Материал: nm1391
|
T E C H N I C A L R E P O R T S |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||||||||||
|
a 20 |
|
|
|
|
b 6,000 |
|
|
|
c |
|
–6,000 |
|
|
|
|
d 2.0 |
|
|
|
|
|
|
|
|
||||||
|
|
|
|
* |
|
|
|
|
|
|
|
|
*†‡ |
|
|
|
|
|
|
†‡ |
AWT diastole (mm) |
1.6 |
|
|
|
|
|
|
|
|
|
|
|
15 |
|
|
* |
|
Max dP/dt (mmHg/s) |
|
|
|
|
* |
Min dP/dt (mmHg/s) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||
|
LVEDP (mmHg) |
|
|
|
4,000 |
|
* |
|
–4,000 |
|
|
* |
|
|
|
|
|
|
|
|
|
|
|||||||||
|
|
|
|
|
|
|
|
|
|
* |
|
1.2 |
|
|
*†‡§ |
|
|
|
Sham |
||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Untreated |
||||||||
|
10 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
DFB |
|
||||||
|
|
|
|
|
*†‡ |
|
|
|
|
|
|
|
|
|
|
|
|
0.8 |
* |
|
* |
|
|
|
|
||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
MSC |
||||||||||
|
|
|
|
|
2,000 |
|
|
|
|
–2,000 |
|
|
|
|
|
|
|
|
|
||||||||||||
|
5 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
0.4 |
* * |
|
* |
|
|
|
|
|
||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||||||||
|
|
0 |
|
|
|
|
|
|
0 |
|
|
|
|
|
|
0 |
|
|
|
|
|
0 |
|
|
|
|
|
|
|
|
|
http://www.nature.com/naturemedicine |
|
|
Sham |
ntreated |
DFB |
MSC |
|
|
|
Sham |
ntreated |
DFB |
MSC |
|
|
|
Sham |
ntreated |
DFB |
MSC |
|
Baseline |
After treatment |
|
|
|
|
||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||||||||
|
|
U |
|
|
|
|
|
U |
|
|
|
|
|
U |
|
|
|
|
|
|
|
|
|
|
|
|
|||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||
e |
11 |
|
|
|
*§ |
f |
50 |
|
|
|
g |
|
|
2,000 |
|
|
|
*§ |
h |
Coronary ligation |
Transplantation |
|
|
||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
100 |
|
|
|
|
|
|
|
|
|||||||
|
10 |
* |
|
*§ |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||||
|
|
|
40 |
|
|
|
|
(pg/ml) |
1,500 |
|
|
|
|
|
80 |
|
|
|
|
|
|
|
|
||||||||
|
|
* * |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
MSC group |
|||||||||
|
9 |
|
*†‡ |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||||||||||
(mm) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(n = 20) |
|||||||||
|
|
30 |
|
|
|
|
level |
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
60 |
|
|
|
|
|
|
|
|
|||||||
8 |
|
|
|
|
|
|
|
|
|
|
*†‡ |
1,000 |
* |
|
|
*§ |
|
|
|
|
|
|
|
|
|
||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||||||||||
LVDD |
|
|
|
|
|
20 |
|
* |
|
Plasma ANP |
|
|
|
40 |
|
|
|
|
|
|
|
|
|||||||||
7 |
|
|
|
|
|
|
|
* |
|
|
|
|
|
|
|
|
|
|
|
Untreated group |
|||||||||||
|
|
|
|
|
|
|
|
|
|
500 |
* * |
|
|
*†‡ |
|
|
|
|
|
|
|
||||||||||
6 |
|
|
|
§ |
|
10 |
* * |
|
* |
|
|
|
20 |
|
|
|
|
|
|
(n = 20) |
|||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||||||||||||||
|
5 |
|
|
|
|
|
0 |
|
|
|
|
|
|
0 |
|
|
|
|
|
0 |
|
|
|
|
|
|
|
|
|||
|
Baseline |
After treatment |
|
Baseline |
After treatment |
|
|
Baseline |
After treatment |
|
0 |
1 |
2 |
3 |
4 |
5 |
6 |
7 |
8 |
||||||||||||
|
|
|
|
|
|
|
|
|
|||||||||||||||||||||||
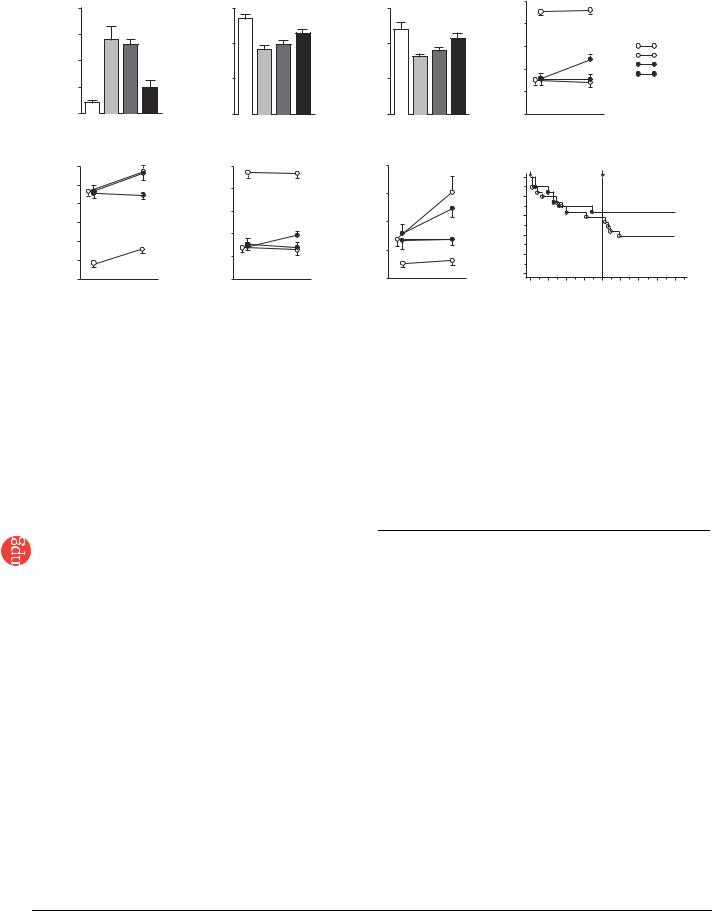
Figure 5 Cardiac structure and function afterPercentageFS |
transplantation of monolayered MSCs. (a–c) Hemodynamic parametersSurvivalrate(percent) |
|
|
Time (weeks) |
|
|
|
||||||||||||||||||||||||
obtained by catheterization. LVEDP, left |
|||||||||||||||||||||||||||||||
Group |
ventricle end-diastolic pressure. (d–f) Echocardiographic findings. AWT, anterior wall thickness; LVDD, left ventricle end-diastolic dimension; FS, fractional |
||||||||||||||||||||||||||||||
shortening. (g) Plasma atrial natriuretic peptide (ANP) level. Baseline represents measurements 4 weeks after coronary ligation; ’after treatment’ represents |
|||||||||||||||||||||||||||||||
measurements taken 4 weeks after transplantation (8 weeks after coronary ligation). Data are mean ± s.e.m. *P o 0.05 versus sham group; wP o 0.05 |
|||||||||||||||||||||||||||||||
versus untreated group; zP o 0.05 versus DFB group; yP o 0.05 versus baseline. (h) Survival of rats with chronic heart failure with or without monolayered |
|||||||||||||||||||||||||||||||
Publishing |
|||||||||||||||||||||||||||||||
MSC transplantation. The Kaplan-Meier survival curve demonstrates an 8-week survival rate of 65% for the MSC group versus 45% for the untreated group. |
|||||||||||||||||||||||||||||||
Survival rate after transplantation was significantly higher in the MSC group than in the untreated group (100% versus 71% 4-week survival rate after |
|||||||||||||||||||||||||||||||
transplantation, log-rank test, P o 0.05). |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||
Nature |
conditions at 37 1C and becomes reversibly hydrophilic below 32 1C. Therefore, |
|
COMPETING INTERESTS STATEMENT |
|
|
|
|
|
|
|
|||||||||||||||||||||
cultured cells that adhere to the dish surface spontaneously detach from the |
|
The authors declare competing financial interests (see the Nature Medicine website |
|||||||||||||||||||||||||||||
grafted surface without enzymatic digestion. |
|
|
|
|
|
|
|
for details). |
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||
2006 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||||||||||||
Preparation of monolayered cell grafts. We suspended MSCs at the third or |
|
Published online at http://www.nature.com/naturemedicine/ |
|
|
|
|
|||||||||||||||||||||||||
© |
fourth |
passage from adipose |
tissue or DFBs at the second passage by |
|
Reprints and permissions information is available online at http://npg.nature.com/ |
||||||||||||||||||||||||||
|
trypsinization, and plated the cell suspension containing 3 ml of complete |
|
reprintsandpermissions/ |
|
|
|
|
|
|
|
|
|
|
||||||||||||||||||
|
medium onto a 60-mm temperature-responsive dish at 5 105 cells per dish |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||||||||||||||
|
(MSCs) or 8 105 cells per dish (DFBs) and cultured cells at 37 1C. After 3 d of |
|
1. |
Liu, J. et al. Autologous stem cell transplantation for myocardial repair. Am. J. Physiol. |
|||||||||||||||||||||||||||
|
culture, confluently cultured MSCs or DFBs on the temperature-responsive |
|
|
Heart Circ. Physiol. 287, H501–H511 (2004). |
|
|
|
|
|
|
|||||||||||||||||||||
|
dishes were incubated at 20 1C. By 40 min, both MSCs and DFBs detached |
|
2. |
Reinlib, L. & Field, L. Cell transplantation as future therapy for cardiovascular disease?: |
|||||||||||||||||||||||||||
|
spontaneously and floated up into the medium as monolayered cell grafts. |
|
|
A workshop of the National Heart, Lung, and Blood Institute. Circulation 101, E182– |
|||||||||||||||||||||||||||
|
Immediately after detachment, we gently aspirated the monolayered cell grafts |
|
|
E187 (2000). |
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||
|
|
3. Schuster, M.D. et al. Myocardial neovascularization by bone marrow angioblasts results |
|||||||||||||||||||||||||||||
|
using a 1,000 ml pipette tip and transferred them onto an elastic plastic sheet. |
|
|||||||||||||||||||||||||||||
|
|
|
in cardiomyocyte regeneration. Am. J. Physiol. Heart Circ. Physiol. 287, H525–H532 |
||||||||||||||||||||||||||||
|
Statistical analysis. Numerical values are expressed as mean ± s.e.m. There are |
|
|
(2004). |
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||
|
|
4. |
Kocher, A.A. et al. Neovascularization of ischemic myocardium by human bone- |
||||||||||||||||||||||||||||
|
four groups of continuous variables in this study. Therefore, for multiple |
|
|
marrow-derived angioblasts prevents cardiomyocyte apoptosis, reduces remodeling |
|||||||||||||||||||||||||||
|
comparisons of more than two groups, we performed one-way analysis of |
|
|
and improves cardiac function. Nat. Med. 7, 430–436 (2001). |
|
|
|
||||||||||||||||||||||||
|
variance (ANOVA). If the ANOVA was significant, we used the Newman-Keul |
|
5. Bel, A. et al. Transplantation of autologous fresh bone marrow into infarcted myocar- |
||||||||||||||||||||||||||||
|
|
|
dium: a word of caution. Circulation 108, II247–II252 (2003). |
|
|
|
|||||||||||||||||||||||||
|
procedure as a post hoc test. For repeated measurement such as echocardio- |
|
|
|
|
|
|||||||||||||||||||||||||
|
|
6. |
Yamada, N. et al. Thermo-responsive polymeric surface: control of attachment and |
||||||||||||||||||||||||||||
|
graphic parameters, we performed two-way repeated ANOVA with the |
|
|
detachment of cultured cells. Makromol. Chem. Rapid Commun. 11, 571–576 |
|||||||||||||||||||||||||||
|
Newman-Keul test. Comparisons of parameters between two groups were made |
|
|
(1990). |
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||
|
by unpaired Student t-test. A value of P o 0.05 was considered significant. |
|
|
7. Okano, T., Yamada, H., Sakai, H. & Sakurai, Y. A novel recovery system for cultured |
|||||||||||||||||||||||||||
|
|
|
|
cells using plasma-treated polystyrene dishes grafted with poly (N-isopropylacryla- |
|||||||||||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||
|
Note: Supplementary information is available on the Nature Medicine website. |
|
|
|
mide). J. Biomed. Mater. Res. 27, 1243–1251 (1993). |
|
|
|
|
|
|||||||||||||||||||||
|
|
|
8. |
Shimizu, T. et al. Fabrication of pulsatile cardiac tissue grafts using a novel 3- |
|||||||||||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
dimensional cell sheet manipulation technique and temperature-responsive cell |
||||||||||||||
|
ACKNOWLEDGMENTS |
|
|
|
|
|
|
|
|
|
|
|
culture surfaces. Circ. Res. 90, e40–e48 (2002). |
|
|
|
|
|
|
||||||||||||
|
We thank J.I. Hoffman for his statistical advice. We thank T. Iwase, T. Ito, S. |
|
|
9. |
Hirose, M., Kwon, O.H., Yamato, M., Kikuchi, A. & Okano, T. Creation of designed |
||||||||||||||||||||||||||
|
Murakami, N. Sakata and Y. Isono for their technical support. We thank Y. Tsuboi |
|
|
shape cell sheets that are noninvasively harvested and moved onto another surface. |
|||||||||||||||||||||||||||
|
|
|
Biomacromolecules 1, 377–381 (2000). |
|
|
|
|
|
|
|
|||||||||||||||||||||
|
and H. Sonoda for their assistance with microscopic analysis of monolayered cell |
|
|
|
|
|
|
|
|
|
|
||||||||||||||||||||
|
|
|
10. Kushida, A. et al. Decrease in culture temperature releases monolayer endothelial cell |
||||||||||||||||||||||||||||
|
grafts. We also thank Y. Sawa for his suggestions on this study. This work was |
|
|
||||||||||||||||||||||||||||
|
|
|
|
sheets together with deposited fibronectin matrix from temperature-responsive culture |
|||||||||||||||||||||||||||
|
supported by research grants for Cardiovascular Disease (16C-6) and Human |
|
|
|
|||||||||||||||||||||||||||
|
|
|
|
surfaces. J. Biomed. Mater. Res. 45, 355–362 (1999). |
|
|
|
|
|
||||||||||||||||||||||
|
Genome Tissue Engineering 005 and 009 from the Japanese Ministry of Health, |
|
|
|
|
|
|
|
|
||||||||||||||||||||||
|
|
|
11. Herreros, J. et al. Autologous intramyocardial injection of cultured skeletal muscle- |
||||||||||||||||||||||||||||
|
Labor and Welfare, and the Program for Promotion of Fundamental Studies in |
|
|
|
derived stem cells in patients with non-acute myocardial infarction. Eur. Heart J. 24, |
||||||||||||||||||||||||||
|
Health Science of the Japanese National Institute of Biomedical Innovation. |
|
|
|
2012–2020 (2003). |
|
|
|
|
|
|
|
|
|
|
||||||||||||||||
|
4 6 4 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
VOLUME 12 [ NUMBER 4 [ |
APRIL 2006 |
NATURE MEDICINE |
||||||||||

T E C H N I C A L R E P O R T S
© 2006 Nature Publishing Group http://www.nature.com/naturemedicine
12.Skobel, E. et al. Transplantation of fetal cardiomyocytes into infarcted rat hearts results in long-term functional improvement. Tissue Eng. 10, 849–864 (2004).
13.Hodgson, D.M. et al. Stable benefit of embryonic stem cell therapy in myocardial infarction. Am. J. Physiol. Heart Circ. Physiol. 287, H471–H479 (2004).
14.Makino, S. et al. Cardiomyocytes can be generated from marrow stromal cells in vitro.
J.Clin. Invest. 103, 697–705 (1999).
15.Pittenger, M.F. et al. Multilineage potential of adult human mesenchymal stem cells. Science 284, 143–147 (1999).
16.Reyes, M. et al. Origin of endothelial progenitors in human postnatal bone marrow.
J.Clin. Invest. 109, 337–346 (2002).
17.Toma, C., Pittenger, M.F., Cahill, K.S., Byrne, B.J. & Kessler, P.D. Human mesenchymal stem cells differentiate to a cardiomyocyte phenotype in the adult murine heart. Circulation 105, 93–98 (2002).
18.Wang, J.S. et al. Marrow stromal cells for cellular cardiomyoplasty: feasibility and potential clinical advantages. J. Thorac. Cardiovasc. Surg. 120, 999–1005 (2000).
19.Jiang, Y. et al. Pluripotency of mesenchymal stem cells derived from adult marrow. Nature 418, 41–49 (2002).
20.Nagaya, N. et al. Transplantation of mesenchymal stem cells improves cardiac function in a rat model of dilated cardiomyopathy. Circulation 112, 1128–1135 (2005).
21.Rangappa, S., Fen, C., Lee, E.H., Bongso, A. & Wei, E.S. Transformation of adult mesenchymal stem cells isolated from the fatty tissue into cardiomyocytes. Ann. Thorac. Surg. 75, 775–779 (2003).
22.Zuk, P.A. et al. Human adipose tissue is a source of multipotent stem cells. Mol. Biol. Cell 13, 4279–4295 (2002).
23.Gaustad, K.G., Boquest, A.C., Anderson, B.E., Gerdes, A.M. & Collas, P. Differentiation of human adipose tissue stem cells using extracts of rat cardiomyocytes. Biochem. Biophys. Res. Commun. 314, 420–427 (2004).
24.Planat-Benard, V. et al. Plasticity of human adipose lineage cells toward endothelial cells: physiological and therapeutic perspectives. Circulation 109, 656–663 (2004).
25.Lee, R.H. et al. Characterization and expression analysis of mesenchymal stem cells from human bone marrow and adipose tissue. Cell. Physiol. Biochem. 14, 311–324 (2004).
26.Li, J., Takaishi, K., Cook, W., McCorkle, S.K. & Unger, R.H. Insig-1 ‘‘brakes’’ lipogenesis in adipocytes and inhibits differentiation of preadipocytes. Proc. Natl. Acad. Sci. USA 100, 9476–9481 (2003).
27.Vande Berg, J.S., Rudolph, R. & Woodward, M. Comparative growth dynamics and morphology between cultured myofibroblasts from granulating wounds and dermal fibroblasts. Am. J. Pathol. 114, 187–200 (1984).
28.Nishida, K. et al. Corneal reconstruction with tissue-engineered cell sheets composed of autologous oral mucosal epithelium. N. Engl. J. Med. 351, 1187–1196 (2004).
29.Shimizu, T., Yamato, M., Kikuchi, A. & Okano, T. Cell sheet engineering for myocardial tissue reconstruction. Biomaterials 24, 2309–2316 (2003).
30.Nishikimi, T., Uchino, K. & Frohlich, E.D. Effects of a1-adrenergic blockade on intrarenal hemodynamics in heart failure rats. Am. J. Physiol. Regul. Integr. Comp. Physiol. 262, R198–R203 (1998).
NATURE MEDICINE VOLUME 12 [ NUMBER 4 [ APRIL 2006 |
4 6 5 |