from the DICOM SR header, or from specialized templates with content for particular uses.
C.4.4.1 Procedure Indications
The DICOM SR Document General Module may specify the Reason for the Requested Procedure as either free text in attribute (0040,1002), and/or as multiple coded values in attribute (0040,100A). These are mapped to the Procedure Indications subsection of the Clinical Information section of the CDA Imaging Report.
Note
ProcedureindicationsmayalsobespecifiedasSRcontentitemsinthe(18785-6,LN,"IndicationsforProcedure")CONTAINER, which may be mapped to the CDA instance in accordance with Section C.4.3. It is an implementation decision how to handle multiple representations of indications in the SR document.
Table C.4-10. Clinical Information Procedure Indications mapping from SR
CDA Business Name |
DICOM SR |
ClinicalInformation: ProcedureIndications: Text |
Referenced Request Sequence (0040,A370) > Reason for the |
|
Requested Procedure (0040,1002) |
ClinicalInformation: ProcedureIndications: |
(432678004, SNOMED, "Indication for procedure") |
CodedObservation[*]: ObsName |
|
ClinicalInformation: ProcedureIndications: |
Referenced Request Sequence (0040,A370) > Reason for the |
CodedObservation[*]: ObsValue |
Requested Procedure Code Sequence (0040,100A) |
C.4.4.2 Current Procedure Descriptions
SR Instances using TID 2006 have a Current Procedure Descriptions section specified using TID 2007. Source data in that template and from the General Study Module is mapped into the CDA Procedure Description section.
Table C.4-11. Current Procedure Description mapping from SR
CDA Business Name |
DICOM SR |
ProcedureDescription: ProcedureTechnique: ProcedureCode |
Procedure Code Sequence (0008,1032) |
- Standard -