Материал: part20
Page 96 |
DICOM PS3.20 2020a - Imaging Reports using HL7 Clinical Document Architecture |
|
||||||
Business |
Nest |
Element/ |
Card |
Elem/Attr Data |
Value |
Value |
Subsidiary |
|
Name |
Level Attribute |
|
Conf |
Type |
Conf |
|
Template |
|
Actionable |
>> |
translation |
0..1 |
MAY |
CD |
MAY |
ValueSet CID 7035 |
|
Priority |
|
|
|
|
|
CWE |
“Actionable Finding |
|
|
|
|
|
|
|
|
Classification” |
|
|
|
|
|
|
|
|
[See 10.1.3 |
|
|
|
|
|
|
|
|
interpretationCode and |
|
|
|
|
|
|
|
|
translation For Actionable |
|
|
|
|
|
|
|
|
Findings] |
|
TargetSite |
> |
targetSiteCode1..1 |
COND |
CD |
|
ConceptDomain |
|
|
|
|
|
|
|
|
|
ObservationSite |
|
|
>> |
qualifier |
0..1 |
COND |
|
|
|
|
|
>>> |
name |
1..1 |
SHALL |
CD |
SHALL (272741003, SNOMED |
|
|
|
|
|
|
|
|
|
CT, "laterality") |
|
Laterality |
>>> |
value |
1..1 |
SHALL |
CD |
SHALL ValueSet CID 244 |
|
|
|
|
|
|
|
|
CNE |
“Laterality” |
|
|
>> |
qualifier |
0..1 |
COND |
|
|
|
|
|
>>> |
name |
1..1 |
SHALL |
CD |
SHALL (106233006, SNOMED |
|
|
|
|
|
|
|
|
|
CT, "topographical |
|
|
|
|
|
|
|
|
modifier") |
|
TopoModifier |
>>> |
value |
1..1 |
SHALL |
CD |
SHALL ValueSetCID2“Anatomic |
||
|
|
|
|
|
|
CNE |
Modifier” |
|
Method |
> |
methodCode 0..1 |
MAY |
CD |
|
ConceptDomain |
|
|
|
|
|
|
|
|
|
ObservationMethod |
|
|
> |
entry |
0..* |
MAY |
|
|
|
|
|
|
Relationship |
|
|
|
|
|
|
|
>@ |
@typeCode |
1..1 |
SHALL |
CS |
SHALL SPRT |
|
|
SOPInstance[*]>> |
observation |
1..1 |
SHALL |
|
|
|
10.8 SOP Instance |
|
|
|
|
|
|
|
|
|
Observation |
|
|
|
|
|
|
|
|
1.2.840.10008.9.18 |
|
> |
entry |
0..* |
MAY |
|
|
|
|
|
|
Relationship |
|
|
|
|
|
|
|
>@ |
@typeCode |
1..1 |
SHALL |
CS |
SHALL SPRT |
|
|
Quantity |
> |
observation |
1..1 |
SHALL |
|
|
|
10.5 Quantity |
Measurement[*] |
|
|
|
|
|
|
Measurement |
|
|
|
|
|
|
|
|
|
2.16.840.1.113883. |
|
|
|
|
|
|
|
|
10.20.6.2.14 |
|
> |
entry |
0..* |
MAY |
|
|
|
|
|
|
Relationship |
|
|
|
|
|
|
|
>@ |
@typeCode |
1..1 |
SHALL |
CS |
SHALL SUBJ |
|
|
Coded |
> |
observation |
1..1 |
SHALL |
|
|
|
10.1 Coded |
Observation[*] |
|
|
|
|
|
|
|
Observation |
|
|
|
|
|
|
|
|
2.16.840.1.113883. |
|
|
|
|
|
|
|
|
10.20.6.2.13 |
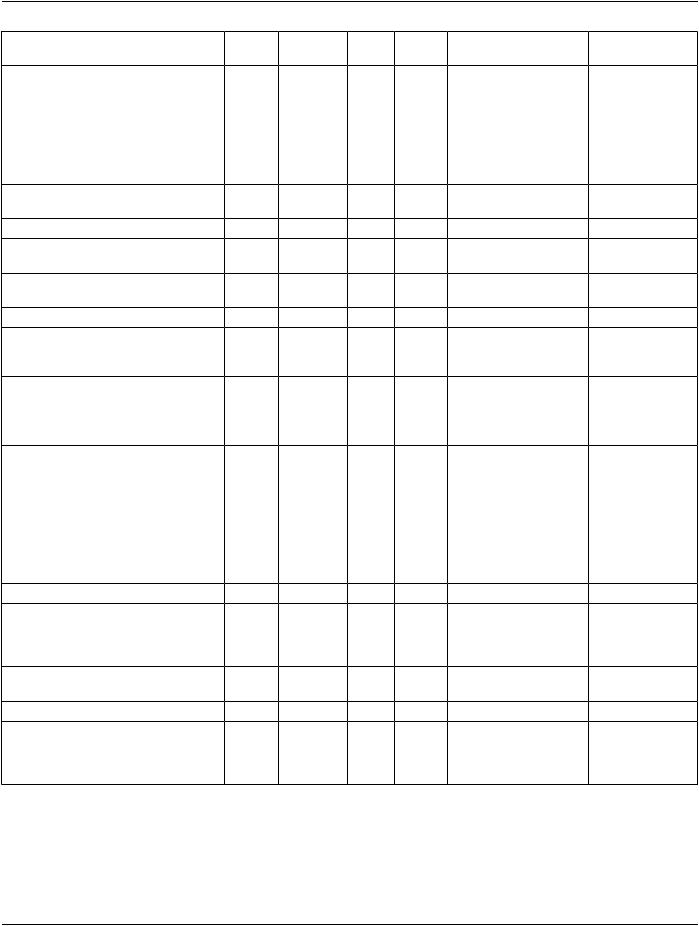
10.1.1 code and @negationInd
TheObservationcodeelementhasanassociatedConceptDomainObservationType.ArepresentativebindingforthisConceptDomain is to the value (ASSERTION, actcode[2.16.840.1.113883.5.4], "Assertion"), providing an assertion of a finding concept in the value element.
- Standard -

DICOM PS3.20 2020a - Imaging Reports using HL7 Clinical Document Architecture |
Page 97 |
The Observation may have @negationInd attribute "true", which together with the code "ASSERTION" indicates that the finding was not observed, e.g., to represent "No finding of stroke".
Note
This is the pattern used in Consolidated CDA for negative findings.
10.1.2 text/reference and Related Narrative Block Markup
The Observation entry SHOULD include a text/reference element, whose value attribute (not to be confused with the value element oftheObservationclass)SHALLbeginwitha'#'andSHALLpointtoitscorrespondingnarrativeintheparentsection(usingtheapproach defined in CDA Release 2, section 4.3.5.1). See Section 9.1.1.1.
10.1.3 interpretationCode and translation For Actionable Findings
When an observation is unexpected or "actionable" (one type of which is denoted a "critical finding"), it may be flagged using the in- terpretationCode.ForveryabnormalfindingstheinterpretationCodeelementSHALLbesetto(AA,ObservationInterpretation,"abnormal alert"). Unexpected normal findings, e.g., no findings of disease when patient treatment had been planned on the presumption of disease, may also be flagged using interpretationCode (N, ObservationInterpretation, "normal").
The translation element of the interpretationCode may be used to provide a further classification as defined in a regionallyor profes- sionally-specified value set. This template identifies an optional value set for the ACR Actionable Finding categories 1, 2, and 3, as defined by: Larson PA, et al. J Am Coll Radiol 2014; published online. DOI 10.1016/j.jacr.2013.12.016.
The narrative text associated with the actionable finding SHOULD be highlighted using styleCode Bold. See Section 9.5.1 and Sec- tion 9.1.1.1.
Actionable findings that require a specific follow-up action or procedure SHOULD be referenced from a recommendation in the 9.8.11 Recommendation section.
Communication of actionable findings SHOULD be documented in the 9.8.10 Communication of Actionable Findings section.
10.1.4 targetSiteCode
Each observation needs to fully specify its site/location.
COND: If the observation site is not pre-coordinated in the observation/code or observation/value, it SHALL be specified in the obser- vation/targetSiteCode.
COND: The qualifier element for laterality SHALL be present if the targetSiteCode represents a paired body part and laterality is not pre-coordinated in the targetSiteCode.
Note that inclusion in a labeled subsection (see Section 9.8.9) does not imply a finding site for the observation from the title. The title is not semantically part of the post-coordination.
10.1.5 entryRelationship/@typeCode=SUBJ/observation - Coded
TheCodedObservationentryMAYincludeanactRelationshipoftypeSUBJ(hassubject)toasubsidiaryCodedObservation(recursively invoking this same template). This allows the constructions of complex clinical statements.
Example 10.1-1. Coded observation example
<text>
...
<content ID="fnd-1"> ...finding of a right hilar mass (abnormal - class 1) ...</content> </text>
...
<entry>
<observation classCode="OBS" moodCode="EVN">
- Standard -

Page 98 |
DICOM PS3.20 2020a - Imaging Reports using HL7 Clinical Document Architecture |
<templateId root="2.16.840.1.113883.10.20.6.2.13"/> <id root="1.2.840.10213.2.62.7044779.114265201"/>
<code code="ASSERTION" codeSystem="2.16.840.1.113883.5.4" codeSystemName="actCode"
displayName="Assertion"/> <text><reference value="#fnd-1"/></text> <statusCode code="completed"/> <effectiveTime value="20140914171504+0500"/> <value xsi:type="CD" code="309530007"
codeSystem="2.16.840.1.113883.6.96" codeSystemName="SNOMED CT" displayName="Hilar mass"/>
<interpretationCode code = "AA" codeSystem="2.16.840.1.113883.5.83" codeSystemName="ObservationInterpretation" displayName="Abnormal Alert">
<translation code="RID49480" codeSystem="2.16.840.1.113883.6.256" codeSystemName="RADLEX"
displayName="ACR Category 1 Actionable Finding"/> </interpretationCode>
<!-- although "hilar mass" is by definition in the lung, the observation.value does not describe right or left lung, so targetSite is required -->
<targetSiteCode code="3341006" codeSystem="2.16.840.1.113883.6.96" codeSystemName="SNOMED CT" displayName="right lung">
</targetSiteCode>
<!-- entryRelationship elements referring to SOP Instance Observations or Quantity Measurement Observations may appear here -->
</observation>
</entry>
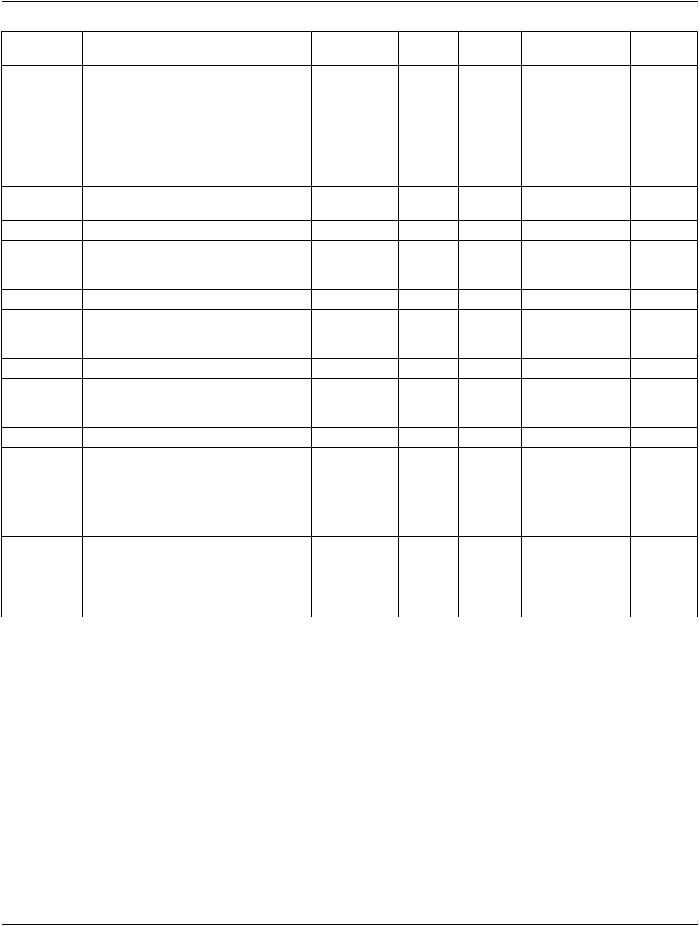
10.2 Procedural Medication
Template ID |
1.2.840.10008.9.13 |
Name |
Procedural Medication |
Effective Date |
2015/03/24 |
Version Label |
DICOM-20150324 |
Status |
Active |
Description |
Procedural medication describes a substance administration that has actually |
|
occurred prior to or during a procedure (e.g., imaging contrast/agents, |
|
anti-histamines, anti-anxiety, beta blockers to control heart rate during procedure, |
|
etc.). |
Classification |
CDA Entry Level |
Relationships |
Included in 9.3 Imaging Procedure Description |
Context |
parent node |
Open/Closed |
Open |
Revision History |
DICOM-20150324: Initial version |
Business |
Nest |
Element/Attribute Card |
Elem/AttrConf Data |
Value |
Value |
Subsidiary |
|
Name |
Level |
|
|
Type |
Conf |
|
Template |
Procedural |
|
substance |
1..1 |
SHALL |
|
|
|
Medication[*] |
|
Administration |
|
|
|
|
|
orContrast[*] |
|
|
|
|
|
|
|
- Standard -
|
DICOM PS3.20 2020a - Imaging Reports using HL7 Clinical Document Architecture |
Page 99 |
||||||
Business |
Nest |
Element/Attribute Card |
Elem/AttrConf Data |
Value |
Value |
Subsidiary |
||
Name |
Level |
|
|
|
Type |
Conf |
|
Template |
|
@ |
@classCode |
1..1 |
SHALL |
CS |
SHALL |
SBADM |
|
|
@ |
@moodCode |
1..1 |
SHALL |
CS |
SHALL |
EVN |
|
|
> |
templateId |
1..1 |
SHALL |
II |
|
|
|
|
>@ |
@root |
1..1 |
SHALL |
UID |
SHALL |
1.2.840.10008.9.13 |
|
|
> |
id |
1..1 |
SHALL |
II |
|
|
|
|
> |
text |
0..1 |
SHOULD |
ED |
|
|
|
Ref |
>> |
reference |
0..1 |
SHOULD |
URL(XML |
#contentRef |
|
|
|
|
|
|
|
IDREF) |
|
|
|
|
> |
statusCode |
1..1 |
SHALL |
CS |
SHALL |
COMPLETED |
|
Route |
> |
routeCode |
0..1 |
MAY |
CE |
SHOULDValueSet CID 11 |
|
|
|
|
|
|
|
|
CWE |
“Route of |
|
|
|
|
|
|
|
|
Administration” |
|
Dose |
> |
doseQuantity |
0..1 |
SHOULD |
PQ |
|
|
|
DoseUnit |
>@ |
@unit |
0..1 |
SHOULD |
|
SHALL |
ValueSet CID 82 |
|
|
|
|
|
|
|
CNE |
“Units of |
|
|
|
|
|
|
|
|
Measurement” |
|
Rate |
> |
rateQuantity |
0..1 |
MAY |
PQ |
|
|
|
RateUnit |
>@ |
@unit |
1..1 |
SHALL |
CS |
SHALL |
ValueSet CID 82 |
|
|
|
|
|
|
|
CNE |
“Units of |
|
|
|
|
|
|
|
|
Measurement” |
|
|
> |
consumable |
1..1 |
SHALL |
|
|
|
|
|
>> |
manufactured |
1..1 |
SHALL |
|
|
|
|
|
|
Product |
|
|
|
|
|
|
|
>>@ |
@classCode |
1..1 |
SHALL |
CS |
SHALL |
MANU |
|
|
>>> |
manufactured |
1..1 |
SHALL |
|
|
|
|
|
|
Material |
|
|
|
|
|
|
CodedProduct>>>> |
code |
1..1 |
SHALL |
CE |
|
ConceptDomain |
|
|
Name |
|
|
|
|
|
|
MedContrastName |
|
|
|
|
|
|
|
|
||
FreeText |
>>>>> |
original Text |
0..1 |
SHOULD |
ED |
|
|
|
ProductName |
|
|
|
|
|
|
|
|
10.2.1 Business Name Alias
This template defines a primary scoping business name "ProceduralMedication" and an alias "Contrast". This allows production logic to use either term, although the structure is identical.
10.2.2 text/reference and Related Narrative Block Markup
The substanceAdministration entry SHOULD include a text/reference element, whose value attribute SHALL begin with a '#' and SHALL point to its corresponding narrative in the parent section (using the approach defined in CDA Release 2, section 4.3.5.1). See Section 9.1.1.1.
10.2.3 doseQuantity
•Pre-coordinated consumable: If the consumable code is a pre-coordinated unit dose (e.g., "metoprolol 25mg tablet") then doseQuantity is a unitless number that indicates the number of products given per administration (e.g., "2", meaning 2 x "metoprolol 25mg tablet").
- Standard -

Page 100 |
DICOM PS3.20 2020a - Imaging Reports using HL7 Clinical Document Architecture |
•Not pre-coordinated consumable: If the consumable code is not pre-coordinated (e.g., is simply "metoprolol"), then doseQuantity must represent a physical quantity with @unit, e.g., "25" and "mg", specifying the amount of product given per administration.
Example 10.2-1. Procedural Medication activity example
<substanceAdministration classCode="SBADM" moodCode="EVN"> <templateId root="1.2.840.10008.9.13"/>
<id root="cdbd33f0-6cde-11db-9fe1-0800200c9a66"/> <text>
<reference value="#med1"/> </text>
<statusCode code="completed"/>
<routeCode code="47625008" codeSystem="2.16.840.1.113883.6.96" codeSystemName="SNOMED CT" displayName="intravenous route"/>
<doseQuantity value="100" unit="ml"/> <consumable>
<manufacturedProduct classCode="MANU">
<templateId root="2.16.840.1.113883.10.20.22.4.23"/> <id/>
<manufacturedMaterial> <code code="412372002"
codeSystem="2.16.840.1.113883.6.96" codeSystemName="SNOMED CT" displayName="Meglumine Diatrizoate">
<originalText>
<reference value="#manmat1"/> </originalText>
<translation code="3320"
codeSystem="2.16.840.1.113883.6.88" codeSystemName="RxNorm" displayName="Diatrizoate Meglumine"/>
</code>
</manufacturedMaterial>
</manufacturedProduct>
</consumable>
</substanceAdministration>
10.3 observationMedia
Template ID |
1.3.6.1.4.1.19376.1.4.1.4.7 |
Name |
observationMedia Entry |
Effective Date |
2011-07 |
Version Label |
IHECIRC-TI |
Status |
Active |
Description |
The observationMedia Entry provides an in-line graphic depiction of the section |
|
findings.Itisreferencedbya<renderMultiMedia>elementinthesectiontext.Typical |
|
usesareforgraphicrepresentationoffindings(e.g.,arterialtreediagrams)orin-line |
|
representations of key images. |
Classification |
CDA Entry Level |
Relationships |
|
Context |
parent node |
Open/Closed |
Open |
- Standard -