Page 1638 |
DICOM PS3.3 2020a - Information Object Definitions |
C.36.6.1.5 Parent RT Prescription
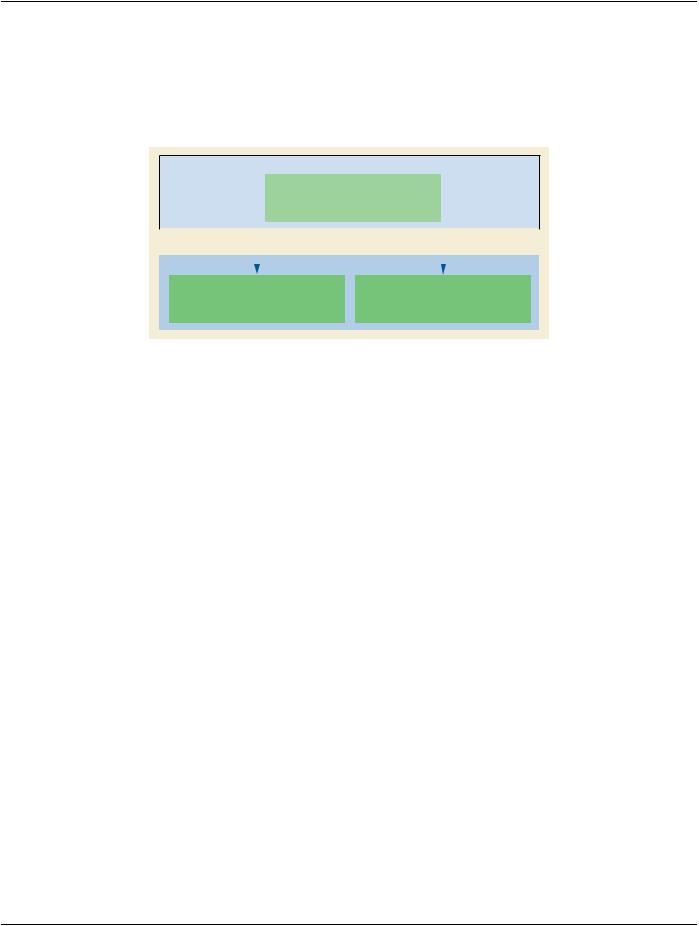
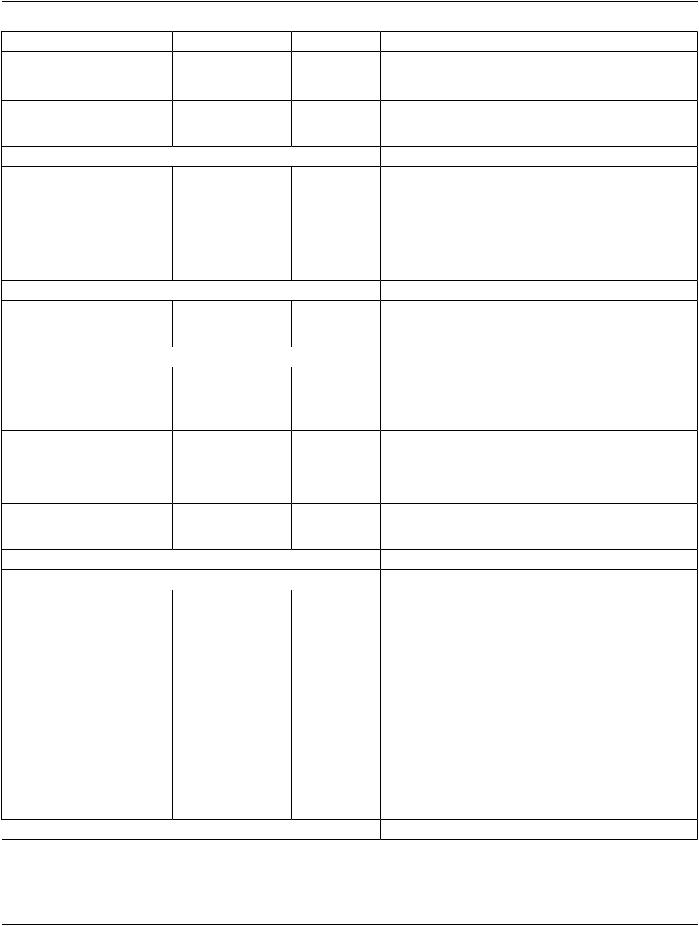
TheRTEnhancedPrescriptionModulesupportsatwo-levelprescriptionmodel(seeFigureC.36.6.1-2)inordertodocumentanapproach where a high-level prescription is created and then more details may be added. The first level would typically be created during the time of prescription definition with only high-level prescription information based on the RT Physician Intent. The second level, if present, would represent a derived prescription containing more detailed information, such as constraints and objectives. A third or subsequent levels are not permitted.
Parent Prescription:
Right Oropharynx Cancer Treatment
•Curative
•Target Dose:: 80 Gy
•Two Phase with FIeld Reduction
|
|
|
|
|
|
|
|
|
|
|
|
Child Prescriptions: |
|
|
|
Right Oropharynx and Right Neck Nodal Volumes |
Right Oropharynx PTV only |
• Target Dose: 28 x 2 Gy = 56 Gy |
• Target Dose: 7 x 2 Gy = 14 Gy |
• IMRT 9 FIelds |
• IMRT Field Boost |
• Photons 6X |
• Photons 6X |
|
|
|
|
Figure C.36.6.1-2. Parent and Child RT Prescriptions
C.36.6.1.6 Dosimetric Objective Sequence
The Dosimetric Objective Sequence (3010,006C) specifies a set of intended dosimetric goals.
Each item of the Dosimetric Objective Sequence (3010,006C) shall be referenced by at least one item of the Referenced Dosimetric Objectives Sequence (3010,0071) in the current Instance.
WithinanRTPhysicianIntentSOPInstance,aDosimetricObjectiveisapplicabletoallPrescriptionsinwhichtheDosimetricObjective UID (3010,006E) is referenced.
A Dosimetric Objective is intended to be satisfied by the combined effect of all treatments associated with prescriptions that reference
the Dosimetric Objective UID (3010,006F).
For example, if there are primary and boost prescriptions that reference the same Dosimetric Objective UID (3010,006E) then the combined effect of both prescriptions is intended to comply with the limit in this Dosimetric Objective.
C.36.6.1.7 Dosimetric Objective Evaluation Scope
For Dosimetric Objectives that include a dose value, the value defines the total dose for all fractions of all prescriptions referencing this Dosimetric Objective, and potentially dose from previous treatment, depending on the value of Dosimetric Objective Evaluation Scope (3010,0063).
If Dosimetric Objective Evaluation Scope (3010,0063) has a value of LIFETIME, information from prior treatments shall be included in the evalution of the Dosimetric Objective.
For example, if an organ has received dose in a prior treatment, and Dosimetric Objective Evaluation Scope (3010,0063) is marked as LIFETIME, then the previously delivered dose shall be included in the evalution of this objective. Information about prior treatments may be described in Prior Treatment Dose Description (3010,0061) or by Instances referenced by the Prior Treatment Reference Sequence (3010,0062) in RT Prescription Sequence (3010,006B).
IfDosimetricObjectiveEvaluationScope(3010,0063)hasavalueofCURRENT,theobjectiveincludesonlythoseprescriptionswhich reference the same Dosimetric Objective, even if information of a prior treatment is available.