Материал: 37_herricks2011
Published on 11 July 2011. Downloaded by Universita Degli Studi di Napoli Federico II on 18/07/2013 15:37:23.
|
|
|
|
|
|
|
|
|
View Article Online |
|||
in the micro-channels, the cross-sectional area of the channel |
the overall pressure drop across the channel or the overall flow |
|
||||||||||
decreases, resulting in unequal flow through different sections of |
rate through the channel based on an infusion pump. |
|
|
|||||||||
the device. A method to estimate the local wall shear stress was |
|
|
|
|
|
|
|
|
|
|
|
|
developed using erythrocytes as particles for tracking fluid |
Rolling velocity versus wall shear stress |
|
|
|
|
|||||||
velocities. Particle image velocimetry (PIV) has been performed |
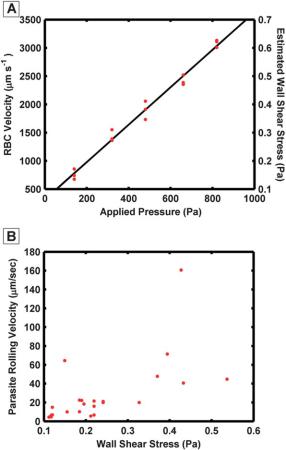
A calibration plot showed that the average velocity of erythro- |
|||||||||||
using the erythrocytes to estimate the local flow environment.30,31 |
||||||||||||
PIV is measured using specialized high-speed cameras, laser light |
cytes increased with |
the measured pressure |
drop across |
the |
||||||||
channels (Fig. 4A). The range of velocities in the calibration plot |
||||||||||||
sources, and image correlation methods to describe the velocity |
||||||||||||
covers |
the velocities |
observed |
in experiments at a constant |
|||||||||
field across the field of view. In this study, an average velocity |
||||||||||||
applied pressure. Reduced flow rates with higher pressure are due |
||||||||||||
estimation was used. The average velocity method uses conven- |
||||||||||||
to endothelial cells growing in the channels and reducing the |
||||||||||||
tional optics and cameras and is more appropriate for field use. |
||||||||||||
effective cross-sectional area. Erythrocyte velocities of 5–20 mm s 1 |
||||||||||||
The average velocity method does not resolve the parabolic |
||||||||||||
have been observed in capillaries, which translates to wall shear |
||||||||||||
velocity field across the width of the micro-channel. Therefore, |
||||||||||||
stresses range of 0.1 to 0.7 kPa.32,33 These wall shear stresses are |
||||||||||||
this method is restricted in areas where flow is relatively laminar |
||||||||||||
in the same range observed in the mF-D described here.34–37 The |
||||||||||||
and unperturbed by obstructions. The method provides a useful |
||||||||||||
rolling velocity |
of |
individual |
parasitized |
erythrocytes |
was |
|||||||
estimate of local velocities when particles are present in at least |
||||||||||||
observed |
over |
a |
variety of wall shear |
stresses (Fig. 4B). As |
||||||||
two consecutive images across the field of view. Over the velocity |
||||||||||||
expected, |
the |
rolling |
velocity increased |
with |
the applied |
wall |
||||||
range measured, the average velocity appears to be linearly |
||||||||||||
shear stress and at higher shear stresses fewer parasitized eryth- |
||||||||||||
related to the pressure applied across the channel (Fig. 4A). This |
||||||||||||
rocytes tended to adhere. The relatively few number of parasit- |
||||||||||||
technique provides a reasonable estimate of the local flow rate |
||||||||||||
ized |
erythrocytes observed |
rolling |
on |
endothelial |
cells |
|||||||
through the channels, and is superior to relying on a measure of |
||||||||||||
underscores the difficulty and unique characteristics of working |
||||||||||||
|
||||||||||||
|
with fresh parasite field isolates. Normally for similar experi- |
|||||||||||
|
ments in non-endemic countries, parasite cultures are selected to |
|||||||||||
|
enrich for expression of adhesive characteristics before binding |
|||||||||||
|
experiments are performed. In an effort to keep the microfluidic |
|||||||||||
|
system as close to physiologic conditions as possible, a field |
|||||||||||
|
parasite isolate was chosen to directly demonstrate that the |
|||||||||||
|
techniques described capture parasite-endothelial cell interac- |
|||||||||||
|
tions over a variety of flow conditions. While a detailed investi- |
|||||||||||
|
gation across multiple parasite isolates and primary brain |
|||||||||||
|
endothelial cells is not presented here, the present work |
|||||||||||
|
demonstrates that these microfluidic technologies are ready for |
|||||||||||
|
field applications. |
|
|
|
|
|
|
|
||||
|
Conclusion |
|
As the parasitized erythrocytes accumulate in the microcircula- |
|
tion it is important to understand the conditions under which |
|
they cytoadhere and how they migrate under various flow |
|
conditions. The mF-CS described here was developed for field |
|
experimentation to observe parasitized erythrocyte cytoadhesion |
|
to primary endothelial cells. Quantifying the rolling behavior of |
|
parasitized erythrocytes over a variety of shear stresses can help |
|
describe the behavior of parasitized cells in micro-circulation. |
|
These types of measurements could help future investigations |
|
into interactions between endothelial cells and parasitized |
|
erythrocytes. This report demonstrates that microfluidic systems |
|
can be utilized to perform experiments in a malaria-endemic |
|
area. Such a system can mimic the micro-circulatory conditions |
|
in the deep capillary beds of organs and may improve our |
|
understanding of malaria pathogenesis. The mF-CS and image |
|
analysis tools described here provide a promising new resource |
|
for investigating how cytoadhesion contributes to severe malarial |
Fig. 4 The mean erythrocyte velocity increased linearly with the applied |
infections. |
pressure drop across the device (A). The wall shear stress was estimated |
|
using the erythrocyte velocity and the depth of field of the objective lens. |
Acknowledgements |
The rolling velocity of parasitized erythrocytes’s increased as the esti- |
|
mated wall shear stress increased (B). Each dot indicates an individual |
This work was supported by the NIH under the following |
parasitized erythrocyte. |
grants R21 AI081234 (P.K.R.), K23AI079402 (K.B.S), and |
|
|
This journal is ª The Royal Society of Chemistry 2011 |
Lab Chip, 2011, 11, 2994–3000 | 2999 |
Published on 11 July 2011. Downloaded by Universita Degli Studi di Napoli Federico II on 18/07/2013 15:37:23.
|
|
|
View Article Online |
||
U19AI089688 (P.K.R.). We specifically thank Jason Stage, Dave |
15 |
M. Frank, R. Dzikowski, B. Amulic and K. Deitsch, Mol. Microbiol., |
|
||
Tucker and the late George Turner of Games4you LLC for |
|
2007, 64, 1486–1498. |
|||
16 |
D. J. Roberts, A. G. Craig, A. R. Berendt, R. Pinches, G. Nash, |
||||
developing software for the microcontroller pump. |
|||||
|
K. Marsh and C. I. Newbold, Nature, 1992, 357, 689–692. |
||||
|
|
|
|||
|
|
17 |
M. Antia, T. Herricks and P. K. Rathod, PLoS Pathog., 2007, 3, e99. |
||
References |
18 |
J. P. Shelby, J. White, K. Ganesan, P. K. Rathod and D. T. Chiu, |
|||
|
|
|
Proc. Natl. Acad. Sci. U. S. A., 2003, 100, 14618–14622. |
||
1 |
L. H. Miller, D. I. Baruch, K. Marsh and O. K. Doumbo, Nature, |
19 |
T. Herricks, M. Antia and P. K. Rathod, Cell Microbiol., 2009. |
||
|
2002, 415, 673–679. |
20 |
B. M. Cooke, S. Usami, I. Perry and G. B. Nash, Microvasc. Res., |
||
2 |
L. H. Miller, S. Usami and S. Chien, J. Clin. Invest., 1971, 50, 1451– |
|
1993, 45, 33–45. |
||
|
1455. |
21 |
T. D’Amico Oblak, P. Root and D. M. Spence, Anal. Chem., 2006, 78, |
||
3 |
A. M. Dondorp, E. Pongponratn and N. J. White, Acta Trop., 2004, |
|
3193–3197. |
||
|
89, 309–317. |
22 |
W. Karunarathne, C. J. Ku and D. M. Spence, Integr. Biol., 2009, 1, |
||
4 |
H. A. Cranston, C. W. Boylan, G. L. Carroll, S. P. Sutera, |
|
655–663. |
||
|
J. R. Williamson, I. Y. Gluzman and D. J. Krogstad, Science, 1984, |
23 |
D. H. Kotsis and D. M. Spence, Anal. Chem., 2003, 75, 145–151. |
||
|
223, 400–403. |
24 |
D. M. Spence, N. J. Torrence, M. L. Kovarik and R. S. Martin, |
||
5 |
A. M. Dondorp, C. Ince, P. Charunwatthana, J. Hanson, A. van |
|
Analyst, 2004, 129, 995–1000. |
||
|
Kuijen, M. A. Faiz, M. R. Rahman, M. Hasan, E. Bin Yunus, |
25 |
S. Usami, H. H. Chen, Y. Zhao, S. Chien and R. Skalak, Ann. |
||
|
A. Ghose, R. Ruangveerayut, D. Limmathurotsakul, K. Mathura, |
|
Biomed. Eng., 1993, 21, 77–83. |
||
|
N. J. White and N. P. Day, J. Infect. Dis., 2008, 197, 79–84. |
26 |
J. M. Rosano, N. Tousi, R. C. Scott, B. Krynska, V. Rizzo, |
||
6 |
I. Ljunstrom, H. Perlmann, M. Schlichthere, A. Scherf and M. |
|
B. Prabhakarpandian, K. Pant, S. Sundaram and M. F. Kiani, |
||
|
Wahlgren, ed., Methods in Malaria Research, Manassas, Virginia, |
|
Biomed. Microdevices, 2009. |
||
|
2004. |
27 |
S. Chung, R. Sudo, P. J. Mack, C. R. Wan, V. Vickerman and |
||
7 |
B. M. Cooke, A. R. Berendt, A. G. Craig, J. MacGregor, |
|
R. D. Kamm, Lab Chip, 2009, 9, 269–275. |
||
|
C. I. Newbold and G. B. Nash, Br. J. Haematol., 1994, 87, 162–170. |
28 |
W. Trager and J. B. Jensen, Science, 1976, 193, 673–675. |
||
8 |
G. B. Nash, B. M. Cooke, K. Marsh, A. Berendt, C. Newbold and |
29 |
Z. Wang, M. C. Kim, M. Marquez and T. Thorsen, Lab Chip, 2007, 7, |
||
|
J. Stuart, Blood, 1992, 79, 798–807. |
|
740–745. |
||
9 |
C. J. McCormick, A. Craig, D. Roberts, C. I. Newbold and |
30 |
Y. Sugii, S. Nishio and K. Okamoto, Ann. N. Y. Acad. Sci., 2002, 972, |
||
|
A. R. Berendt, J. Clin. Invest., 1997, 100, 2521–2529. |
|
331–336. |
||
10 |
C. Newbold, P. Warn, G. Black, A. Berendt, A. Craig, B. Snow, |
31 |
Y. Sugii, S. Nishio and K. Okamoto, Physiol. Meas., 2002, 23, 403– |
||
|
M. Msobo, N. Peshu and K. Marsh, Am. J. Trop. Med. Hyg., 1997, |
|
416. |
|
|
|
57, 389–398. |
32 |
C. M. Rovainen, T. A. Woolsey, N. C. Blocher, D. B. Wang and |
||
11 |
K. R. Hughes, G. A. Biagini and A. G. Craig, Molecular and |
|
O. F. Robinson, J. Cereb. Blood Flow Metab., 1993, 13, 359–371. |
||
|
biochemical parasitology, 169, pp. 71–78. |
33 |
A. C. Ngai and H. R. Winn, Am. J. Physiol., 1996, 270, H1712–1717. |
||
12 |
S. J. Chakravorty, K. R. Hughes and A. G. Craig, Biochem. Soc. |
34 |
M. Oshima, T. Kobayashi and K. Takagi, Ann. N. Y. Acad. Sci., 2002, |
||
|
Trans., 2008, 36, 221–228. |
|
972, 337–344. |
||
13 |
D. J. Bridges, J. Bunn, J. A. van Mourik, G. Grau, R. J. Preston, |
35 |
H. H. Lipowsky, S. Kovalcheck and B. W. Zweifach, Circ. Res., 1978, |
||
|
M. Molyneux, V. Combes, J. S. O’Donnell, B. de Laat and |
|
43, 738–749. |
||
|
A. Craig, Blood, 115, pp. 1472–1474. |
36 |
P. Ganesan, S. He and H. Xu, Microvasc. Res., 80, pp. 99–109. |
||
14 |
J. D. Smith and A. G. Craig, Curr. Issues Mol. Biol., 2005, 7, 81–93. |
37 |
P. Ganesan, S. He and H. Xu, Ann. Biomed. Eng., 38, pp. 1566–1585. |
||
3000 | Lab Chip, 2011, 11, 2994–3000 |
This journal is ª The Royal Society of Chemistry 2011 |